Diabetes macrovascular disease Prevalence of Diabetes Males ages

Diabetes & macrovascular disease
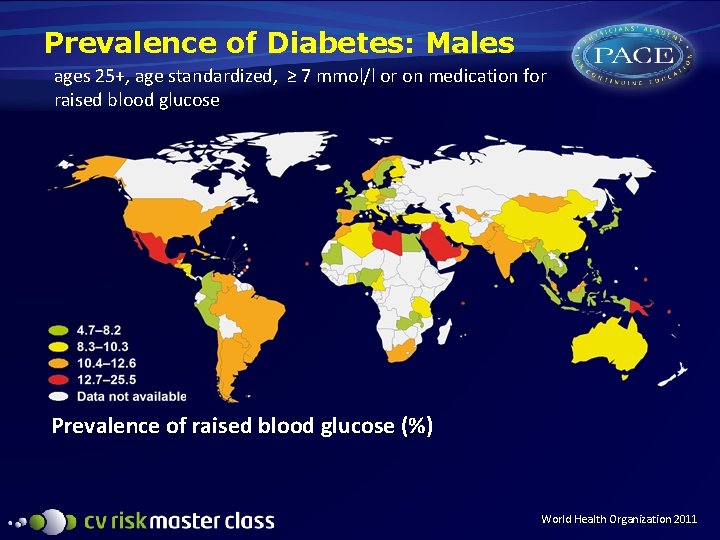
Prevalence of Diabetes: Males ages 25+, age standardized, ≥ 7 mmol/l or on medication for raised blood glucose Prevalence of raised blood glucose (%) World Health Organization 2011
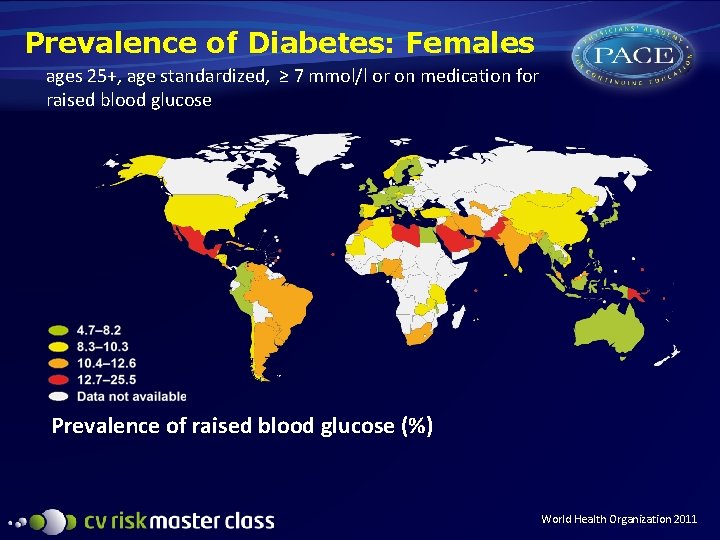
Prevalence of Diabetes: Females ages 25+, age standardized, ≥ 7 mmol/l or on medication for raised blood glucose Prevalence of raised blood glucose (%) World Health Organization 2011
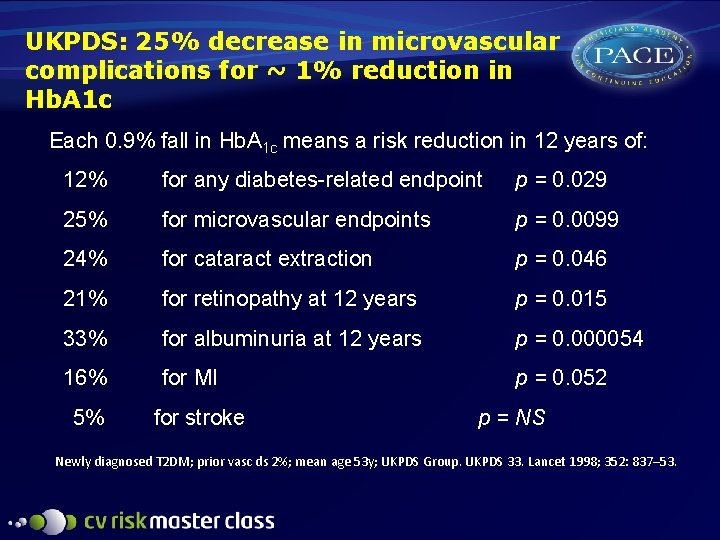
UKPDS: 25% decrease in microvascular complications for ~ 1% reduction in Hb. A 1 c Each 0. 9% fall in Hb. A 1 c means a risk reduction in 12 years of: 12% for any diabetes-related endpoint p = 0. 029 25% for microvascular endpoints p = 0. 0099 24% for cataract extraction p = 0. 046 21% for retinopathy at 12 years p = 0. 015 33% for albuminuria at 12 years p = 0. 000054 16% for MI p = 0. 052 5% for stroke p = NS Newly diagnosed T 2 DM; prior vasc ds 2%; mean age 53 y; UKPDS Group. UKPDS 33. Lancet 1998; 352: 837– 53.
