Photosynthesis Supramolecular Chemistry UAF 2005 Photosynthesis Where does













- Slides: 24

Photosynthesis Supramolecular Chemistry, UAF, 2005

Photosynthesis Where does the energy come from that sustains all life? The Sun!! Well most of it anyway…… Supramolecular Chemistry, UAF, 2005

Respiration vs. Photosynthesis and respiration as complementary processes in the living world. Photosynthesis uses the energy of sunlight to produce sugars and other organic molecules. These molecules in turn serve as food. Respiration is a process that uses O 2 and forms CO 2 from the same carbon atoms that had been taken up as CO 2 and converted into sugars by photosynthesis. In respiration, organisms obtain the energy that they need to survive. Photosynthesis preceded respiration on the earth for probably billions of years before enough O 2 was released to create an atmosphere rich in oxygen. (The earth's atmosphere presently contains 20% O 2. ) Supramolecular Chemistry, UAF, 2005

Cellular Respiration -vs- Photosynthesis Supramolecular Chemistry, UAF, 2005

Overview of Photosynthesis 6 CO 2 + 12 H 2 O C 6 H 12 O 6 + 6 O 2 + 6 H 2 O DG = + 686 kcal/mol • Photosynthesis, like cellular respiration, is a series of redox reactions! What is being oxidized and what is being reduced in the summarizing equation above? Supramolecular Chemistry, UAF, 2005

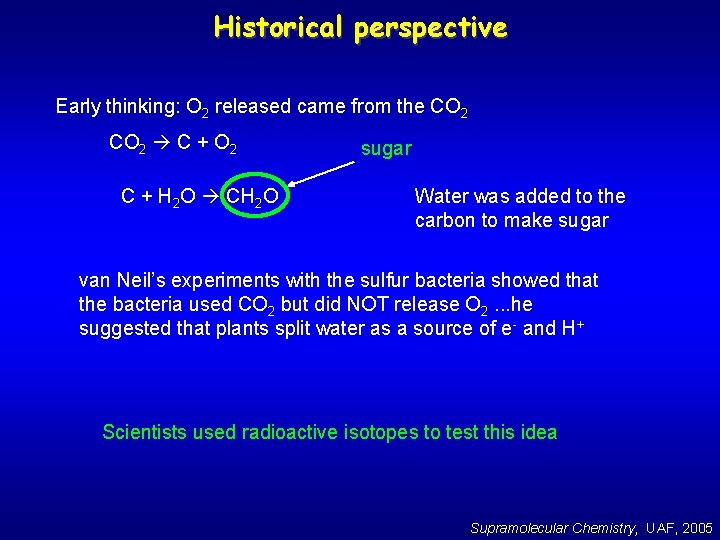
Historical perspective Early thinking: O 2 released came from the CO 2 C + O 2 C + H 2 O CH 2 O sugar Water was added to the carbon to make sugar van Neil’s experiments with the sulfur bacteria showed that the bacteria used CO 2 but did NOT release O 2. . . he suggested that plants split water as a source of e- and H+ Scientists used radioactive isotopes to test this idea Supramolecular Chemistry, UAF, 2005

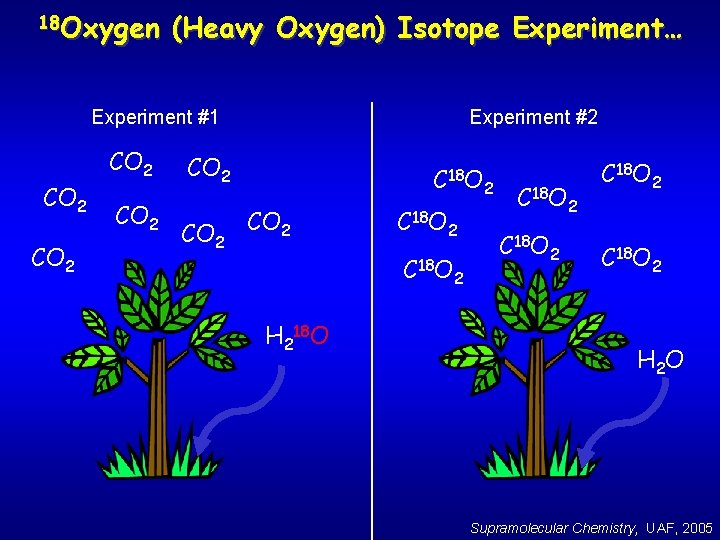
18 Oxygen (Heavy Oxygen) Isotope Experiment… Experiment #1 CO 2 Experiment #2 CO 2 C 18 O H 218 O 2 2 C 18 O 2 H 2 O Supramolecular Chemistry, UAF, 2005

