Isentropic flow through Nozzles The main idea of










- Slides: 19

Isentropic flow through Nozzles

The main idea of jet engine is reverse reaction The reverse reaction is mainly depend on Newton laws which state that “any action have equal another reaction equal in Magnitude and opposite in direction “ The force acting on the body to move is equal The change in momentum The momentum = mass of the body * velocity To get a maximum force you should increase 1 - mass 2 - velocity

• Subsonic, incompressible • Subsonic, compressible • Supersonic


Stagnation Point Locations

Isentropic relation in tubular form

One-Dimensional Isentropic Flow Property Relations for Isentropic Flow of Ideal Gases

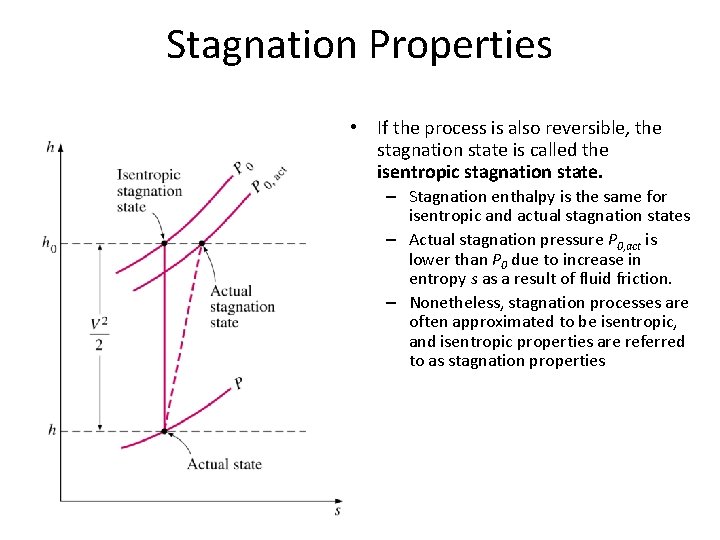
Stagnation Properties • If the process is also reversible, the stagnation state is called the isentropic stagnation state. – Stagnation enthalpy is the same for isentropic and actual stagnation states – Actual stagnation pressure P 0, act is lower than P 0 due to increase in entropy s as a result of fluid friction. – Nonetheless, stagnation processes are often approximated to be isentropic, and isentropic properties are referred to as stagnation properties

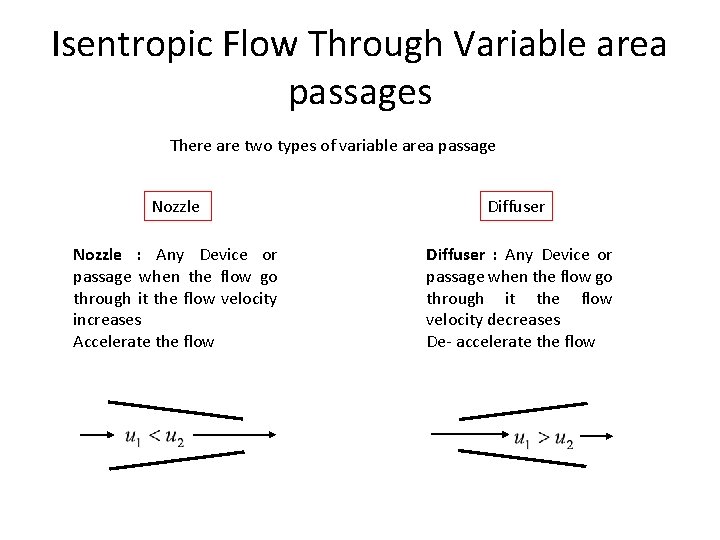
Isentropic Flow Through Variable area passages There are two types of variable area passage Nozzle Diffuser Nozzle : Any Device or passage when the flow go through it the flow velocity increases Accelerate the flow Diffuser : Any Device or passage when the flow go through it the flow velocity decreases De- accelerate the flow

Isentropic Flow Through Variable area passages Converging or converging-diverging nozzles are found in many engineering applications Steam and gas turbines, aircraft and spacecraft propulsion, industrial blast nozzles, torch nozzles • Here, we will study the effects of back pressure (pressure at discharge) on the exit velocity, mass flow rate, and pressure distribution along the nozzle