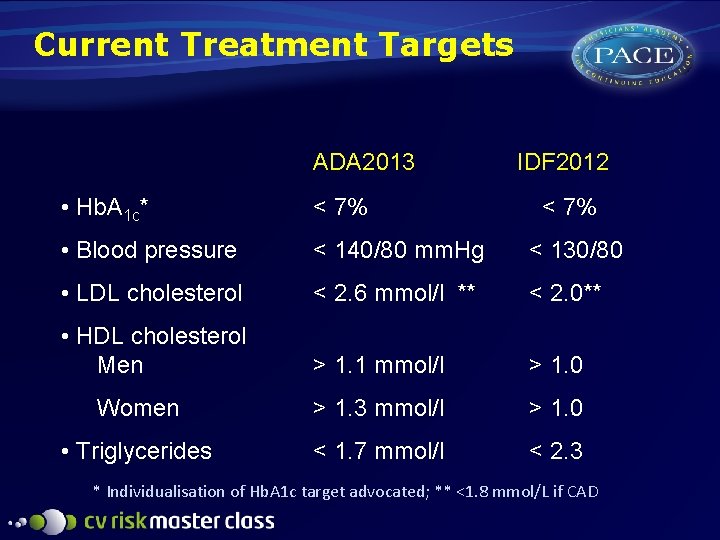
Current Treatment Targets ADA 2013 IDF 2012 • Hb. A 1 c* < 7% • Blood pressure < 140/80 mm. Hg < 130/80 • LDL cholesterol < 2. 6 mmol/l ** < 2. 0** • HDL cholesterol Men > 1. 1 mmol/l > 1. 0 Women > 1. 3 mmol/l > 1. 0 • Triglycerides < 1. 7 mmol/l < 2. 3 < 7% * Individualisation of Hb. A 1 c target advocated; ** <1. 8 mmol/L if CAD
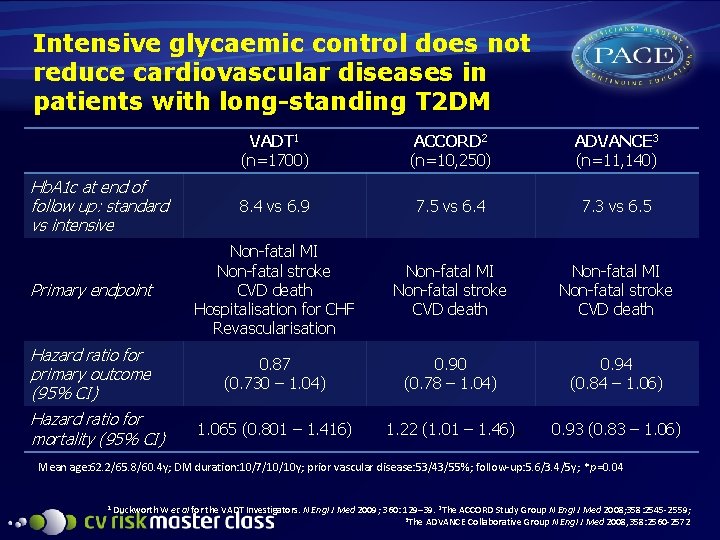
Intensive glycaemic control does not reduce cardiovascular diseases in patients with long-standing T 2 DM VADT 1 (n=1700) ACCORD 2 (n=10, 250) ADVANCE 3 (n=11, 140) 8. 4 vs 6. 9 7. 5 vs 6. 4 7. 3 vs 6. 5 Primary endpoint Non-fatal MI Non-fatal stroke CVD death Hospitalisation for CHF Revascularisation Non-fatal MI Non-fatal stroke CVD death Hazard ratio for primary outcome (95% CI) 0. 87 (0. 730 – 1. 04) 0. 90 (0. 78 – 1. 04) 0. 94 (0. 84 – 1. 06) 1. 065 (0. 801 – 1. 416) 1. 22 (1. 01 – 1. 46) Hb. A 1 c at end of follow up: standard vs intensive Hazard ratio for mortality (95% CI) * 0. 93 (0. 83 – 1. 06) Mean age: 62. 2/65. 8/60. 4 y; DM duration: 10/7/10/10 y; prior vascular disease: 53/43/55%; follow-up: 5. 