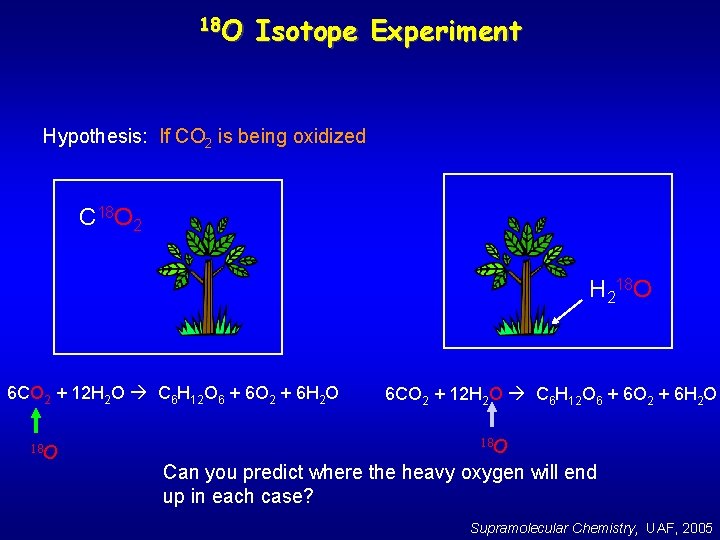
18 O Isotope Experiment Hypothesis: If CO 2 is being oxidized C 18 O 2 H 218 O 6 CO 2 + 12 H 2 O C 6 H 12 O 6 + 6 O 2 + 6 H 2 O 18 O Can you predict where the heavy oxygen will end up in each case? Supramolecular Chemistry, UAF, 2005

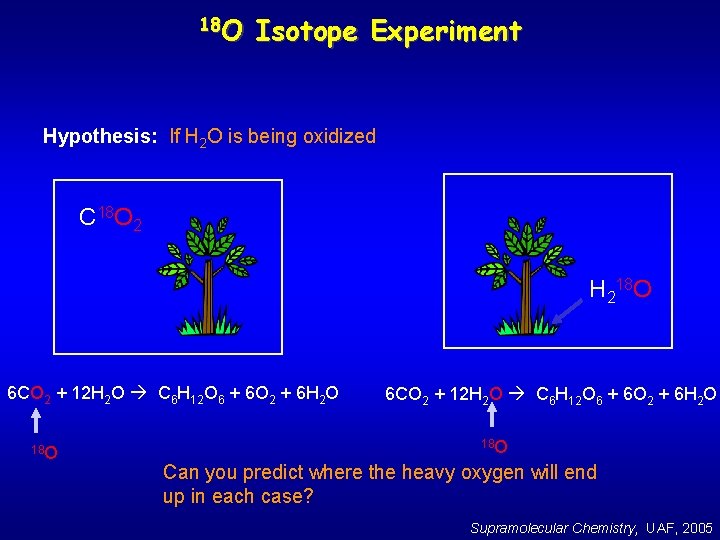
18 O Isotope Experiment Hypothesis: If H 2 O is being oxidized C 18 O 2 H 218 O 6 CO 2 + 12 H 2 O C 6 H 12 O 6 + 6 O 2 + 6 H 2 O 18 O Can you predict where the heavy oxygen will end up in each case? Supramolecular Chemistry, UAF, 2005

