Determination of the hydrofluoric acid concentration in the






![The determination of [H 2 Si. F 6] or [HF] in the same bath The determination of [H 2 Si. F 6] or [HF] in the same bath](https://slidetodoc.com/presentation_image/7eb4a4b44ca3b0a49e6fa1ff774aca94/image-8.jpg)
![[H 2 Si. F 6] is determinate by the volume difference DVm. L = [H 2 Si. F 6] is determinate by the volume difference DVm. L =](https://slidetodoc.com/presentation_image/7eb4a4b44ca3b0a49e6fa1ff774aca94/image-10.jpg)
![[F-I] = [F -] + 6 [Si. F 62 -] Potentiometric Acido/Basic VEP 3 [F-I] = [F -] + 6 [Si. F 62 -] Potentiometric Acido/Basic VEP 3](https://slidetodoc.com/presentation_image/7eb4a4b44ca3b0a49e6fa1ff774aca94/image-11.jpg)



- Slides: 14

Determination of the hydrofluoric acid concentration in the solutions used for the chemical polishing of crystal. E. Brient Cristallerie de Baccarat, 20 rue des Cristalleries - 54120 Baccarat Y. Pillet, M. Vilasi LCSM UMR 755, Faculté des Sciences et techniques UHP – Nancy I Bd des Aiguillettes BP 239 - 54506 Vandoeuvre

polishing before after

INDUSTRIAL EXPECTATIONS Savings in consumption of acids Optimization of the polishing cycles : improvement of the capacity on the existing machines Control of the « strength » of the bath to avoid quality problems as stuck salts or loss of acceptable dimensional tolerances

CONTEXT Solution of HF + H 2 SO 4 + H 2 Si. F 6 (leaching of the glass) HF/H 2 SO 4 ratio : a key factor for the quality of the process AIM OF THE STUDY Determination of the [F-] in a hyper-acidic medium (H 2 SO 4) Determination of [HF] to control ageing of polishing bath Data should be achievable by a simple method

The classical titration method consists on the following reaction: La 3+ + 3 F- => La. F 3(solid) acted at p. H=5 and the determination of the equivalent volume is realized thanks to a potentiometric measurement In the industrial polishing baths, this reaction is hindered by the presence of H 2 Si. F 6 produced by the leaching of the glass This technique cannot be used directly

Laboratory experiments were performed on standard solutions containing : - pure H 2 Si. F 6 - pure HF - a mixture of H 2 Si. F 6 and HF The titration of H 2 Si. F 6 is feasible according to the following reaction : 2 La 3+ + Si. F 62 - + 4 H 2 O => 2 La. F 3(solid) + Si(OH)4 + 4 H+ at p. H= 5, H 2 Si. F 6 is existing as Si. F 62 - The titration of a mixture of H 2 Si. F 6 + HF determines the total amounts of F-I provided by the both acidic species

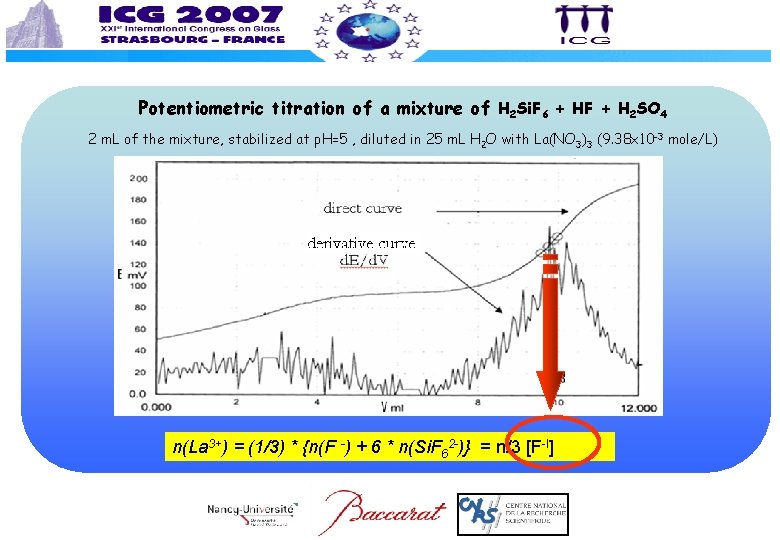
Potentiometric titration of a mixture of H 2 Si. F 6 + HF + H 2 SO 4 2 m. L of the mixture, stabilized at p. H=5 , diluted in 25 m. L H 2 O with La(NO 3)3 (9. 38 x 10 -3 mole/L) n(La 3+) = (1/3) * {n(F -) + 6 * n(Si. F 62 -)} = n/3 [F-I]
![The determination of H 2 Si F 6 or HF in the same bath The determination of [H 2 Si. F 6] or [HF] in the same bath](https://slidetodoc.com/presentation_image/7eb4a4b44ca3b0a49e6fa1ff774aca94/image-8.jpg)
The determination of [H 2 Si. F 6] or [HF] in the same bath needs to use an other technique of titration The easiest titration technique coming in mind is the acid/base method The titration of standard solutions containing H 2 Si. F 6 was studied at the laboratory