6/3. 4/5 y; *p=0. 04 1 Duckworth W et al for the VADT Investigators. N Engl J Med 2009; 360: 129– 39. 2 The ACCORD Study Group N Engl J Med 2008; 358: 2545 -2559; 3 The ADVANCE Collaborative Group N Engl J Med 2008, 358: 2560 -2572
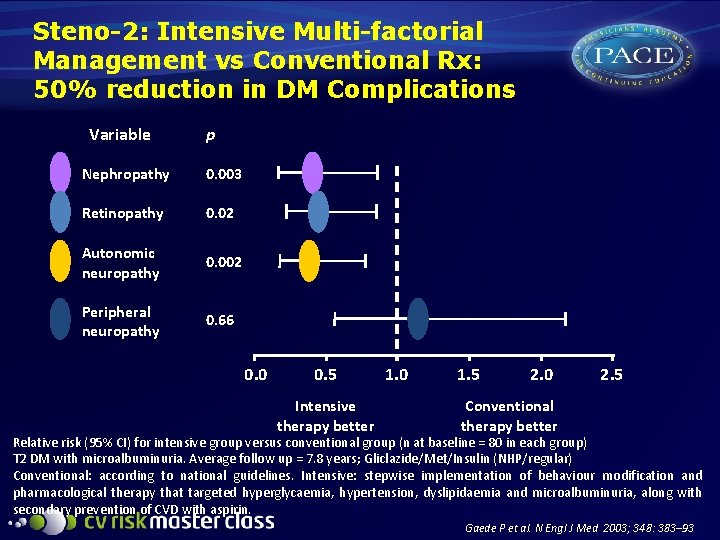
Steno-2: Intensive Multi-factorial Management vs Conventional Rx: 50% reduction in DM Complications Variable p Nephropathy 0. 003 Retinopathy 0. 02 Autonomic neuropathy 0. 002 Peripheral neuropathy 0. 66 0. 0 0. 5 Intensive therapy better 1. 0 1. 5 2. 0 2. 5 Conventional therapy better Relative risk (95% CI) for intensive group versus conventional group (n at baseline = 80 in each group) T 2 DM with microalbuminuria. Average follow up = 7. 8 years; Gliclazide/Met/Insulin (NHP/regular) Conventional: according to national guidelines. Intensive: stepwise implementation of behaviour modification and pharmacological therapy that targeted hyperglycaemia, hypertension, dyslipidaemia and microalbuminuria, along with secondary prevention of CVD with aspirin. Gaede P et al. N Engl J Med 2003; 348: 383– 93 .
