Pharmacy 483 QI and DUE in Pharmacy Practice


















- Slides: 23

Pharmacy 483: QI and DUE in Pharmacy Practice Steve Riddle, BS Pharm, BCPS QI and Medication Utilization Lead HMC Pharmacy February 24, 2004

Acute Myocardial Infarction • HA, 52 yo male admitted via ER with severe, “crushing chest pain”, ST elevation with positive enzyme elevations. • What should be done for this patient?

Why do we need QI in pharmacy or in healthcare

How do we assess quality? • Quality Assurance (QA): quality assurance is any systematic process of checking to see whether a product or service is meeting specified requirements – Implies “maintenance of standard” • Quality Improvement (QI) – Focus is on improvement of product or service or process

Continuous Quality Improvement (CQI) “Doing things right first time" • Implies that there is only one way to do something and that good quality care is static and unchanging. • It is essential to strive for continuous quality improvement and not to assume that because things are "done right first time" they cannot be done better.

Three Categories of Quality Improvement • Eliminating quality problems – Remove unsafe on ineffective agents from formulary – Facilitating use of most appropriate agent – Reducing order-drug turnaround times (ie, automation) • Reducing costs while maintaining or improving quality – Optimize drug acquisition cost: contract negotiations, Group Purchasing Organizations (GPOs) – Therapeutic substitution initiatives (ex. , PPIs) – Generic utilization • Expanding customer expectations – Development of innovative products and services to attract customers (ie, CDTM, mail order)

QI Methodology Many QI theories or methods. Most share key steps…. • Identify What are you improving? • Analyze Understand the problem(s) • Develop Hypothesize solutions/changes • Test or Implement Put it into practice • Assess Outcomes What worked? • Sustain Hold the gains • Spread Broaden scope of gains

AMI Treatment: 3 QI Examples In Pharmacy. #1 Disease State Management #2 Pharmacologic Class Review #3 Drug Use Evaluation (DUE)

AMI Drug Treatment: Assessing Quality Indicators • What are goals? – Current Clinical Recommendations (AHA & NCEP Guidelines) – Benchmarking (CMS Data, UHC) • Review patient data for EBM drug indicators – Retrospective: Disch Dx (ICD-9 Codes), – Prospective (”Real Time”) • Identify areas for improvement – Where are major deficiencies?

Quality of Care for AMI: Disease State Management Focus on provision of key elements of care that optimize outcomes • Interventions (Arteriogram, PCTA, CABG) • Labs and Diagnostic Eval. (ECG, enzymes, Echo, EF) • Messages (Life Style Modification, Smoking Cessation, Medication Adherence) • Drug Therapy (Thrombolytics, Heparin, GP-2 B 3 A inhibitors, ASA, ACEIs, Beta-Blockers, Statins) • Timeliness of therapy (door-to-drug)

HMC Care Goals for AMI Measure Goal Sampling Plan AMI patient discharged on ASA AMI patient discharged on ACEI 100% Chart Review AMI patient discharged on Beta Blocker AMI patient discharged on Statin (if LDL > 130) 100% Chart Review Smokers with CV Condition will have documented cessation advice/counseling 75% Chart and CIS documentation review

