ChemistryWater Hot Spot Review Molecule of Life Molecule




![p. H and [H+] conversions Ex. p. H= 8 or [H+]= 1 x 10 p. H and [H+] conversions Ex. p. H= 8 or [H+]= 1 x 10](https://slidetodoc.com/presentation_image_h2/a5fc5e817f452cd4e6393103bb523ecf/image-18.jpg)


![Acids- Strong versus Weak Acid- donor of [H+] , increases [H+] in sol’n ●Strong Acids- Strong versus Weak Acid- donor of [H+] , increases [H+] in sol’n ●Strong](https://slidetodoc.com/presentation_image_h2/a5fc5e817f452cd4e6393103bb523ecf/image-21.jpg)
![Base- Strong versus Weak ● Base- acceptor of [H+] , dec. [H+] in sol’n Base- Strong versus Weak ● Base- acceptor of [H+] , dec. [H+] in sol’n](https://slidetodoc.com/presentation_image_h2/a5fc5e817f452cd4e6393103bb523ecf/image-22.jpg)





- Slides: 29

Chemistry/Water Hot Spot Review

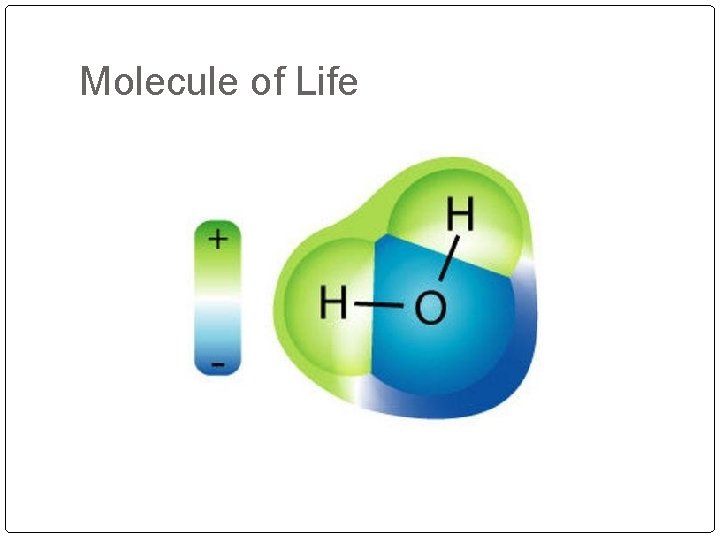
Molecule of Life

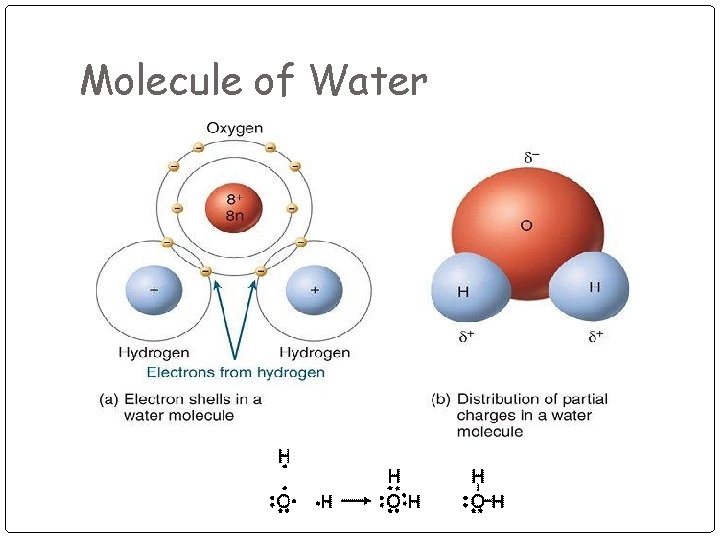
Molecule of Water

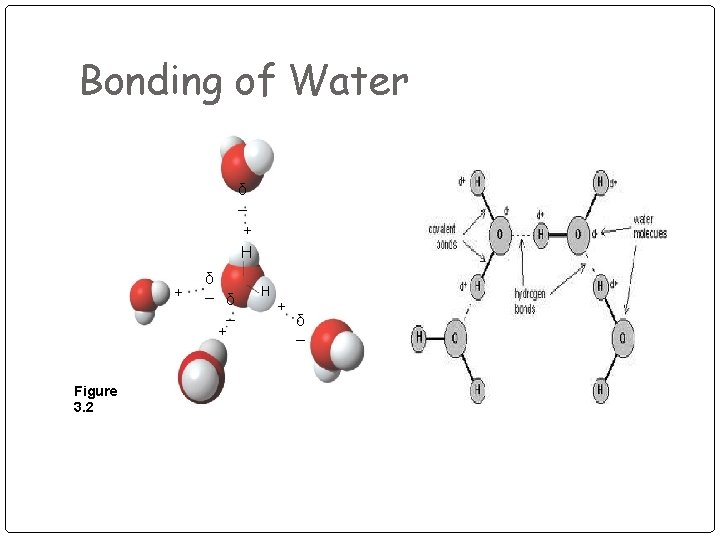
Bonding of Water δ – + H + Figure 3. 2 δ – + H + δ –