The Actual Results!! C 18 O 2 H 218 O 6 CO 2 + 12 H 2 O C 6 H 12 O 6 + 6 O 2 + 6 H 2 O 18 O So. . it’s the water that’s being oxidized! Supramolecular Chemistry, UAF, 2005

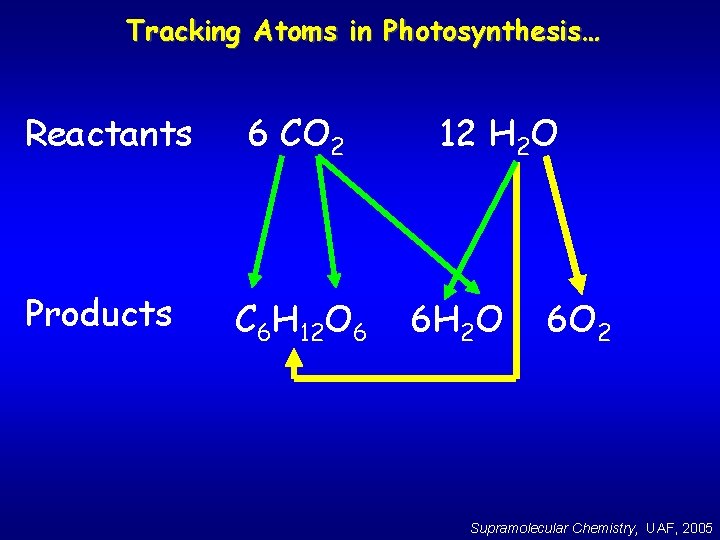
Tracking Atoms in Photosynthesis… Reactants 6 CO 2 Products C 6 H 12 O 6 12 H 2 O 6 O 2 Supramolecular Chemistry, UAF, 2005

What does this suggest about photosynthesis? The sugars that result from photosynthesis are produced by adding the hydrogen ions and electrons from water to carbon dioxide, NOT by splitting CO 2 and adding water! oxidized H+ and e- 6 CO 2 + 12 H 2 O C 6 H 12 O 6 + 6 O 2 + 6 H 2 O reduced Supramolecular Chemistry, UAF, 2005

Plant Photosynthesis internal leaf structure outer membrane chloroplasts inner membrane thylakoid Supramolecular Chemistry, UAF, 2005

The stages of photosynthesis… Light Dependent Reactions • Occurs along the thylakoid membrane • this is where H 2 O is oxidized. • chemiosmotic production of ATP! • electron carrier (NADPH) is synthesized Supramolecular Chemistry, UAF, 2005

The stages of photosynthesis… Light Independent Reactions • Occurs in the stroma of the chloroplast. • Where CO 2 is “fixed” into sugars! Supramolecular Chemistry, UAF, 2005

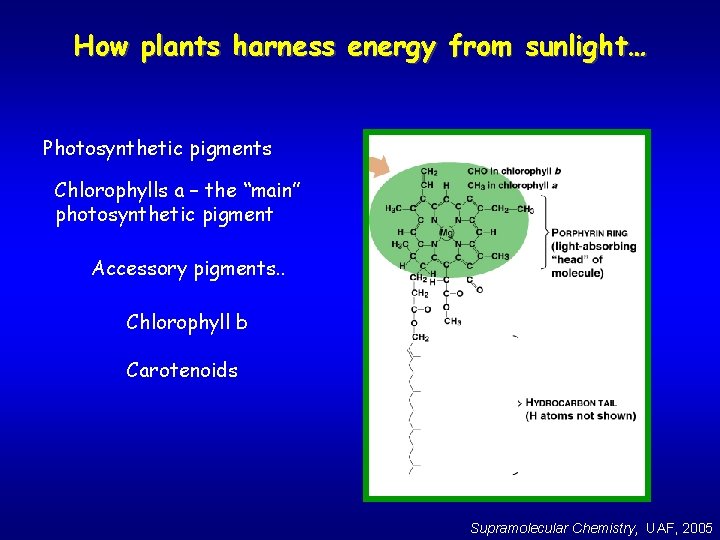
How plants harness energy from sunlight… Photosynthetic pigments Chlorophylls a – the “main” photosynthetic pigment Accessory pigments. . Chlorophyll b Carotenoids Supramolecular Chemistry, UAF, 2005