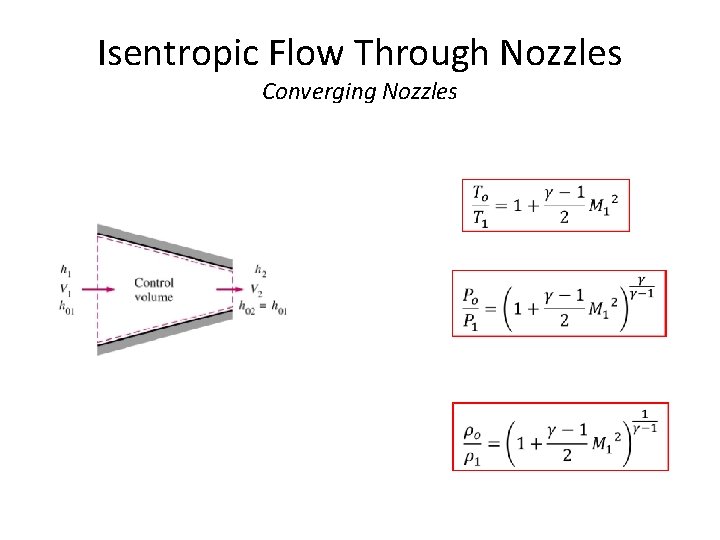
Isentropic Flow Through Nozzles Converging Nozzles • Under steady flow conditions, mass flow rate is constant

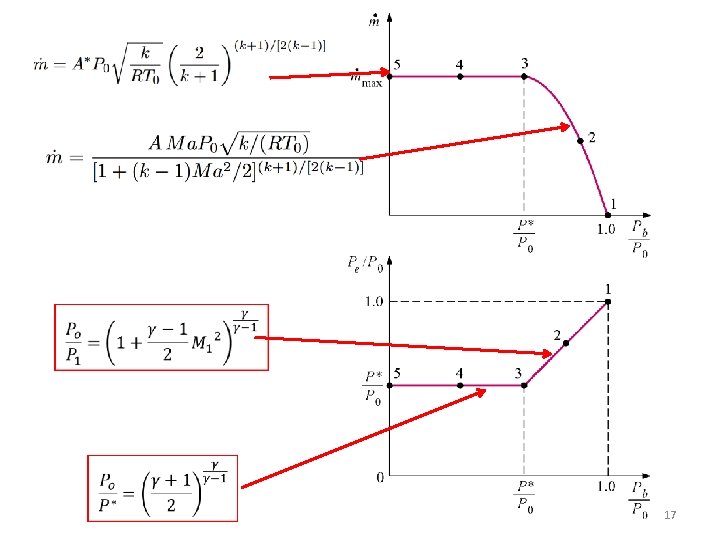
Substituting T and P from the expressions Mass flow rate is a function of stagnation properties, flow area, and Mach No

Isentropic Flow Through Nozzles Converging Nozzles • The maximum mass flow rate through a nozzle with a given throat area A* is fixed by the P 0 and T 0 and occurs at Ma = 1 • This principal is important for chemical processes, medical devices, flow meters, and anywhere the mass flux of a gas must be known and controlled.

Isentropic Flow Through Nozzles Converging Nozzles

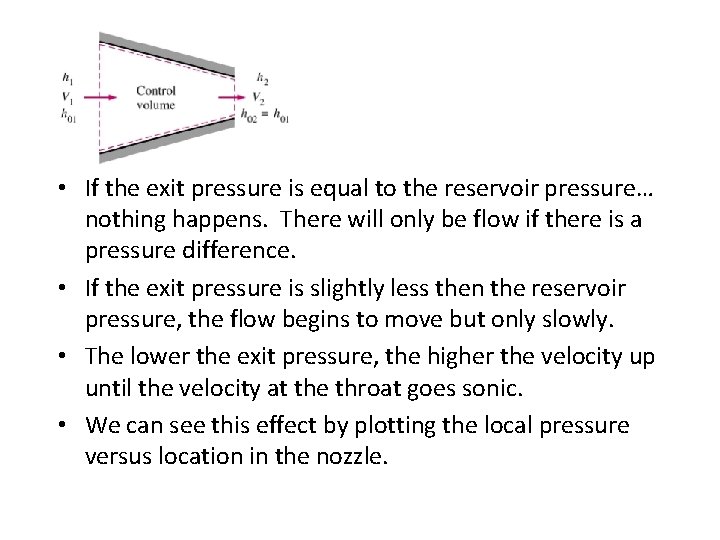
• If the exit pressure is equal to the reservoir pressure… nothing happens. There will only be flow if there is a pressure difference. • If the exit pressure is slightly less then the reservoir pressure, the flow begins to move but only slowly. • The lower the exit pressure, the higher the velocity up until the velocity at the throat goes sonic. • We can see this effect by plotting the local pressure versus location in the nozzle.