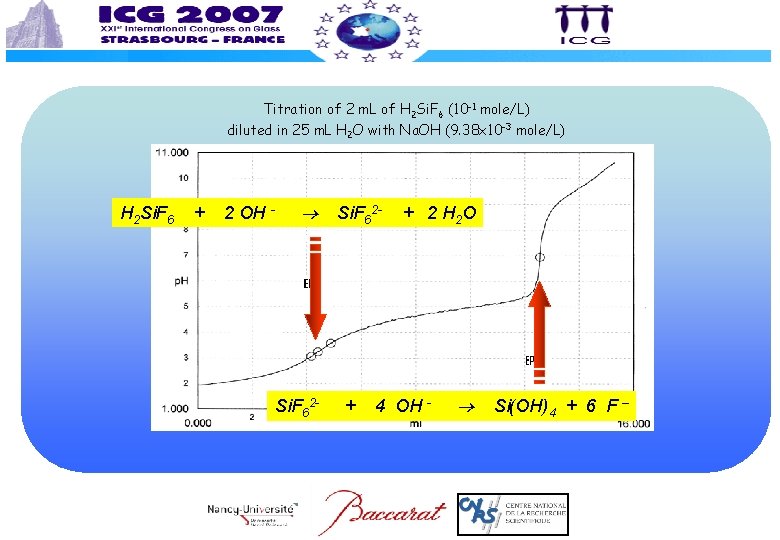
Titration of 2 m. L of H 2 Si. F 6 (10 -1 mole/L) diluted in 25 m. L H 2 O with Na. OH (9. 38 x 10 -3 mole/L) H 2 Si. F 6 + 2 OH - Si. F 62 - + + 2 H 2 O 4 OH - Si(OH)4 + 6 F –
![H 2 Si F 6 is determinate by the volume difference DVm L [H 2 Si. F 6] is determinate by the volume difference DVm. L =](https://slidetodoc.com/presentation_image/7eb4a4b44ca3b0a49e6fa1ff774aca94/image-10.jpg)
[H 2 Si. F 6] is determinate by the volume difference DVm. L = VEP 2 – VEP 1 DVm. L Titration of a standard mixture of H 2 SO 4 + HF + H 2 Si. F 6 gives a similar curve to the one obtained with pure H 2 Si. F 6 solution Technique is therefore applicable to an industrial bath
![FI F 6 Si F 62 Potentiometric AcidoBasic VEP 3 [F-I] = [F -] + 6 [Si. F 62 -] Potentiometric Acido/Basic VEP 3](https://slidetodoc.com/presentation_image/7eb4a4b44ca3b0a49e6fa1ff774aca94/image-11.jpg)
[F-I] = [F -] + 6 [Si. F 62 -] Potentiometric Acido/Basic VEP 3 DVm. L

CONCLUSIONS Chemical characterization of fluoride in hyperacidic medium can be done with an accuracy of about 10%. This method is validated on industrial solutions for crystal polishing Its simplicity allows the application on the glass making site. It should become a powerful tool to control the ageing of polishing baths

FIRST INDUSTRIAL POSITIVE CONSEQUENCE The “virtuous circle” Decrease of the ratio HF/H 2 SO 4 (by additional H 2 SO 4 and constant HF) corresponds to a lower value of H 2 Si. F 6 in the bath and lower necessary additions of HF to maintain a constant level of active HF in the bath The hypothesis to be checked is the following one: 1. Ions Si. F 62 - react better with the additional H+ than with K+ to form H 2 Si. F 6 instead of K 2 Si. F 6 2. the following reaction permits to re-create active HF: H 2 Si. F 6 + 2 H 2 O => Si. O 2 + 6 HF
