Chapter 2 Chemistry of Life Section 2 Properties


























































- Slides: 66

Chapter 2: Chemistry of Life Section 2 Properties of Water

Bellwork • • • Turn in your lab and conclusions stapled together from yesterday Person #3 get textbooks for your group Person #1 get 1 piece of colored paper for each person at your table Person #4 get an admit slip for each person at your table. Complete the Admit slip using the following pictures:

Agenda Essential Question? • What properties of water make it so important? Learning Goal: • I will be able to – Describe the special properties of water and explain its importance. – Explain the formation of hydrogen bonds • Notes on Properties of Water • HW: – study for 2. 2 quiz tomorrow at the end of class – 2. 2 worksheet due tomorrow

Frayer Fold Notes • You need ONE piece of paper folded “Frayer Style” • Turn to page 40 in your book.

Water & Its Importance • Why is water important for our bodies? • Water makes up 70 to 95 percent of most organisms.

Water Characteristics Page 40

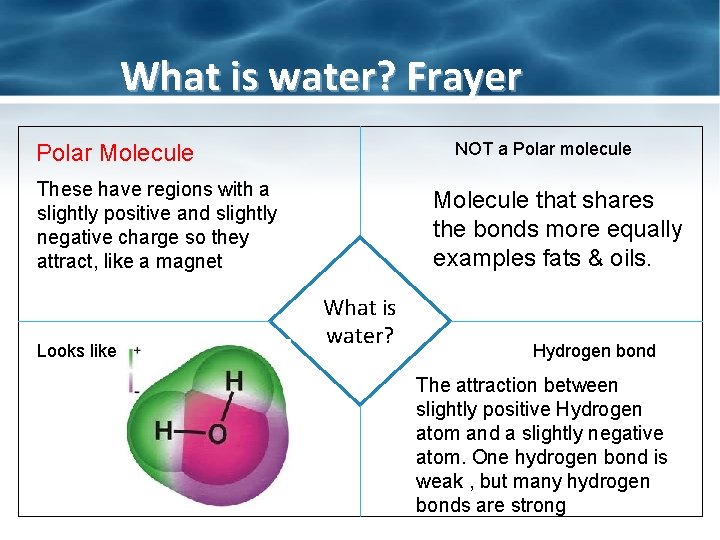
What is water? Frayer NOT a Polar molecule Polar Molecule These have regions with a slightly positive and slightly negative charge so they attract, like a magnet Looks like Molecule that shares the bonds more equally examples fats & oils. What is water? Hydrogen bond The attraction between slightly positive Hydrogen atom and a slightly negative atom. One hydrogen bond is weak , but many hydrogen bonds are strong

Hydrogen bonding Demonstration • How many drops of water do you think will fit on a penny before the water breaks? • Write it on the corner of your paper and lets see who’s closest. • http: //www. youtube. com/watch? v=tv 4 Jrc 0 6 y. LA&feature=related&safety_mode=true& persist_safety_mode=1&safe=active

Water Properties Cohesion High Specific Heat Water resists temperature changes which helps regulate cell temperatures. Adhesion Properties of water Water is attracted to other substances-creates capillary action Ex. transpiration-process when plants and trees remove water from the soil Water sticks to each other- this creates surface tension. Solvent Water is usually present in a greater amount and its polarity helps dissolve other substances.

Properties of water • High Specific Heat & Solvent http: //www. youtube. com/watch? v=QH 1 yphfgf. FI&safety_mode=tru e&persist_safety_mode=1&safe=active Return

Cohesion … Return Helps insects walk across water

Adhesion Causes Capillary Action Which gives water the ability to “climb” structures

Adhesion Also Causes Water to … Form spheres & hold onto plant leaves Return Attach to a silken spider web

Cool Water Trick • http: //www. youtube. com/watch? v=7 cta. A 2 m ERz. I&feature=related&safety_mode=true&p ersist_safety_mode=1&safe=active

Another cool water video https: //www. youtube. com/watch? v=KFPvd. Nbft. OY

Quickwrite-Exit Slip • 2 minutes • REMEMBER you are just trying to WRITE for the ENTIRE TWO MINUTES!!! • Explain what you learned about water today.

