Efficacy Paul Meyers MD ViceChairman Dept of Pediatrics






- Slides: 21

Efficacy Paul Meyers, MD Vice-Chairman, Dept of Pediatrics Memorial Sloan-Kettering Cancer Center 2023. 01 1

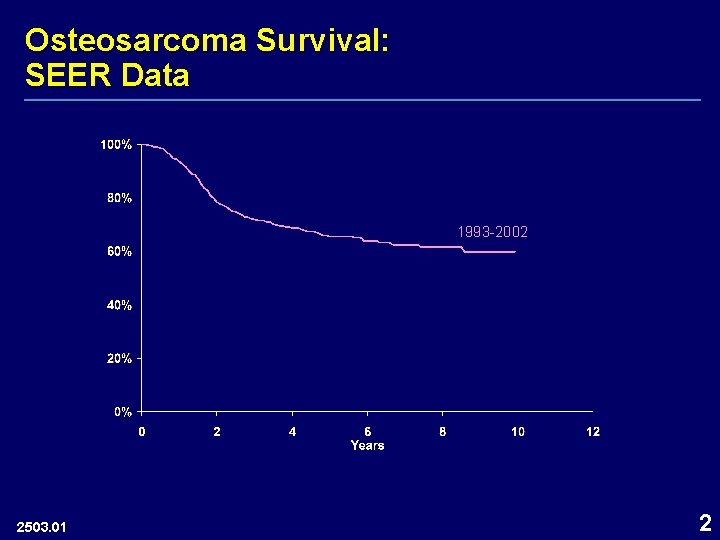
Osteosarcoma Survival: SEER Data 1993 -2002 2503. 01 2

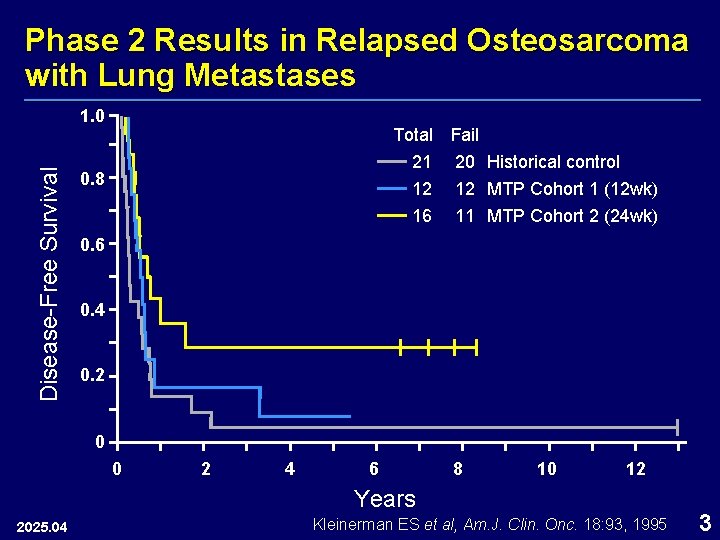
Phase 2 Results in Relapsed Osteosarcoma with Lung Metastases Disease-Free Survival 1. 0 Total Fail 21 20 Historical control 0. 8 12 16 12 MTP Cohort 1 (12 wk) 11 MTP Cohort 2 (24 wk) 0. 6 0. 4 0. 2 0 0 2 4 6 8 10 12 Years 2025. 04 Kleinerman ES et al, Am. J. Clin. Onc. 18: 93, 1995 3

Landmark Phase 3 Trial NCI-sponsored, Cooperative Group study – Designed and conducted independently of corporate sponsor Largest study completed in osteosarcoma – 178 sites – 1/3 of eligible incident cases in US Newly diagnosed (within 30 days; age <31) – 678 non-metastatic resectable disease – 115 with more advanced disease 2027. 01 4

Phase 3 Study Design INDUCTION A Cisplatin Doxorubicin HDMTX B Ifosfamide Doxorubicin HDMTX 2028. 03 D E F I N I T I V E S U R G E R Y MAINTENANCE Cisplatin, Doxorubicin, HDMTX A- Cisplatin, Doxorubicin, HDMTX, MTP A+ Cisplatin, Ifosfamide, Doxorubicin, HDMTX B- Cisplatin, Ifosfamide, Doxorubicin, HDMTX, MTP B+ 20 27 36 Weeks 5