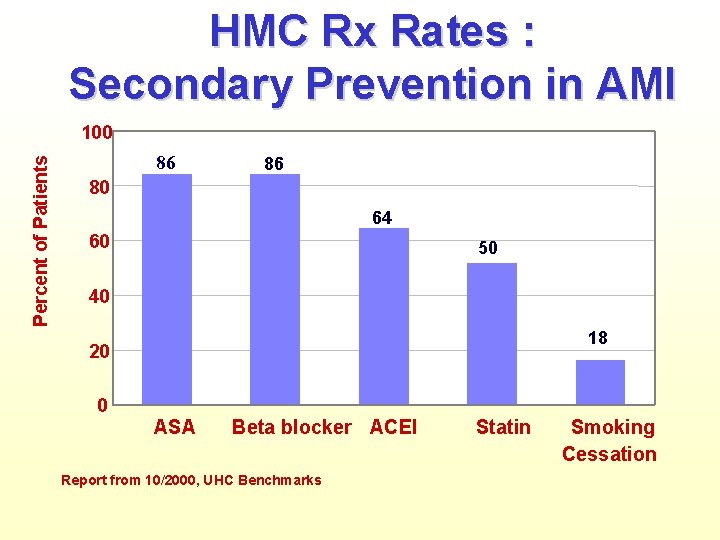
HMC Rx Rates : Secondary Prevention in AMI Percent of Patients 100 86 86 80 64 60 50 40 18 20 0 ASA Beta blocker ACEI Report from 10/2000, UHC Benchmarks Statin Smoking Cessation

AMI Treatment: Indicated Drugs Under Utilized? • • Problems Provider lack of awareness of benefits Inconsistencies in prescribing habits Lack of use of current prescribing aids Complex processes • • • Solutions education/awareness of providers Simplify processes order sets, clinical pathways Designate specific responsibilities Clinical Care Coordinator or pharmacist on clinical team Use data (ie, daily admit printouts)

Pharmacist Role • Collaborate in development of practice guidelines – – • Influence prescribing patterns – – • Committee involvement Standing order and clinical pathway development Daily rounding or clinic interactions Conduct educational programs for residents Provide feedback to prescribers around specific drugs “Counter-detailing” Perform direct patient care roles – – – Anticoagulation service Collaborative disease management protocols Patient education programs

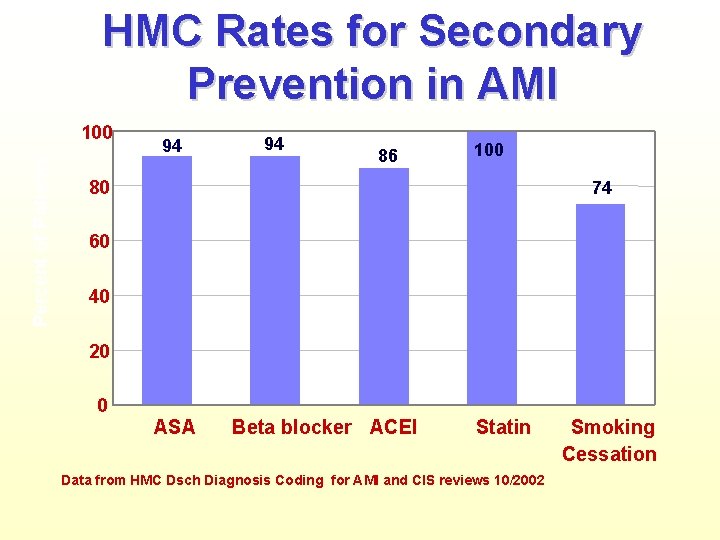
HMC Rates for Secondary Prevention in AMI Percent of Patients 100 94 94 86 100 80 74 60 40 20 0 ASA Beta blocker ACEI Statin Data from HMC Dsch Diagnosis Coding for AMI and CIS reviews 10/2002 Smoking Cessation

ACEI Class Review • Clinical Efficacy – – Numerous agents Varying degrees of literature support FDA approved indications Theoretical differences vs. hard outcomes vs. missing data – “Class Effect”? • Cost – Low-cost generics vs. brand – Pharmaceutical company detailing • Convenience – Once daily vs. BID dosing