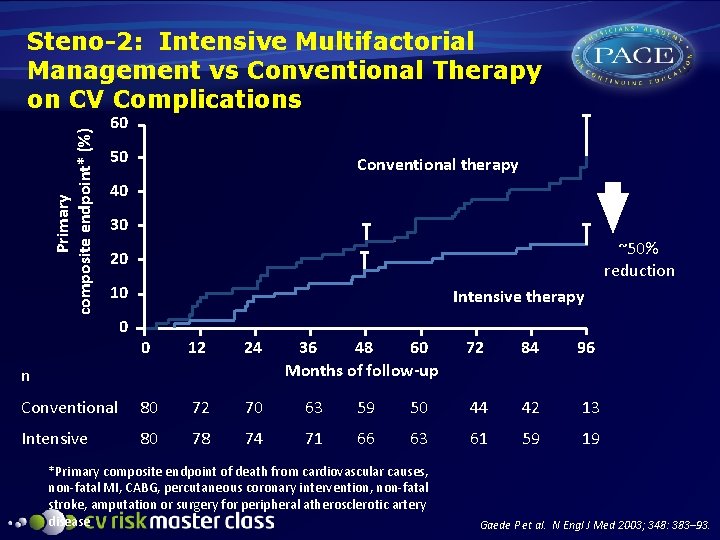
Primary composite endpoint* (%) Steno-2: Intensive Multifactorial Management vs Conventional Therapy on CV Complications 60 50 Conventional therapy 40 30 ~50% reduction 20 10 0 Intensive therapy 0 12 24 Conventional 80 72 70 63 59 Intensive 80 78 74 71 66 n 36 48 60 Months of follow-up 72 84 96 50 44 42 13 63 61 59 19 *Primary composite endpoint of death from cardiovascular causes, non-fatal MI, CABG, percutaneous coronary intervention, non-fatal stroke, amputation or surgery for peripheral atherosclerotic artery disease Gaede P et al. N Engl J Med 2003; 348: 383– 93.

Dyslipidaemia in Type 2 Diabetes Lipid Abnormalities Triglyceride Pathogenetic Mechanisms lipoprotein lipase activity HDL cholesterol lipoprotein lipase; CETP; Small dense LDL triglyceride; CETP; hepatic lipase activity LDL cholesterol LDL glycation Insulin resistance ( FFA + hepatic overproduction) hepatic lipase activity CETP, cholesterol ester transfer protein activity FFA, free fatty acids Lam KS, Diabetes in the New Millenium, 1999; p 327 -35
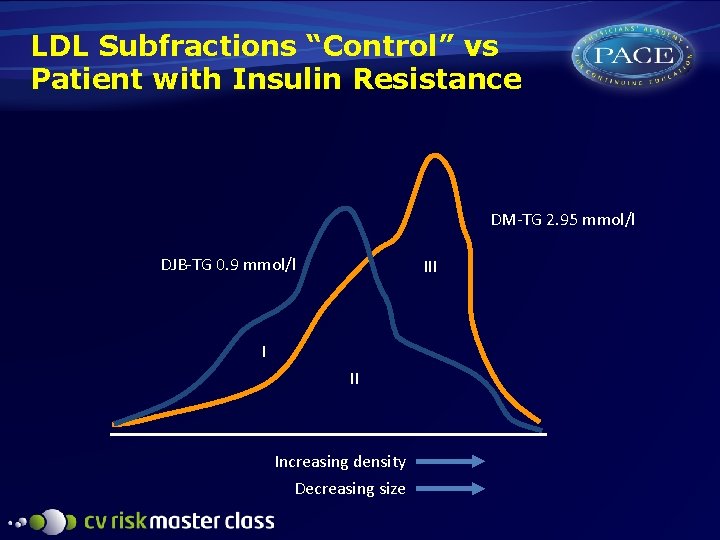
LDL Subfractions “Control” vs Patient with Insulin Resistance DM-TG 2. 95 mmol/l DJB-TG 0. 9 mmol/l III Increasing density Decreasing size