Water Characteristics Day 2 Sept 10 th-11 th

• • • No BW due to sub Collect 2. 2 Worksheet Review Notes “Adhesion and Solvent” Properties of water Paper clip Lab Complete Section 2. 3 of Workbook

Water Characteristics Day 3 Sept 14 th

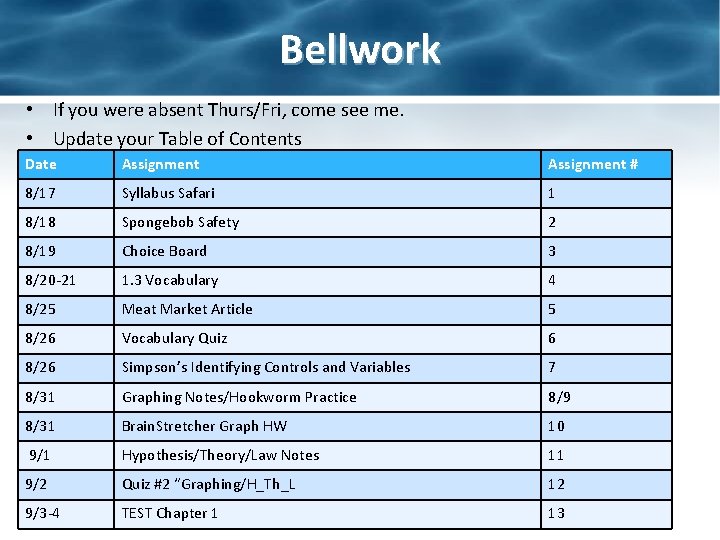
Bellwork • If you were absent Thurs/Fri, come see me. • Update your Table of Contents Date Assignment # 8/17 Syllabus Safari 1 8/18 Spongebob Safety 2 8/19 Choice Board 3 8/20 -21 1. 3 Vocabulary 4 8/25 Meat Market Article 5 8/26 Vocabulary Quiz 6 8/26 Simpson’s Identifying Controls and Variables 7 8/31 Graphing Notes/Hookworm Practice 8/9 8/31 Brain. Stretcher Graph HW 10 9/1 Hypothesis/Theory/Law Notes 11 9/2 Quiz #2 “Graphing/H_Th_L 12 9/3 -4 TEST Chapter 1 13

Agenda • Review Chapter 1 Test – Who’s missing it? – Retest Forms • • Review Properties of Water Notes Review Properties of Water Lab HW: Study for you Properties of Water QUIZ tomorrow! Review 2. 3 Workbook page (did you get this? ) – Due Thursday/Friday

Water Characteristics Day 4 Sept 15 th

Bellwork • If you were absent come see me. • Turn in your Properties of Water Lab and 2. 2 worksheet – If you do not have these complete take out a piece of paper and write a not with the following: • Your name, Name of Assignment, Today’s date, Reason Not completed • Pick up a new bellwork sheet, hole punch it and place it in your bellwork section of your binder. • Complete #1 on the new bellwork sheet • Study for your quiz when finished.

Essential Question? • What properties of water make it so important? Learning Goal: • I will be able to – Describe the special properties of water and explain its importance. – Explain the formation of hydrogen bonds Agenda • Review 2. 2 worksheet • Quiz on Properties of Water • Begin Macromolecules Concept Definition Maps • HW: 2. 3 Worksheet due Thursday

What do you know? Using one post it note per piece of information write down what you know about the 4 macromolecules: Carbohydrates, Nucleic Acids, Lipids, Proteins and what you know about Carbon.

Bellwork 9/16 Turn in Properties of Water and 2. 2 Worksheet if you haven’t done so. Pick up a Yellow sheet, complete it and turn it in if you haven’t done so AND don’t have them complete. If you were absent/testing check the absent file for your work. Complete #3 of your bellwork NOT #2.

Essential Question: What are carbon compounds and why are they important for living things? Learning Goal: I will be able to: Explain why carbon is valuable to living things Compare and Contrast the structure and function of the different types of macromolecules

Agenda Video on Macromolecules Begin the Macromolecules Concept Definition folder HW: Complete 2. 3 Worksheet

Chemistry of Life Video https: //www. youtube. com/watch? v=IJ 7 x. OSCEm. Zw&list=PLw. L 0 Myd 7 Dk 1 Eo. Pe. Klyu. KR 7 obb 5 OKLSn. TN

Exit Ticket… Answer the following in complete sentences based on what you’ve learned today… Why is Carbon so important to living things? What question do you still have from today’s lesson?

Macromolecule Lab and Foldable 9/17 -18

Bellwork 9/17 -18 If you were absent come see me to get your make up work Turn in your 2. 3 Worksheet – If you do not have this pick up and fill out a yellow sheet and turn it in. Also, Properties of Water Lab should be turned in and 2. 2 worksheet. Otherwise, get a Yellow Sheet! Take out your Foldable from yesterday. For example, group 1 gets supplies from the cabinet labeled group 1.

Essential Question: What are carbon compounds and why are they important for living things? Learning Goal: I will be able to: Explain why carbon is valuable to living things Compare and Contrast the structure and function of the different types of macromolecules

Agenda 9/17 -18 Macromolecule Foldable Macromolecule Lab HW: Finish Foldable and Lab – due Monday!

Lab You will be timed! 5 minutes to read and complete a section in your foldable 5 minutes to complete the lab for that section of your foldable Rotate tables and lab stations

Exit slip Answer the following: Did the lab help or hinder your understanding of Macromolecules? Explain! What question do you still have from today’s lesson?