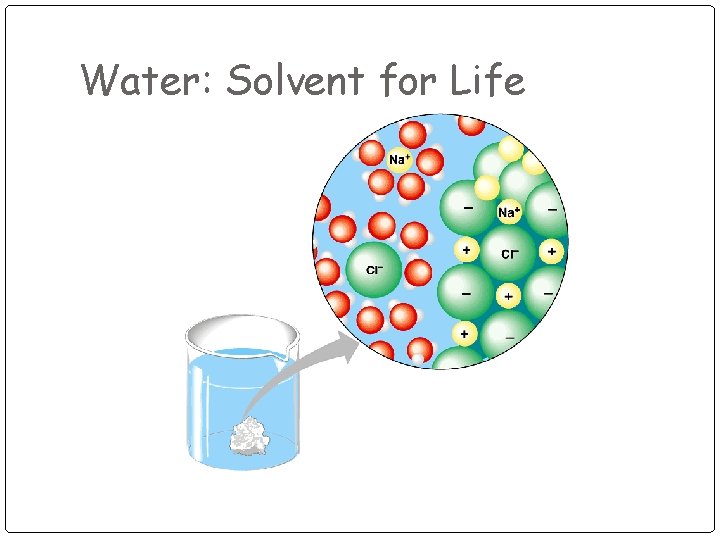
Water: Solvent for Life

Cohesion and Adhesion

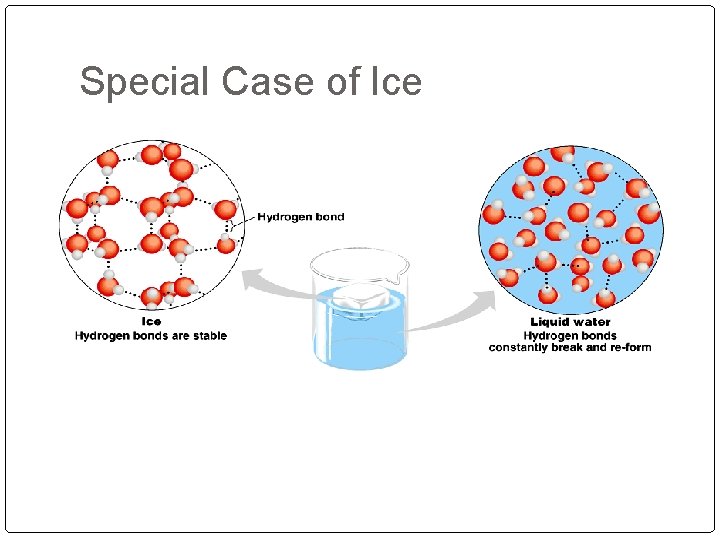
Special Case of Ice

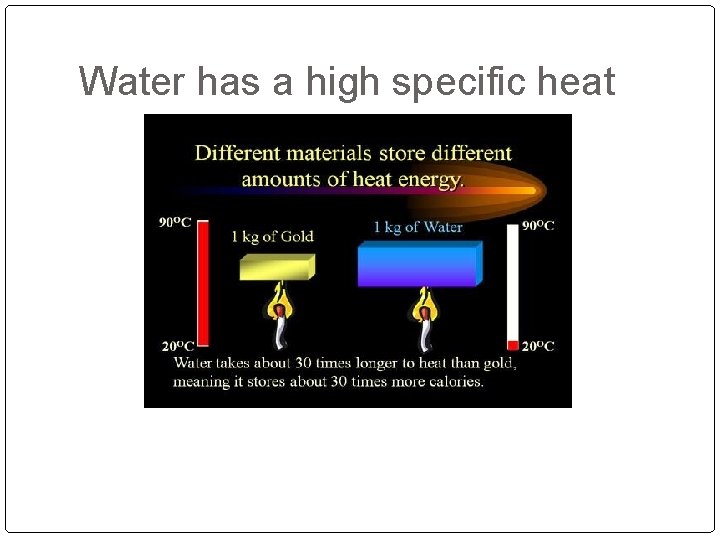
Water has a high specific heat

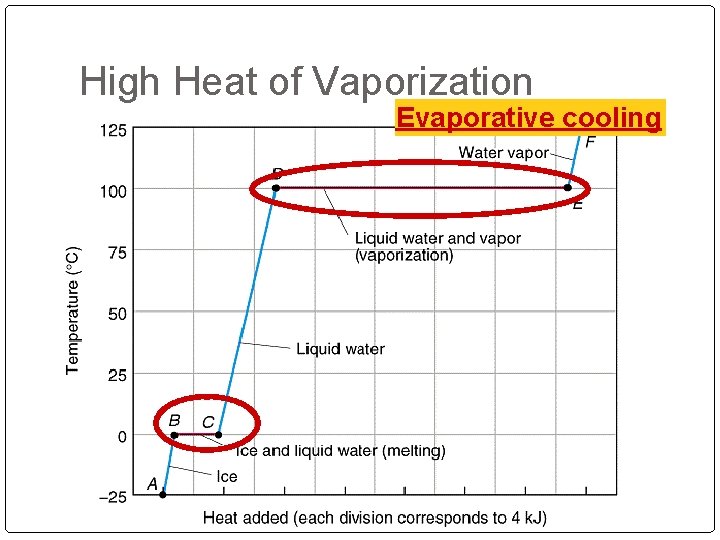
High Heat of Vaporization Evaporative cooling Organisms rely on heat of vaporization to remove body heat

Mr. Anderson Water Review http: //www. youtube. com/watch? v=DVCYl. ST 6 m. YQ&feature=rel ated