Phase 3 Study Endpoints DFS – Putative surrogate for OS – Prospectively defined primary endpoint – “To determine whether MTP can improve DFS…” – “To compare the results of a prospective, randomized trial of two chemotherapeutic regimens…” (A vs. B) Survival – “To improve the survival of patients with osteogenic sarcoma” 2034. 02 6

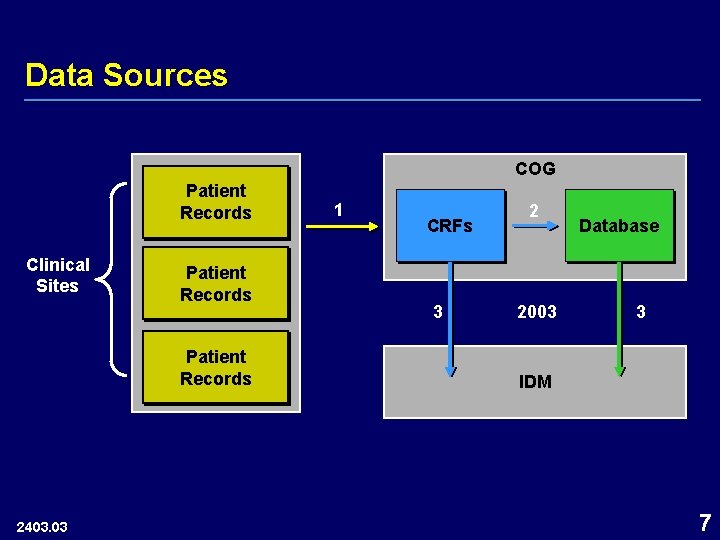
Data Sources COG Patient Records Clinical Sites Patient Records 2403. 03 1 CRFs 3 2 2003 Database 3 IDM 7

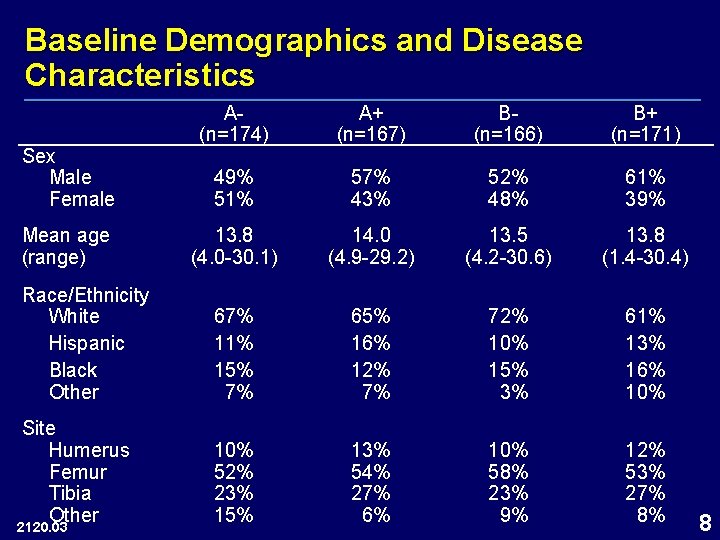
Baseline Demographics and Disease Characteristics A(n=174) A+ (n=167) B(n=166) B+ (n=171) 49% 51% 57% 43% 52% 48% 61% 39% 13. 8 (4. 0 -30. 1) 14. 0 (4. 9 -29. 2) 13. 5 (4. 2 -30. 6) 13. 8 (1. 4 -30. 4) Race/Ethnicity White Hispanic Black Other 67% 11% 15% 7% 65% 16% 12% 7% 72% 10% 15% 3% 61% 13% 16% 10% Site Humerus Femur Tibia Other 10% 52% 23% 15% 13% 54% 27% 6% 10% 58% 23% 9% 12% 53% 27% 8% Sex Male Female Mean age (range) 2120. 03 8