The Absolute Concentration of LDL-C Can be Misleading in Subjects with Small, Dense LDL? Large, buoyant particles Apo B Small, dense particles More apo B At the same LDL-C level, the number of LDL particles is increased, if small and dense Each LDL particle contains one molecule of apo B Apo B concentration increases in direct relation to number of LDL particles Sniderman AD et al Ann Intern Med 2001
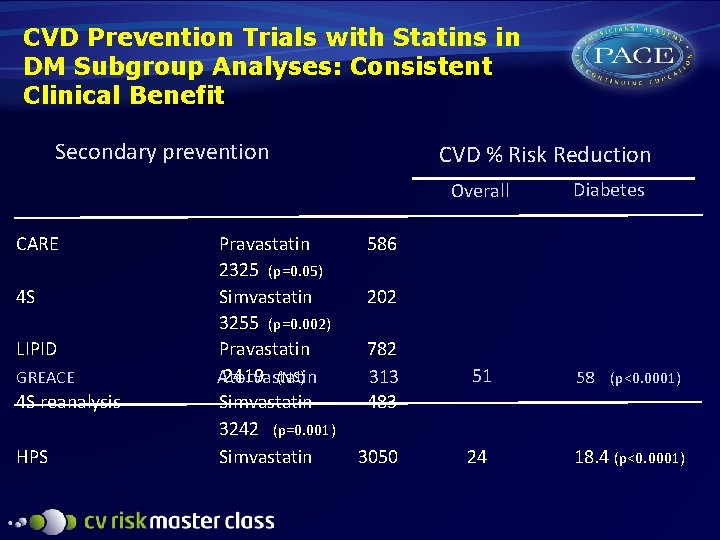
CVD Prevention Trials with Statins in DM Subgroup Analyses: Consistent Clinical Benefit Secondary prevention CVD % Risk Reduction Overall CARE 4 S LIPID GREACE 4 S reanalysis HPS Pravastatin 2325 (p=0. 05) Simvastatin 3255 (p=0. 002) Pravastatin 2419 (NS) Atorvastatin Simvastatin 3242 (p=0. 001) Simvastatin Diabetes 586 202 782 313 483 51 58 (p<0. 0001) 3050 24 18. 4 (p<0. 0001)
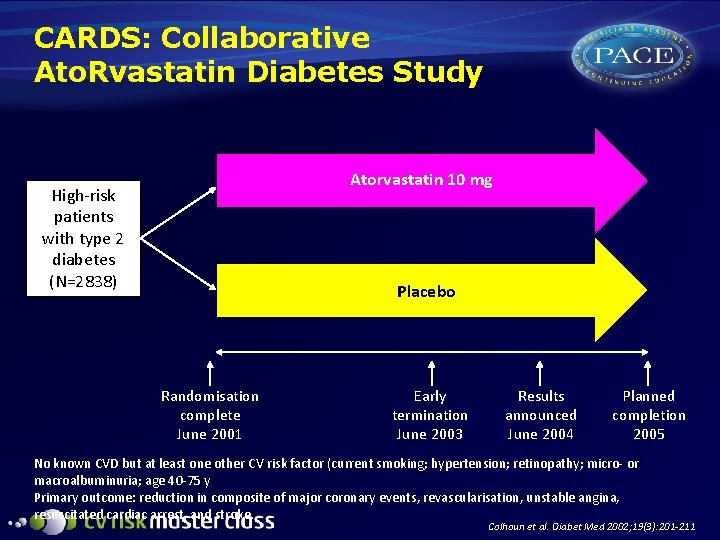
CARDS: Collaborative Ato. Rvastatin Diabetes Study Atorvastatin 10 mg High-risk patients with type 2 diabetes (N=2838) Placebo Randomisation complete June 2001 Early termination June 2003 Results announced June 2004 Planned completion 2005 No known CVD but at least one other CV risk factor (current smoking; hypertension; retinopathy; micro- or macroalbuminuria; age 40 -75 y Primary outcome: reduction in composite of major coronary events, revascularisation, unstable angina, resuscitated cardiac arrest, and stroke. Colhoun et al. Diabet Med 2002; 19(3): 201 -211
