Elephant Toothpaste Aimee Gonzalez Materials Graduated cylinder or
















- Slides: 17

Elephant Toothpaste Aimee Gonzalez

Materials ❏ ❏ ❏ ❏ ❏ Graduated cylinder or 16 ounce soda bottle Hydrogen peroxide Dry yeast Potassium iodide Liquid dishwashing soap Food coloring Small cups Safety goggles Gloves

Safety Precautions ❏ This Experiment should be done outside. ❏ Wear proper protective equipment including gloves, lab coat, and safety glasses when preparing and performing this demonstration. Concentrated hydrogen peroxide and potassium iodide can cause skin and eye irritation. ❏ Disposal: Remaining solution can be flushed down drain with plenty of water.

Step 1 Pour hydrogen peroxide solution into the graduated cylinder.

Step 2 Squirt in a few drops of dishwashing soap

Step 3 Place 5 -10 drops of food coloring into the cylinder to make the foam colorful.

Step 4 Add potassium iodide solution into the graduated cylinder, watch to see what happens

Trial # 1 Trial # 2 Trial # 3 6% of hydrogen peroxide 12% of hydrogen peroxide 3% of hydrogen peroxide Dish Soap Dry Yeast Potassium iodide Dish Soap Food coloring Potassium iodide

Trial 1 ❏ Pour 100 ml of 6% of Hydrogen Peroxide into the graduated cylinder ❏ Add a squeeze of dishwashing soap ❏ Add 1 teaspoon of potassium iodide ❏ Wait to see what happens

Trial 2 ❏ Pour 150 ml of 12% of Hydrogen Peroxide into the graduated cylinder ❏ Squeeze a few drops of dishwashing soap ❏ Add a few drops of food dye ❏ Add 2 teaspoons of potassium iodide ❏ Wait to see what happens

Trail 3 ❏ Pour 200 ml of 3% Hydrogen Peroxide into the graduated cylinder ❏ Squeeze a small amount of dishwashing soap ❏ Add a few drops of food dye ❏ Add 3 teaspoons of potassium iodide ❏ One packet of dry yeast, mixed with hot water ❏ Wait to see what happens

What I learned ❏ Percentage of hydrogen peroxide ❏ Potassium iodide - powerful calyast

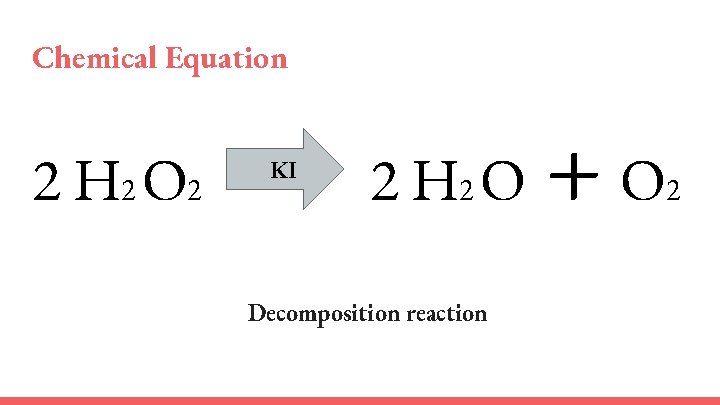
Chemical Equation 2 H 2 O 2 KI 2 H 2 O Decomposition reaction +O 2

Explanation ● The iodide ion (KI) is used as a catalyst to decompose H 2 O 2, liberating water, oxygen and heat. ● The yeast acted as a catalyst to remove the oxygen from the hydrogen peroxide. ● Exothermic Reaction – created both foam, and heat.

Real World ❏ Hydrogen peroxide is used to heal wounds

Stoichiometry

THANK YOU