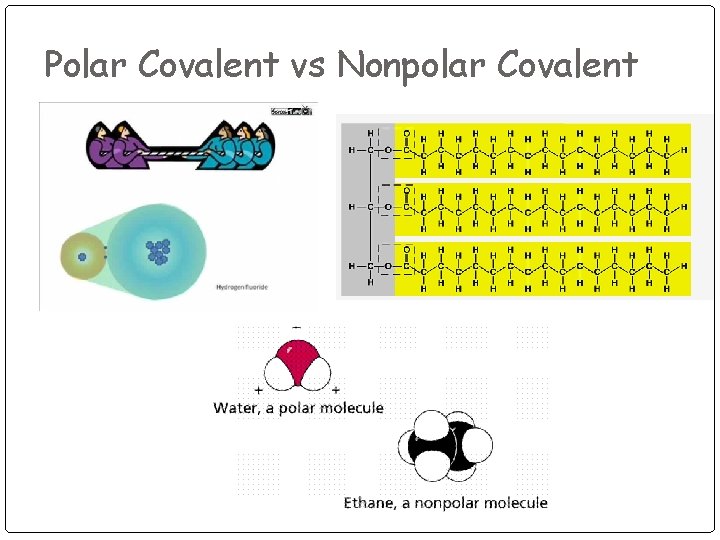
Polar Covalent vs Nonpolar Covalent

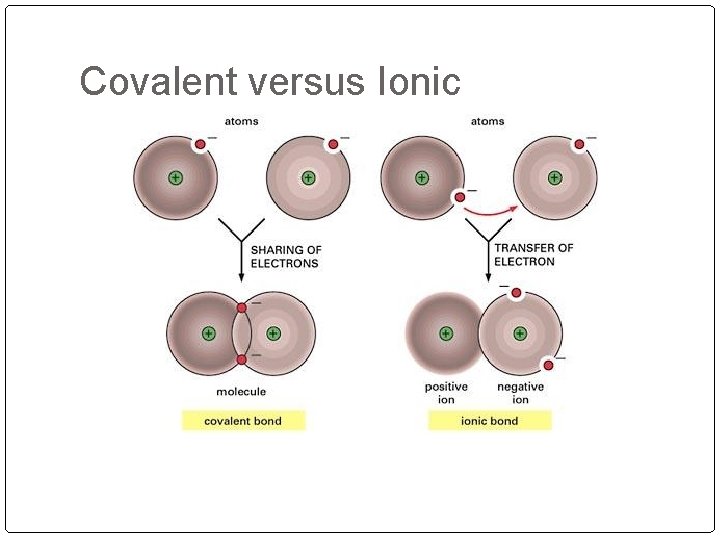
Covalent versus Ionic

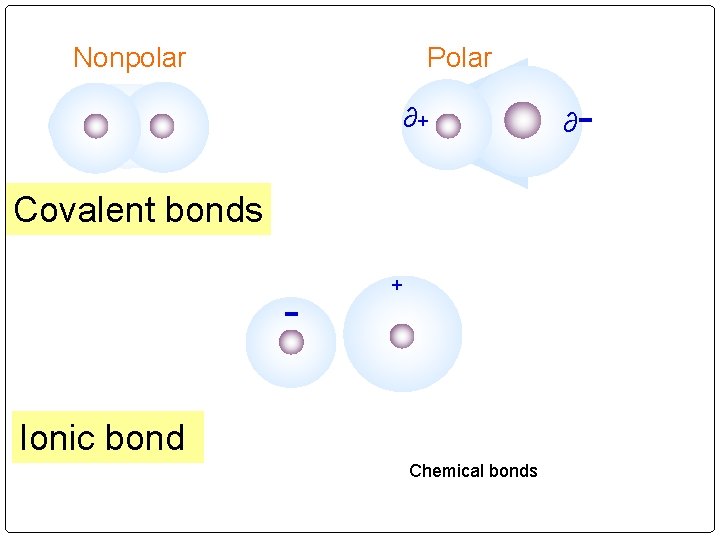
Nonpolar Polar ∂+ Covalent bonds - + Ionic bond Chemical bonds ∂ -

In liquid water, hydrogen bonds continually break and reform

Mr. Anderson p. H Review http: //www. youtube. com/watch? v=V 4 S 1 Kl. Jd. Mb. E&feature=BFa&list=PL 432856 91048 DAD 00


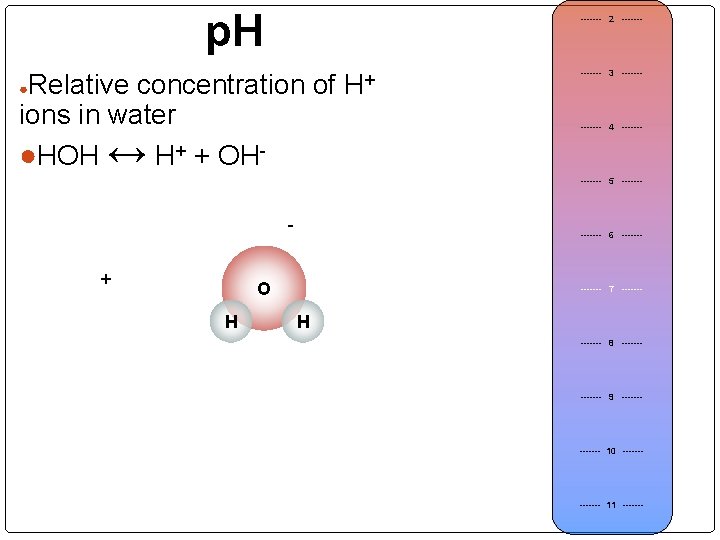
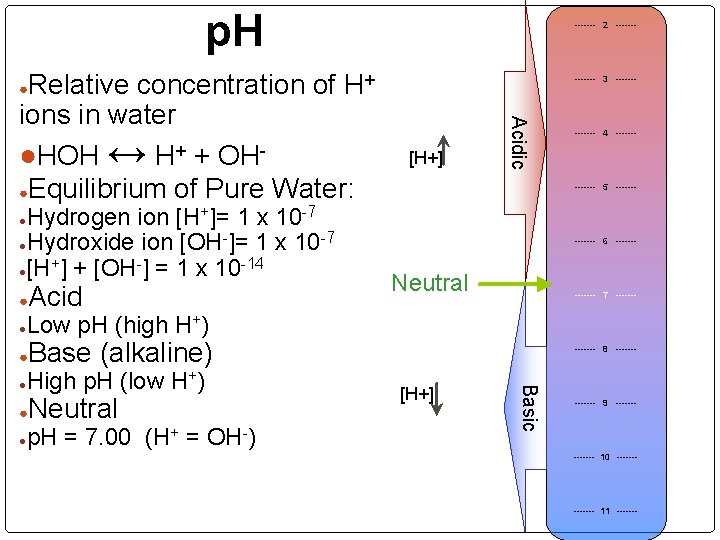
p. H Relative concentration of H+ ions in water ●HOH ↔ H+ + OH●Equilibrium of Pure Water: ------- 2 ------- 3 ------- ● Acidic Hydrogen ion [H+]= 1 x 10 -7 -7 ●Hydroxide ion [OH ]= 1 x 10 + -14 ●[H ] + [OH ] = 1 x 10 [H+] ------- 4 ------- 5 ------- ● ● Acid ------- 6 ------- Neutral ------- 7 ------- Low p. H (high H+) ● ● Base (alkaline) ● Neutral p. H = 7. 00 (H+ = OH-) [H+] Basic High p. H (low H+) ● ------- 8 ------- 9 ------- ● ------- 10 ------- 11 -------
![p H and H conversions Ex p H 8 or H 1 x 10 p. H and [H+] conversions Ex. p. H= 8 or [H+]= 1 x 10](https://slidetodoc.com/presentation_image_h2/a5fc5e817f452cd4e6393103bb523ecf/image-18.jpg)
p. H and [H+] conversions Ex. p. H= 8 or [H+]= 1 x 10 -8 Ex. p. H = 10, [H+]= ? Ex. [H+]= 1 x 10 -8 , p. H? Ex. [H+]= 1 x 10 -4 [OH-]= 1 x 10 -10 Ex. [H+]= 1 x 10 -12 , [OH-]= ? p. H= -log 10 [H+] = 10 -p. H p. OH=-log 10 [OH-] p. H+ p. OH= 14 p. H+ p. OH= 1 x 10 -14