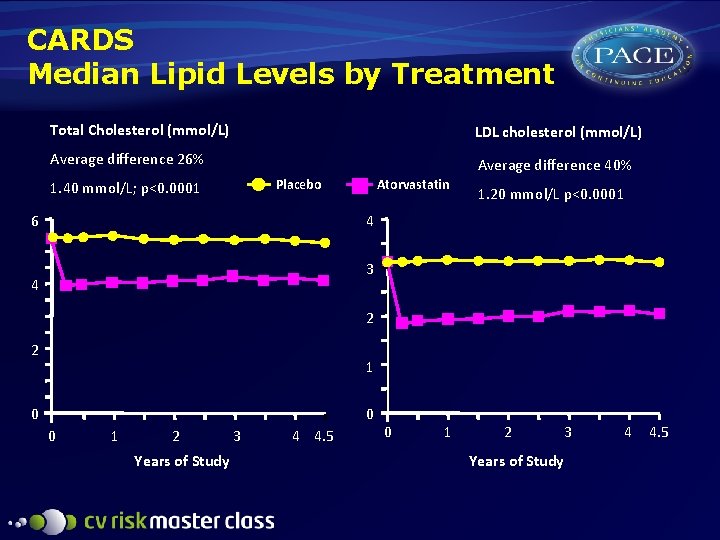
CARDS Median Lipid Levels by Treatment Total Cholesterol (mmol/L) LDL cholesterol (mmol/L) Average difference 26% Average difference 40% Placebo 1. 40 mmol/L; p<0. 0001 6 Atorvastatin 1. 20 mmol/L p<0. 0001 4 3 4 2 2 1 0 0 0 1 2 Years of Study 3 4 4. 5
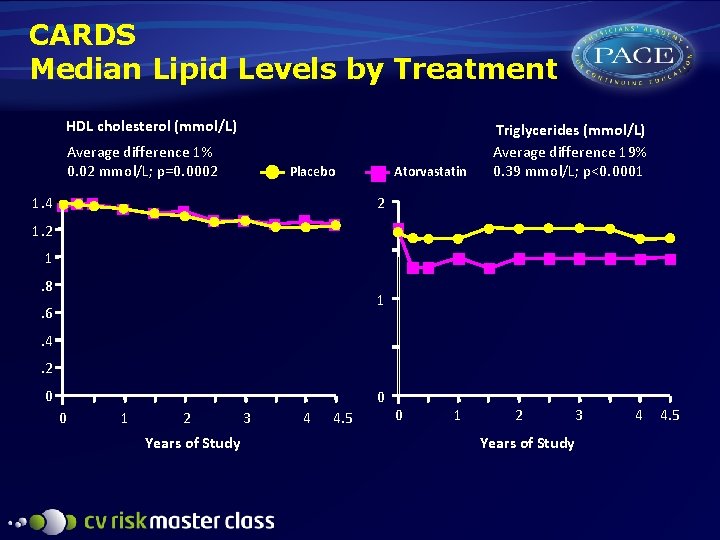
CARDS Median Lipid Levels by Treatment HDL cholesterol (mmol/L) Average difference 1% 0. 02 mmol/L; p=0. 0002 Placebo 1. 4 Atorvastatin Triglycerides (mmol/L) Average difference 19% 0. 39 mmol/L; p<0. 0001 2 1. 8 1 . 6. 4. 2 0 0 0 1 2 Years of Study 3 4 4. 5
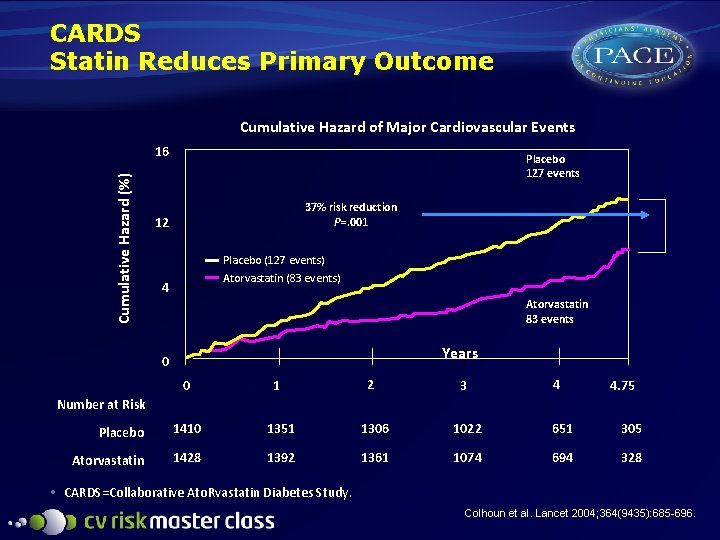
CARDS Statin Reduces Primary Outcome Cumulative Hazard of Major Cardiovascular Events Cumulative Hazard (%) 16 Placebo 127 events 37% risk reduction P=. 001 12 Placebo (127 events) Atorvastatin (83 events) 4 Atorvastatin 83 events Years 0 0 1 2 3 4 Placebo 1410 1351 1306 1022 651 305 Atorvastatin 1428 1392 1361 1074 694 328 4. 75 Number at Risk • CARDS=Collaborative Ato. Rvastatin Diabetes Study. Colhoun et al. Lancet 2004; 364(9435): 685 -696.
