Introduction to Radiation Radiation Types Health Physics Society










- Slides: 12

Introduction to Radiation: Radiation Types ©Health Physics Society

Introduction to Radiation • Objectives • To provide useful information about • • radiation for interested individuals To introduce basic concepts of radiation and radioactivity Improve understanding of radiation – what it is and how it interacts

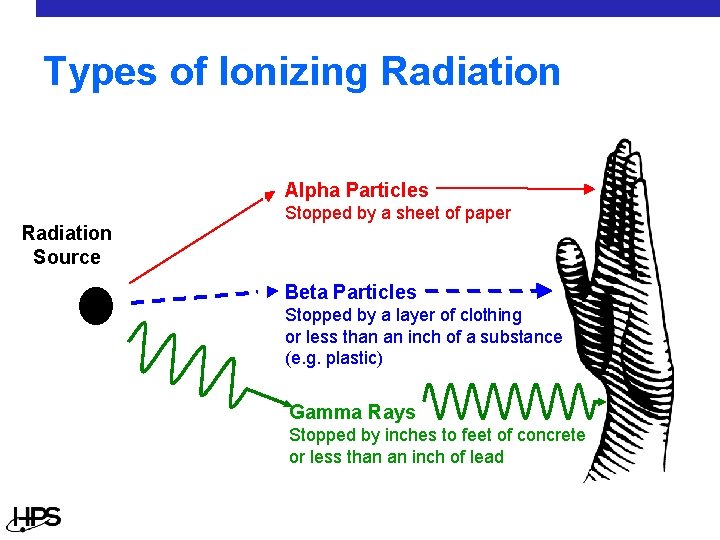
Types of Ionizing Radiation Alpha Particles Radiation Source Stopped by a sheet of paper Beta Particles Stopped by a layer of clothing or less than an inch of a substance (e. g. plastic) Gamma Rays Stopped by inches to feet of concrete or less than an inch of lead

Radiation Types - Alpha • An alpha particle consists of two protons and two neutrons • Very large on an atomic scale • Positively charged • Penetration in materials • Outside the body, an alpha emitter is • not a hazard unless it is on the skin Inside the body, an alpha emitter is a bigger hazard if it deposits energy in sensitive tissue

Radiation Types - Alpha • Common alpha-particle emitters • Radon-222 gas in the environment • Uranium-234 and -238) in the • environment Polonium-210 in tobacco • Common alpha-particle emitter uses • Smoke detectors • Cigarettes/cigars • Static eliminators

Radiation Types - Beta • A beta particle is a charged electron • Has the size and weight of an electron • Can be positively or negatively charged • Penetration in materials • At low energies, a beta particle is not very • penetrating – stopped by the outer layer of skin or a piece of paper At higher energies, a beta particle may penetrate to the live layer of skin and may need 0. 5” of plexiglass to be stopped

Radiation Types - Beta • Penetration in materials, continued • Inside the body, a beta particle is not as • hazardous as an alpha particle because it is not as big Because it is not as big, it travels farther, interacting with more tissue (but each small piece of tissue gets less energy deposited)

Radiation Types - Beta • Common beta-particle emitters • Tritium (hydrogen-3) in the environment • Carbon (14) in the environment • Phosphorus (32) used in research and medicine • Common beta-particle emitter uses • Carbon dating • Basic research • Cancer treatment

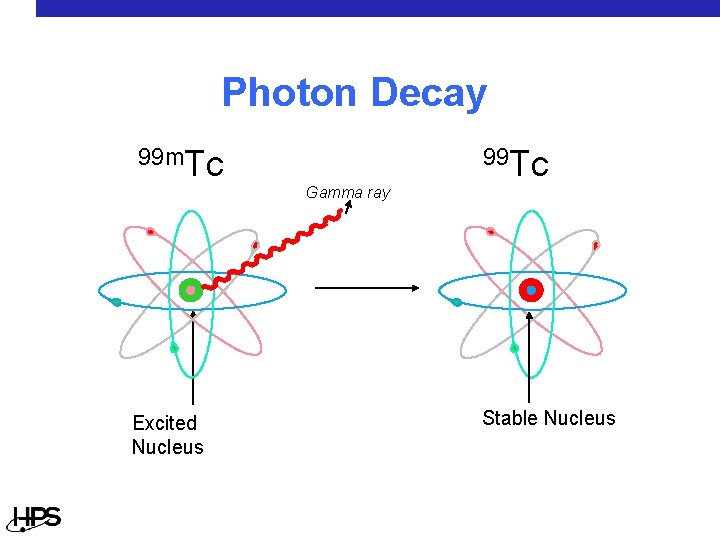
Radiation Types - Photon • A photon is an x or gamma ray • Has no weight • Has no charge • Penetration in materials • At low energies, a photon can be stopped • by a very thin (almost flexible) layer of lead or several centimeters of tissue At higher energies, inches of lead might be necessary to stop a photon and they can pass right through a human

Radiation Types - Photon • Common photon emitters • Cesium (137) • Technetium (99 m) used in medicine • Iodine (131) used in medicine • Common photon emitter uses • Determining the density of soil • Diagnosing disease • Cancer treatment

Photon Decay 99 Tc 99 m. Tc Gamma ray Excited Nucleus Stable Nucleus

Examples of Radioactive Materials Radionuclide Physical Half-Life Cesium-137 30 yrs Cobalt-60 5 yrs Plutonium-239 24, 000 yrs 600 Ci Nuclear Weapon Iridium-192 74 days 100 Ci Industrial Radiography Hydrogen-3 12 yrs 12 Ci Strontium-90 29 yrs 0. 1 Ci Iodine-131 8 days 0. 015 Ci Therapy Technetium-99 m 6 hrs 0. 025 Ci Imaging Americium-241 432 yrs Radon-222 4 days Activity 1. 5 x 106 Ci 15, 000 Ci 0. 000005 Ci 1 p. Ci/l Use Food Irradiator Cancer Therapy Exit Signs Eye Therapy Device Smoke Detectors Environmental