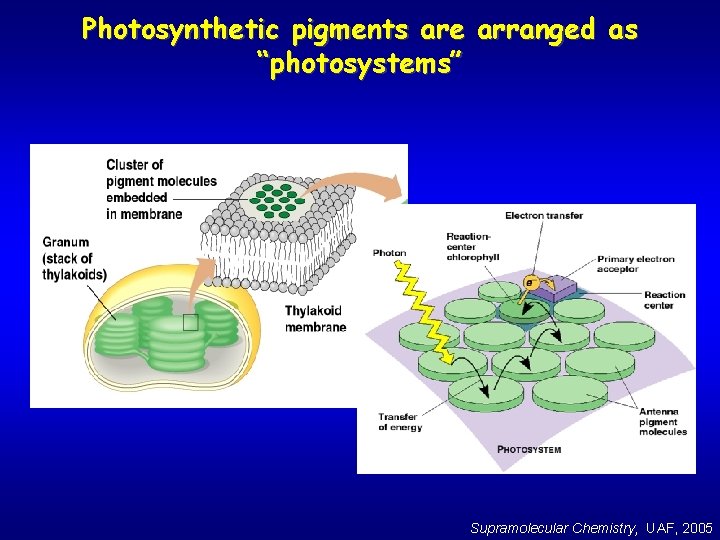
Photosynthetic pigments are arranged as “photosystems” Supramolecular Chemistry, UAF, 2005

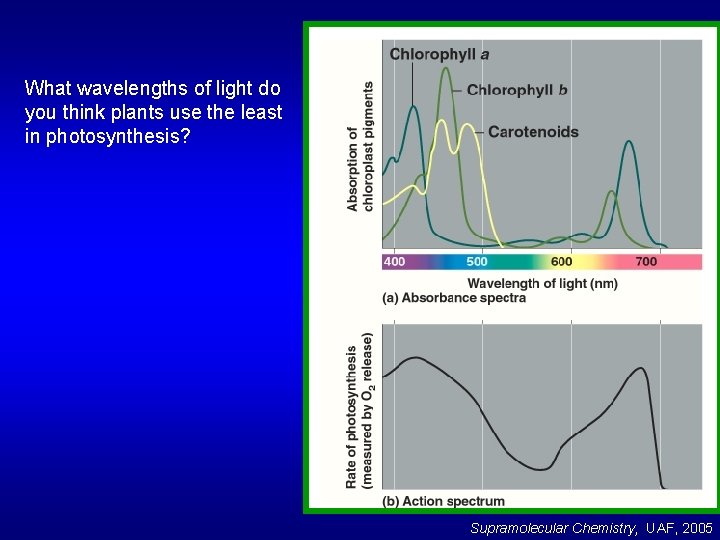
What wavelengths of light do you think plants use the least in photosynthesis? Supramolecular Chemistry, UAF, 2005

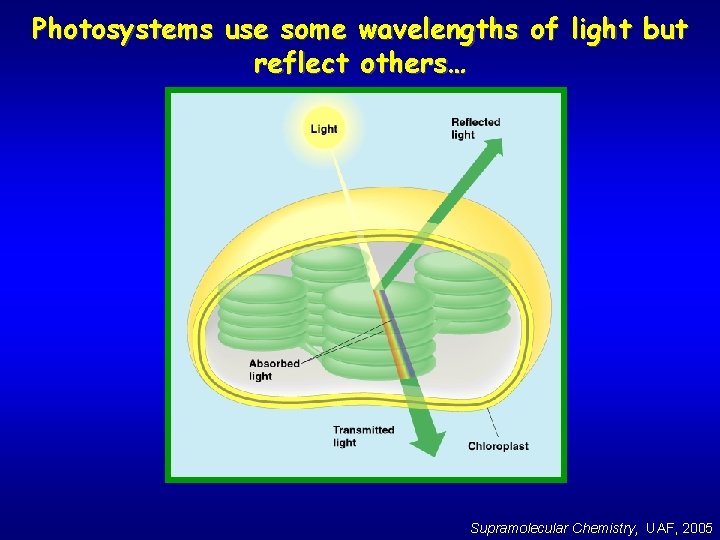
Photosystems use some wavelengths of light but reflect others… Supramolecular Chemistry, UAF, 2005