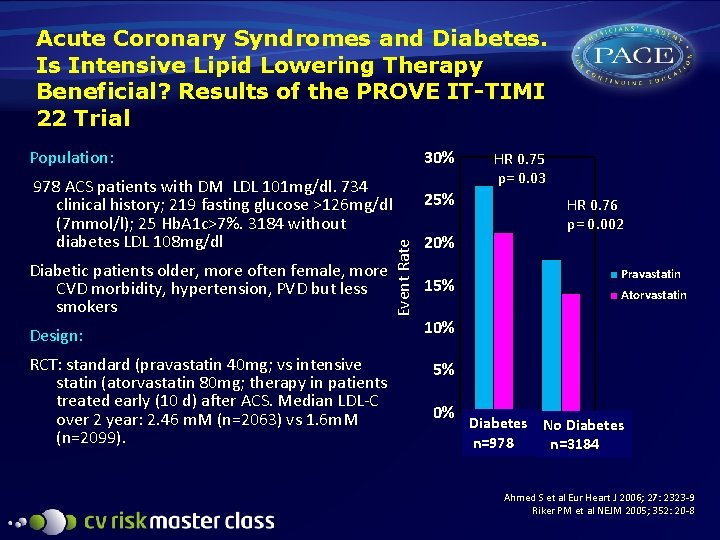
Acute Coronary Syndromes and Diabetes. Is Intensive Lipid Lowering Therapy Beneficial? Results of the PROVE IT-TIMI 22 Trial 30% Population: Diabetic patients older, more often female, more CVD morbidity, hypertension, PVD but less smokers Design: RCT: standard (pravastatin 40 mg; vs intensive statin (atorvastatin 80 mg; therapy in patients treated early (10 d) after ACS. Median LDL-C over 2 year: 2. 46 m. M (n=2063) vs 1. 6 m. M (n=2099). E v e n t R at e 978 ACS patients with DM LDL 101 mg/dl. 734 clinical history; 219 fasting glucose >126 mg/dl (7 mmol/l); 25 Hb. A 1 c>7%. 3184 without diabetes LDL 108 mg/dl 25% 20% 15% HR 0. 75 p= 0. 03 HR 0. 76 p= 0. 002 Pravastatin Atorvastatin 10% 5% 0% Diabetes. Non No Diabetes n=978 n=3184 Diabetes Ahmed S et al Eur Heart J 2006; 27: 2323 -9 Riker PM et al NEJM 2005; 352: 20 -8
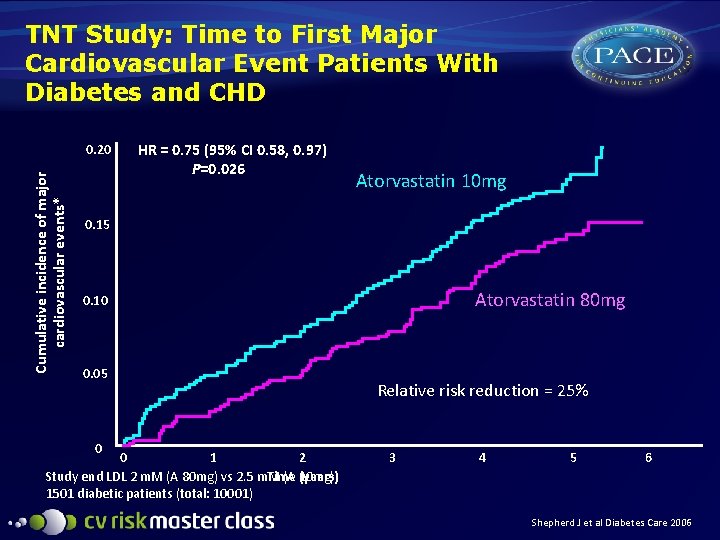
TNT Study: Time to First Major Cardiovascular Event Patients With Diabetes and CHD Cumulative incidence of major cardiovascular events* 0. 20 HR = 0. 75 (95% CI 0. 58, 0. 97) P=0. 026 Atorvastatin 10 mg 0. 15 Atorvastatin 80 mg 0. 10 0. 05 0 0 1 2 Study end LDL 2 m. M (A 80 mg) vs 2. 5 m. M (A 10 mg) Time (years) 1501 diabetic patients (total: 10001) Relative risk reduction = 25% 3 4 5 6 Shepherd J et al Diabetes Care 2006
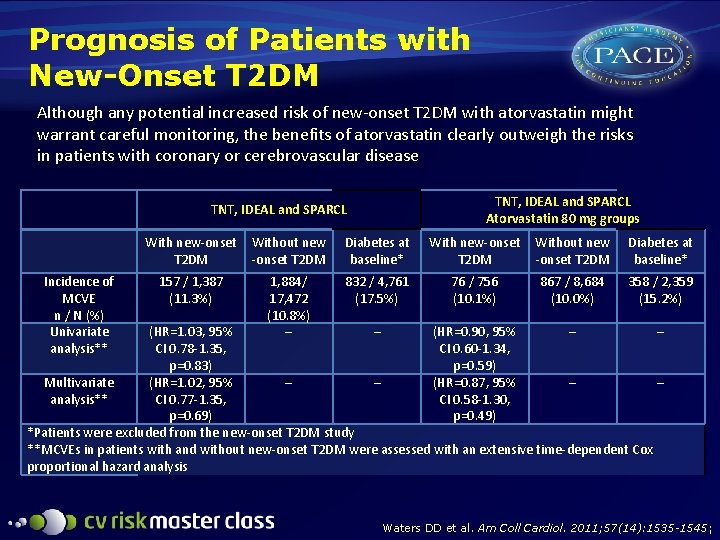
Prognosis of Patients with New-Onset T 2 DM Although any potential increased risk of new-onset T 2 DM with atorvastatin might warrant careful monitoring, the benefits of atorvastatin clearly outweigh the risks in patients with coronary or cerebrovascular disease TNT, IDEAL and SPARCL Atorvastatin 80 mg groups TNT, IDEAL and SPARCL Incidence of MCVE n / N (%) Univariate analysis** With new-onset T 2 DM Without new -onset T 2 DM Diabetes at baseline* 157 / 1, 387 (11. 