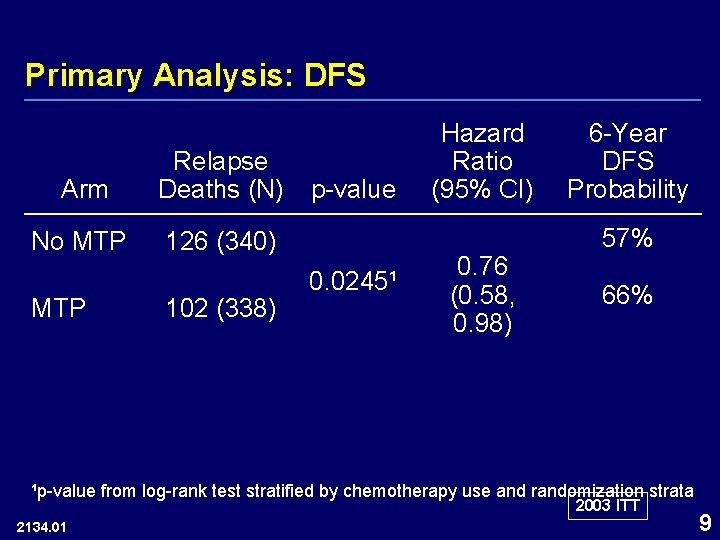
Primary Analysis: DFS Arm Relapse Deaths (N) No MTP 126 (340) MTP 102 (338) p-value 0. 0245¹ Hazard Ratio (95% CI) 0. 76 (0. 58, 0. 98) 6 -Year DFS Probability 57% 66% ¹p-value from log-rank test stratified by chemotherapy use and randomization strata 2003 ITT 2134. 01 9

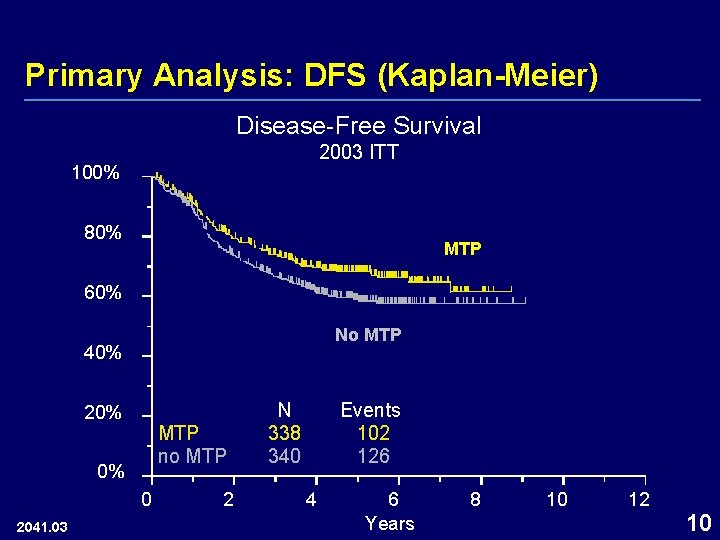
Primary Analysis: DFS (Kaplan-Meier) Disease-Free Survival 2003 ITT 100% 80% MTP 60% No MTP 40% 20% MTP no MTP 0% 0 2041. 03 2 N 338 340 Events 102 126 4 6 Years 8 10 12 10

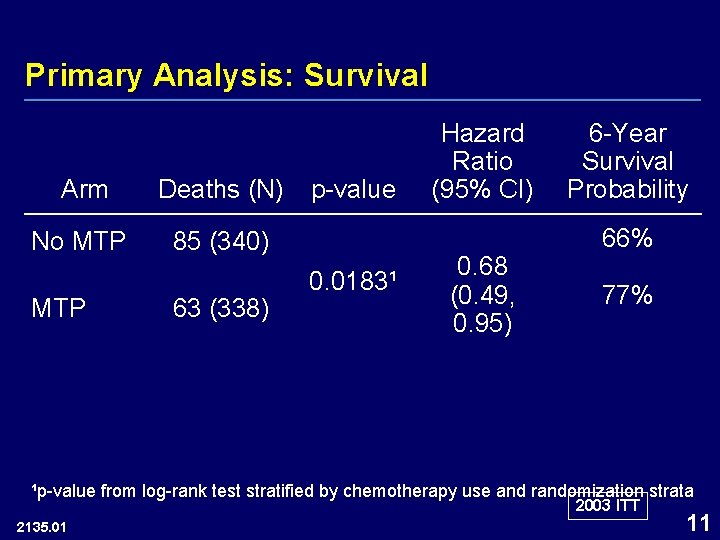
Primary Analysis: Survival Arm Deaths (N) No MTP 85 (340) MTP 63 (338) p-value 0. 0183¹ Hazard Ratio (95% CI) 0. 68 (0. 49, 0. 95) 6 -Year Survival Probability 66% 77% ¹p-value from log-rank test stratified by chemotherapy use and randomization strata 2003 ITT 2135. 01 11