More p. H Ex. p. H= 7, p. OH=7 Ex. p. H=1, p. OH=? Ex. What is the p. H of a solution with [H+]= 1 x 10 -4 ? What is the p. H of a solution with [OH-]= 1 x 10 -4?

p. H=2 is 10 times stronger than p. H=3 p. H =2 is 100 times stronger than p. H=4 p. H=2 is 1000 times stronger than 5 Ex. If p. H is increased from p. H=4 to p. H=2, what does that mean? Ex. If your p. H=4, what is your p. H if you have 100 times less [H+]?
![Acids Strong versus Weak Acid donor of H increases H in soln Strong Acids- Strong versus Weak Acid- donor of [H+] , increases [H+] in sol’n ●Strong](https://slidetodoc.com/presentation_image_h2/a5fc5e817f452cd4e6393103bb523ecf/image-21.jpg)
Acids- Strong versus Weak Acid- donor of [H+] , increases [H+] in sol’n ●Strong Acids- completely dissociated in water ●Weak Acids- do not completely dissociate (Carbonic Acid) H 2 CO 3 HCO 3 - + H+ Carbonic Acid Bicarbonate Ion Hydrogen Ion ●
![Base Strong versus Weak Base acceptor of H dec H in soln Base- Strong versus Weak ● Base- acceptor of [H+] , dec. [H+] in sol’n](https://slidetodoc.com/presentation_image_h2/a5fc5e817f452cd4e6393103bb523ecf/image-22.jpg)
Base- Strong versus Weak ● Base- acceptor of [H+] , dec. [H+] in sol’n 1) Weak Base: reduces [H+] by accepting hydrogen ions NH 3 + H+ NH 4+ ● 2) Strong Base: reduces [H+] by dissociating to form Hydroxide ions [OH-] Na. OH Na+ + OH●