3%) 1, 884/ 17, 472 (10. 8%) – 832 / 4, 761 (17. 5%) 76 / 756 (10. 1%) 867 / 8, 684 (10. 0%) 358 / 2, 359 (15. 2%) (HR=1. 03, 95% – (HR=0. 90, 95% – – CI 0. 78 -1. 35, CI 0. 60 -1. 34, p=0. 83) p=0. 59) Multivariate (HR=1. 02, 95% – – (HR=0. 87, 95% – – analysis** CI 0. 77 -1. 35, CI 0. 58 -1. 30, p=0. 69) p=0. 49) *Patients were excluded from the new-onset T 2 DM study **MCVEs in patients with and without new-onset T 2 DM were assessed with an extensive time-dependent Cox proportional hazard analysis Waters DD et al. Am Coll Cardiol. 2011; 57(14): 1535 -1545;

Is intensive LDL-cholesterol lowering beneficial and safe? PROVE IT-TIMI 22 substudy: No safety issues with 4 m LDL-C <40 mg-<80 mg/dl (n=1428); <40 -60 mg/dl significantly less major CV events than ≥ 80 -100 mg/dl (n=233) SPARCL: Atorvastatin 80 mg vs placebo 1 -6 m after stroke or TIA- HR 0. 8 for major CV event in 5 y (p=0. 002); HR 0. 78 (0. 66 -0. 94) for ischaemic stroke but HR 1. 66 (1. 082. 55; 55/2365 vs 33/2366) for haemorrhagic stroke; LDL-C 1. 89 vs 3. 3 m. M No ↑ in haemorrhagic stroke in PROVE IT-TIMI and TNT (2099 and 4995 on A 80 mg ; median 2 y and 4. 9 y; mainly no previous stroke) SPARCL: ↑ ALT 3 x UNR (2. 2% vs 0. 5%; p<0. 001) Meta-analysis of 13 statin trials: ↑ incident DM (OR 1. 09; 95% CI 1. 01 - 1. 17) Cheung BM & Lam KS. Lancet 2010; 376: 1622 -4 SPARCL Investigators. NEJM 2006; 355: 549 -59; Wiviott SD et al. JACC 2006; 46: 1411 -6; Sattaar N et al. Lancet. 2010: 735 -42
- Slides: 20