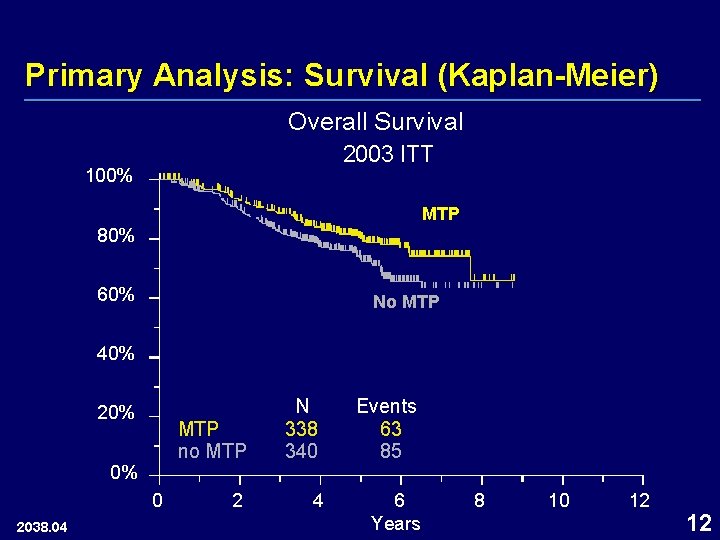
Primary Analysis: Survival (Kaplan-Meier) Overall Survival 2003 ITT 100% MTP 80% 60% No MTP 40% 20% 0% 0 2038. 04 MTP no MTP N 338 340 Events 63 85 2 4 6 Years 8 10 12 12

Data Sources COG Patient Records Clinical Sites Patient Records 2455. 01 1 CRF 2 2006 Database 3 IDM 13

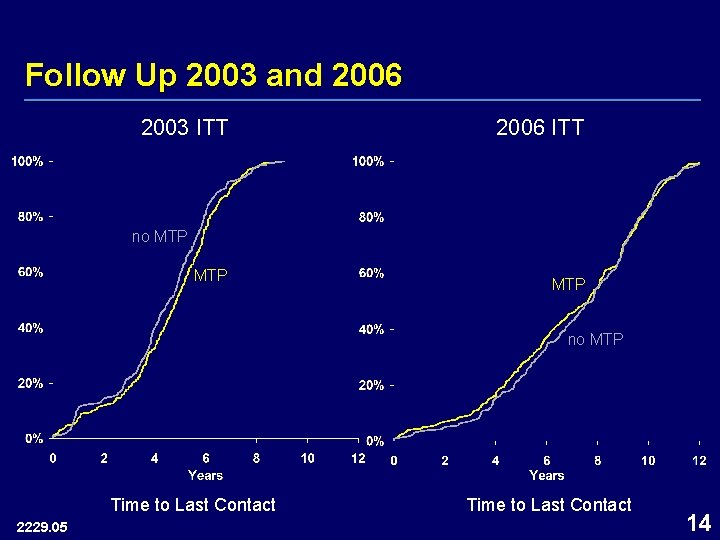
Follow Up 2003 and 2006 2003 ITT 2006 ITT no MTP MTP no MTP Time to Last Contact 2229. 05 Time to Last Contact 14