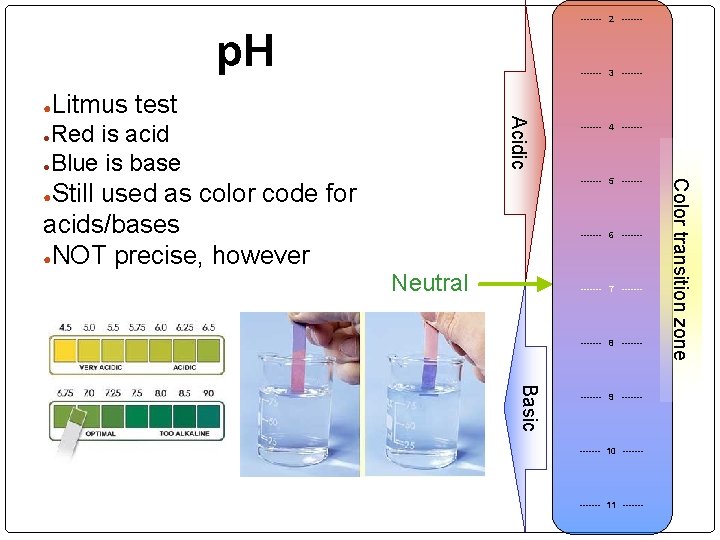
------- 2 ------- p. H ● ------- 3 ------- Acidic Litmus test Red is acid ●Blue is base ● ------- 5 ------- ● ------- 6 ------- Neutral ------- 7 ------- 8 ------- Basic ------- 9 ------- 10 ------- 11 ------- Color transition zone Still used as color code for acids/bases ●NOT precise, however ------- 4 -------

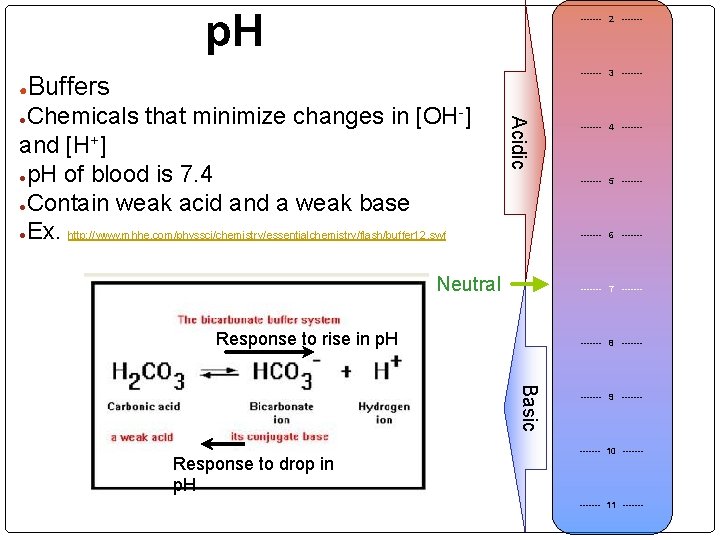
p. H ● ------- 2 ------- 3 ------- Buffers Acidic Chemicals that minimize changes in [OH-] and [H+] ●p. H of blood is 7. 4 ●Contain weak acid and a weak base ●Ex. http: //www. mhhe. com/physsci/chemistry/essentialchemistry/flash/buffer 12. swf ● ------- 5 ------- 6 ------- Neutral ------- 7 ------- Response to rise in p. H ------- 8 ------- Basic Response to drop in p. H ------- 4 ------- 9 ------- 10 ------- 11 -------

M 1 V 1 = M 2 V 2 Ex. How much concentrated 18 M sulfuric acid is needed to prepare 100 m. L of a 2 M solution? M 1 V 1 = M 2 V 2 (18 M) (V 1)= (2 M)(100 m. L) V 1= (2 M)(100 m. L) / (18 M) = 11 m. L

M 1 V 1 = M 2 V 2 Ex. To how much water should 100. m. L of 18 M HCl be added to produce 1. 5 M solution? M 1 V 1 = M 2 V 2 (18 M) (100 m. L)= (1. 5 M)(V 2) V 2= (18 M)(100 m. L) / (1. 5 M) = 1200 m. L 1200 m. L-100 m. L= 1100 m. L ●

0 A. The following are p. H values: cola-2; orange juice 3; beer-4; coffee-5; human blood-7. 4. Which of these liquids has the highest molar concentration of OH-? 1. 2. 3. 4. 5. cola orange juice beer coffee human blood

B. If the p. H of a solution is increased from p. H 8 to p. H 9, it means that the concentration of H+ is 10 times greater than what it was at p. H 8. 2. concentration of H+ is 100 times less than what it was at p. H 8. 3. concentration of OH- is 10 times greater than what it was at p. H 8. 4. concentration of OH- is 100 times less than what it was at p. H 8. 5. concentration of H+ is greater and the concentration of OH- is less than at p. H 8. 1.

C. Acid precipitation has lowered the p. H of a particular lake to 4. 0. What is the hydroxide ion concentration of the lake? 1. 2. 3. 4. 5. 10 -7 M 10 -4 M 10 -10 M 10 -14 M 10 M