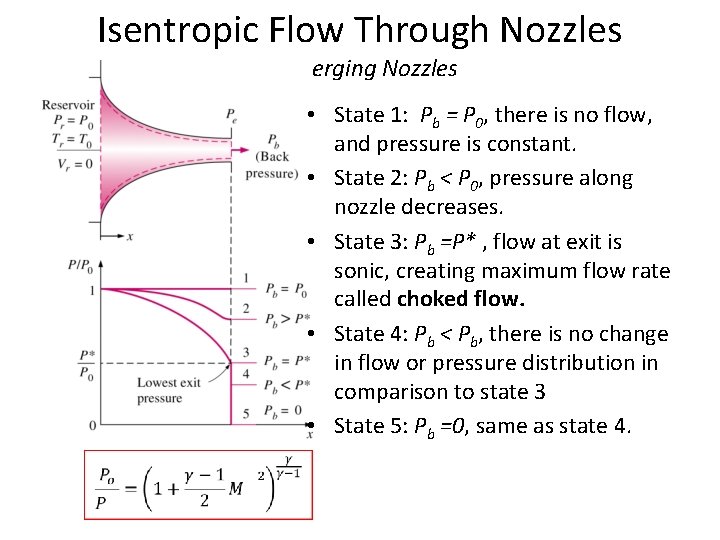
Isentropic Flow Through Nozzles Converging Nozzles • State 1: Pb = P 0, there is no flow, and pressure is constant. • State 2: Pb < P 0, pressure along nozzle decreases. • State 3: Pb =P* , flow at exit is sonic, creating maximum flow rate called choked flow. • State 4: Pb < Pb, there is no change in flow or pressure distribution in comparison to state 3 • State 5: Pb =0, same as state 4.

17

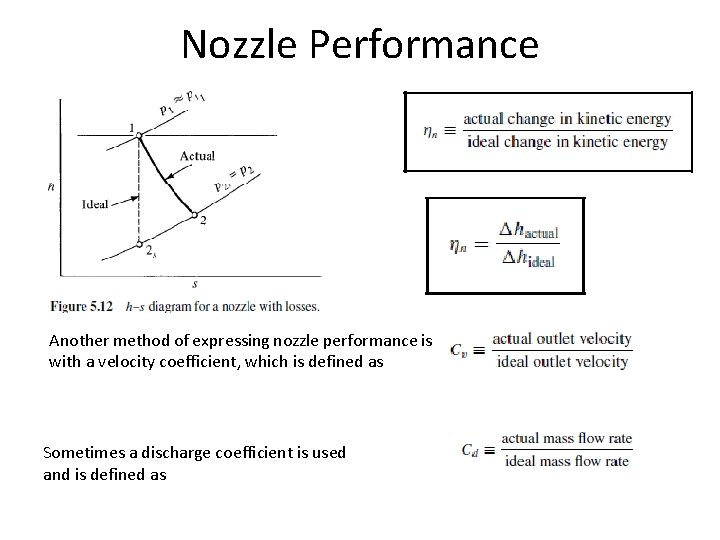
Nozzle Performance Another method of expressing nozzle performance is with a velocity coefficient, which is defined as Sometimes a discharge coefficient is used and is defined as

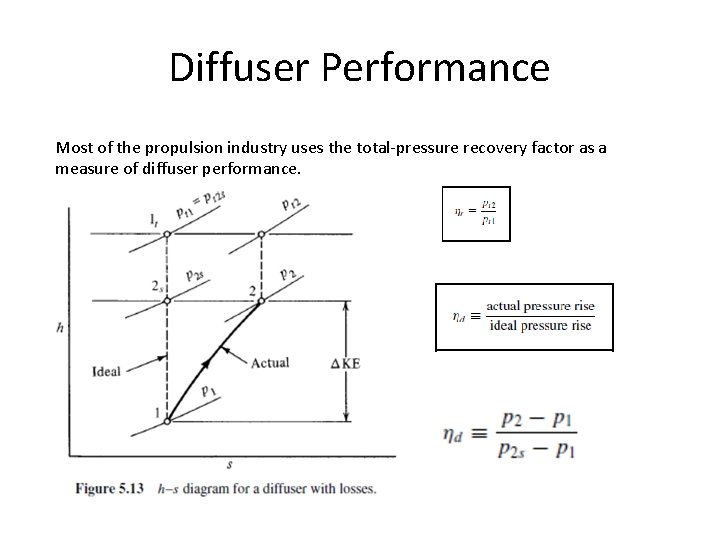
Diffuser Performance Most of the propulsion industry uses the total-pressure recovery factor as a measure of diffuser performance.