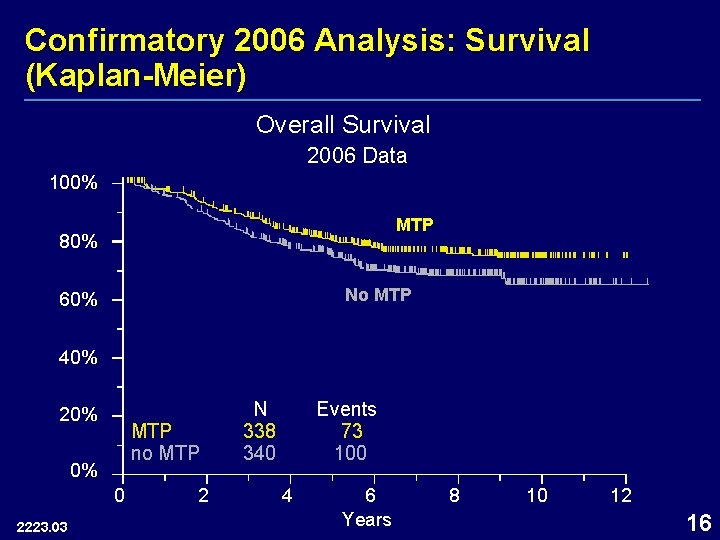
Confirmatory Analysis 2006: Survival Arm Deaths (N) No MTP 100 (340) MTP 73 (338) p-value 0. 0352¹ Hazard Ratio (95% CI) 0. 72 (0. 53, 0. 97) 6 -Year Survival Probability 70% 78% ¹p-value from log-rank test stratified by chemotherapy use and randomization strata 2006 ITT 2545. 01 15

Confirmatory 2006 Analysis: Survival (Kaplan-Meier) Overall Survival 2006 Data 100% MTP 80% No MTP 60% 40% 20% MTP no MTP 0% 0 2223. 03 2 N 338 340 Events 73 100 4 6 Years 8 10 12 16

Safety 2457. 01 17

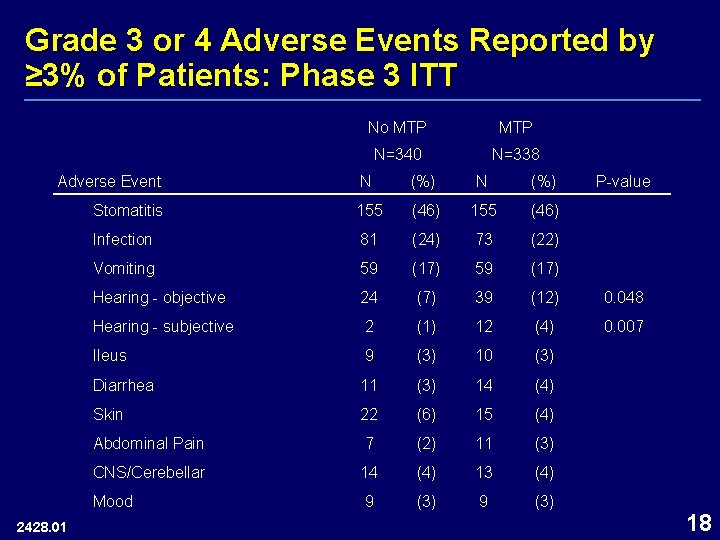
Grade 3 or 4 Adverse Events Reported by ≥ 3% of Patients: Phase 3 ITT Adverse Event 2428. 01 No MTP N=340 N=338 N (%) P-value Stomatitis 155 (46) Infection 81 (24) 73 (22) Vomiting 59 (17) Hearing - objective 24 (7) 39 (12) 0. 048 Hearing - subjective 2 (1) 12 (4) 0. 007 Ileus 9 (3) 10 (3) Diarrhea 11 (3) 14 (4) Skin 22 (6) 15 (4) Abdominal Pain 7 (2) 11 (3) CNS/Cerebellar 14 (4) 13 (4) Mood 9 (3) 18