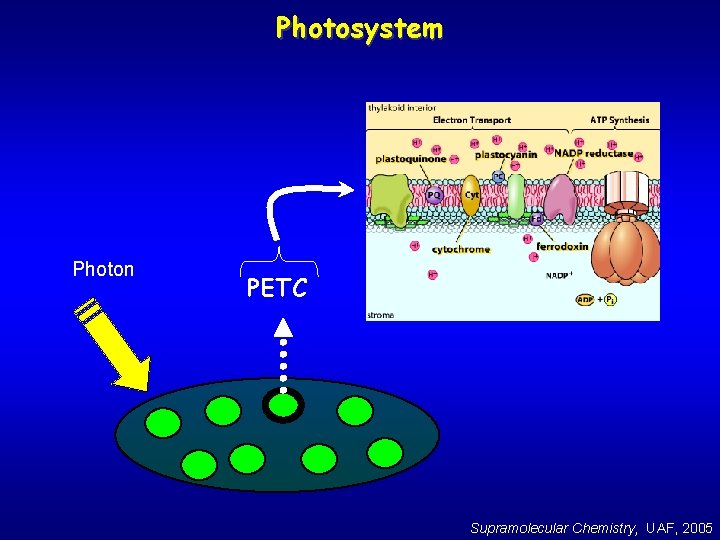
Photosystem From the Photosystem, e- are passed along an Electron Transport Chain. . The Photosynthetic Electron Transport Chain (PETC) Photon Reaction Center Light Harvesting Pigments Supramolecular Chemistry, UAF, 2005

Photosystem Photon PETC Supramolecular Chemistry, UAF, 2005

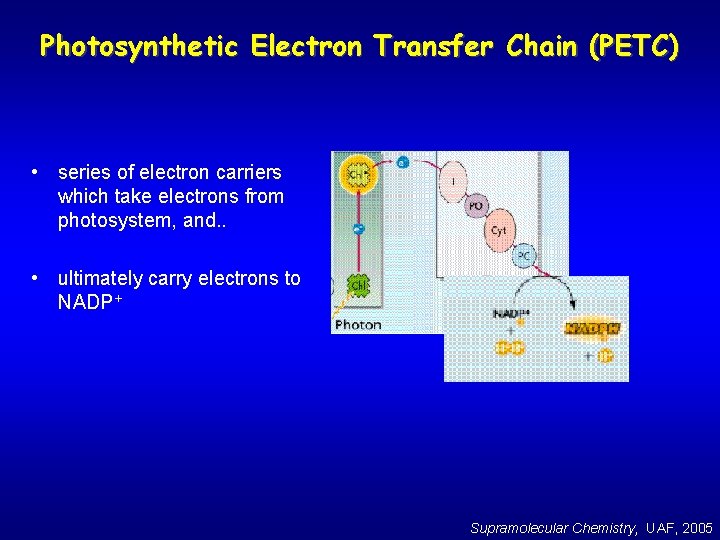
Photosynthetic Electron Transfer Chain (PETC) • series of electron carriers which take electrons from photosystem, and. . • ultimately carry electrons to NADP+ Supramolecular Chemistry, UAF, 2005

Photosystems • Experiments in the 1940’s suggested that light photons are absorbed at 2 different points along the same PETC. … • In fact, there are two Photosystems in operation Supramolecular Chemistry, UAF, 2005

This powerpoint was kindly donated to www. worldofteaching. com http: //www. worldofteaching. com is home to over a thousand powerpoints submitted by teachers. This is a completely free site and requires no registration. Please visit and I hope it will help in your teaching. Supramolecular Chemistry, UAF, 2005