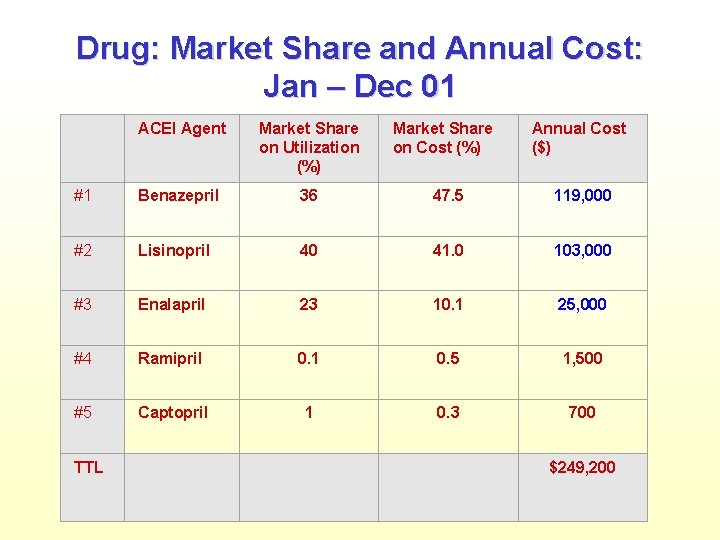
Drug: Market Share and Annual Cost: Jan – Dec 01 ACEI Agent Market Share on Utilization (%) #1 Benazepril 36 47. 5 119, 000 #2 Lisinopril 40 41. 0 103, 000 #3 Enalapril 23 10. 1 25, 000 #4 Ramipril 0. 1 0. 5 1, 500 #5 Captopril 1 0. 3 700 TTL Market Share on Cost (%) Annual Cost ($) $249, 200

Drug Use Evaluation (DUE) • Definition: Authorized, structured, ongoing review of practitioner prescribing, pharmacist dispensing and patient use of medications. • Purpose: To ensure drugs are used appropriately, safely, and effectively to – Improve patient care – Lower the overall cost of care – Foster more efficient use of health care resources • Process – Comprehensive review of medication use data – Identify patterns of prescribing

DUE Targets • Therapeutic appropriateness • Appropriate generic or FLA utilization • Inappropriate dose and/or duration • Over and underutilization • Compliance with polices/guidelines

DUE: Ramipril • Restrictions: – Limited Indications: HOPE Criteria – Cost: Trade name vs. generic alternatives • Appropriate Use – Chart reviews of users – Compare actual use to restriction criteria – Percent compliance rate • Assessment

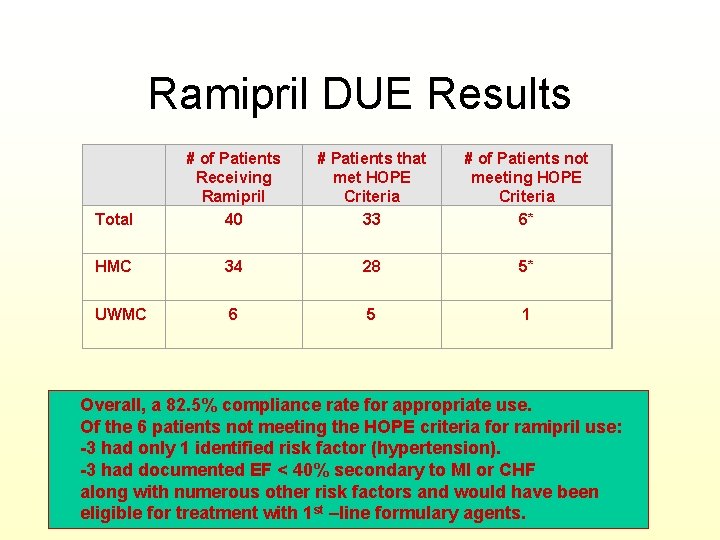
Ramipril DUE Results Total # of Patients Receiving Ramipril 40 # Patients that met HOPE Criteria 33 # of Patients not meeting HOPE Criteria 6* HMC 34 28 5* UWMC 6 5 1 Overall, a 82. 5% compliance rate for appropriate use. Of the 6 patients not meeting the HOPE criteria for ramipril use: -3 had only 1 identified risk factor (hypertension). -3 had documented EF < 40% secondary to MI or CHF along with numerous other risk factors and would have been eligible for treatment with 1 st –line formulary agents.


QUESTIONS?