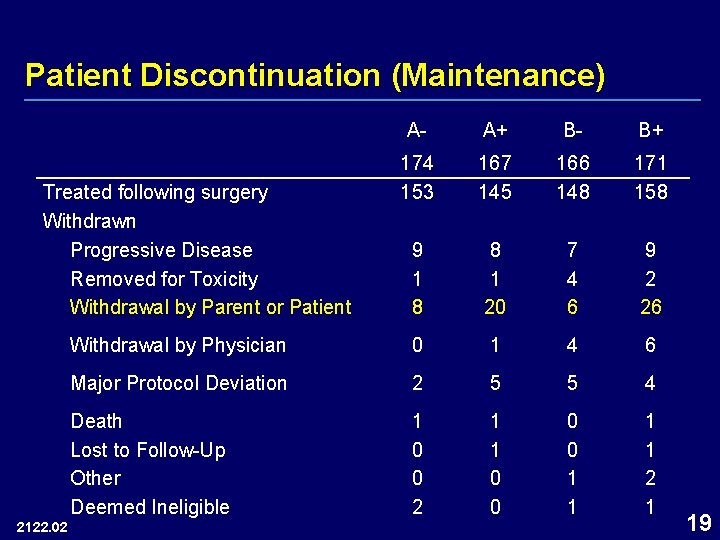
Patient Discontinuation (Maintenance) A- A+ B- B+ 174 153 167 145 166 148 171 158 9 1 8 8 1 20 7 4 6 9 2 26 Withdrawal by Physician 0 1 4 6 Major Protocol Deviation 2 5 5 4 Death Lost to Follow-Up Other Deemed Ineligible 1 0 0 2 1 1 0 0 1 1 2 1 Treated following surgery Withdrawn Progressive Disease Removed for Toxicity Withdrawal by Parent or Patient 2122. 02 19

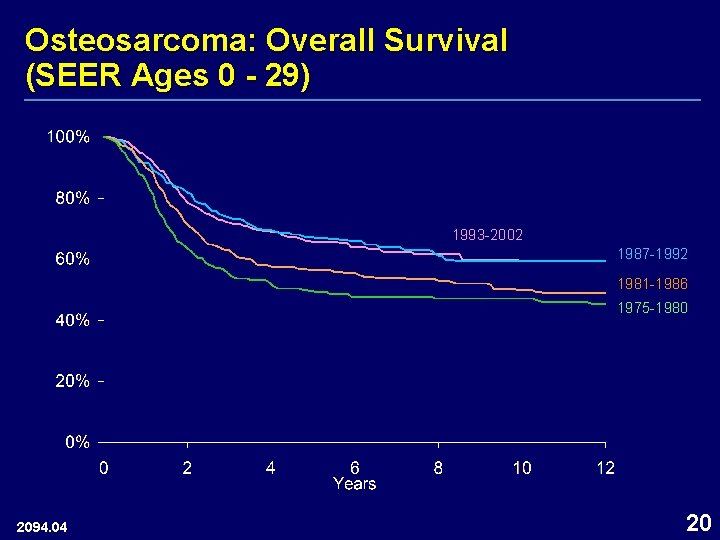
Osteosarcoma: Overall Survival (SEER Ages 0 - 29) 1993 -2002 1987 -1992 1981 -1986 1975 -1980 2094. 04 20

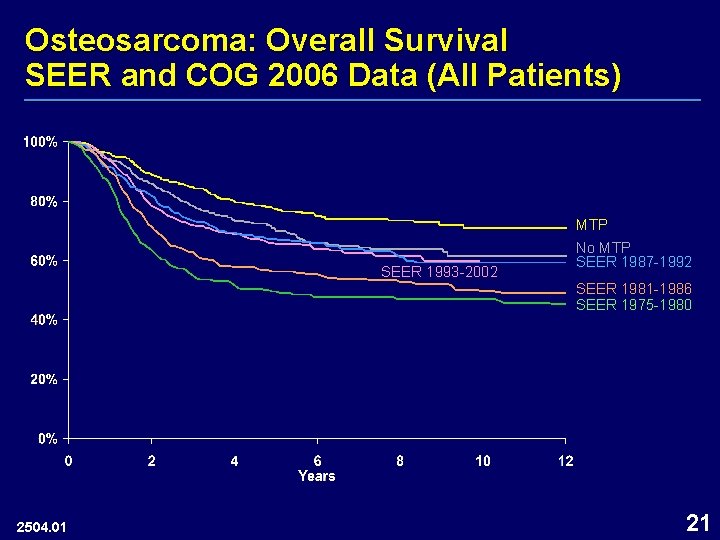
Osteosarcoma: Overall Survival SEER and COG 2006 Data (All Patients) MTP SEER 1993 -2002 No MTP SEER 1987 -1992 SEER 1981 -1986 SEER 1975 -1980 2504. 01 21