Macromolecule Review 9/21

Bellwork 9/21 If you were absent check the absent filing cabinet. READ and DO what your slip says! Turn in your COMPLETED Ch 1 RETEST Packet, Macromolecules LAB and Foldable-if you don’t have it done fill out a Yellow Sheet and turn it in today! Bring your work tomorrow. Pick up your new bellwork sheet and complete #1 for this week.

Essential Question: How are the macromolecules similar and different in their structure and function? Learning Goal: I will be able to: Compare and Contrast the structure and function of the different types of macromolecules

Agenda 9/21 Macromolecules Word Sort and Analogy Poster HW: Macromolecules Analogies Poster– due Tuesday

Word Sort As a team classify the characteristics with their appropriate macromolecule. When finished, get Ms. Black’s attention to be approved. Fill out your WORD Sort Sheetplace in binder

Analogy Begin Working on your Macromolecule Analogies. For Example, Nucleic acids store information (function)which is like a __computer hard drive______ because it also stores information. Two examples of Nucleic acids are ____DNA and RNA____.

Exit ticket Put your name on the slip of paper Answer the Essential question. Be specific and use proper terminology. If you are still confused about something, tell me. Place it in a pocket when you are done.

Enzymes Section 2. 4 and 2. 5

Bellwork 9/22 • If you were absent check the filing cabinet. READ and DO what it says! • Turn in your Macromolecule Analogies-complete and turn in a YELLOW SHEET if you don’t have it and bring it tomorrow! • Complete #2 of you new bellwork sheet for this week. • STUDY for your Quiz- remember to focus on the structure, function, monomers, polymers and examples.

Enzymes Essential Question: • How do enzymes impact living things and what affects the way enzyme work? Learning Goal: • Explain how enzymes work by lowering the activation energy of a reaction. • Identify and explain how the factors like p. H and temperature affect enzymes.

Agenda 9/22 • QUIZ-Macromolecules • Begin Enzyme notes (section 2. 4 and 2. 5) • HW: Concept Maps

Enzymes- Day 2 9/23

Bellwork 9/23 • If you were absent check the filing cabinet. READ and DO what it says! • Take out your Enzyme Notes from yesterday and place on your desk. • Complete #3 of you new bellwork sheet for this week.

Enzymes Essential Question: • How do enzymes impact living things and what affects the way enzyme work? Learning Goal: • Explain how enzymes work by lowering the activation energy of a reaction. • Identify and explain how the factors like p. H and temperature affect enzymes.

Agenda • Quiz Trade Review • Review Enzyme Notes • Exit Ticket • HW: Enzyme Concept Map and Study for your Quiz tomorrow! (All of chapter 2)

Enzyme Notes 1. Copy the overall chemical equation for the process that changes glucose and oxygen into carbon dioxide and water. Circle the reactants and draw a box around the products. 6 O 2 + C 6 H 12 O 6 6 CO 2 + 6 H 2 O

Enzyme Notes 2. What is required to break bonds in the reactants? ▫ Energy is required to break bonds in the reactants. This is called BOND ENERGY which is the amount of energy needed to break a bond between two atoms.

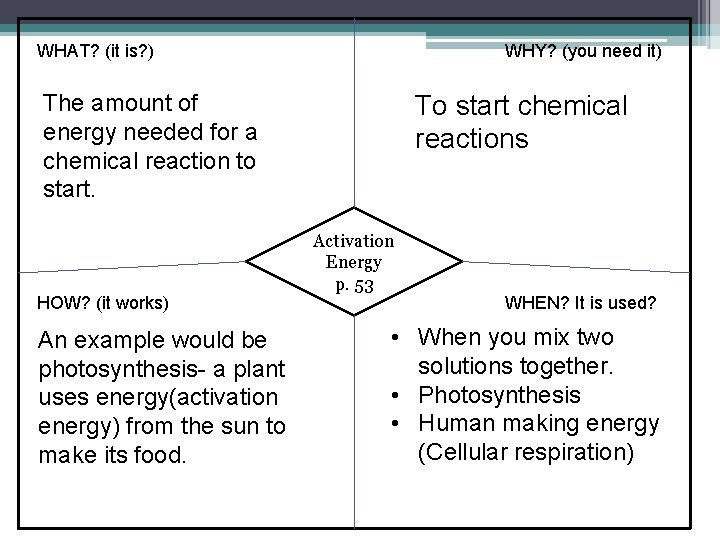
WHAT? (it is? ) WHY? (you need it) The amount of energy needed for a chemical reaction to start. HOW? (it works) An example would be photosynthesis- a plant uses energy(activation energy) from the sun to make its food. To start chemical reactions Activation Energy p. 53 WHEN? It is used? • When you mix two solutions together. • Photosynthesis • Human making energy (Cellular respiration)

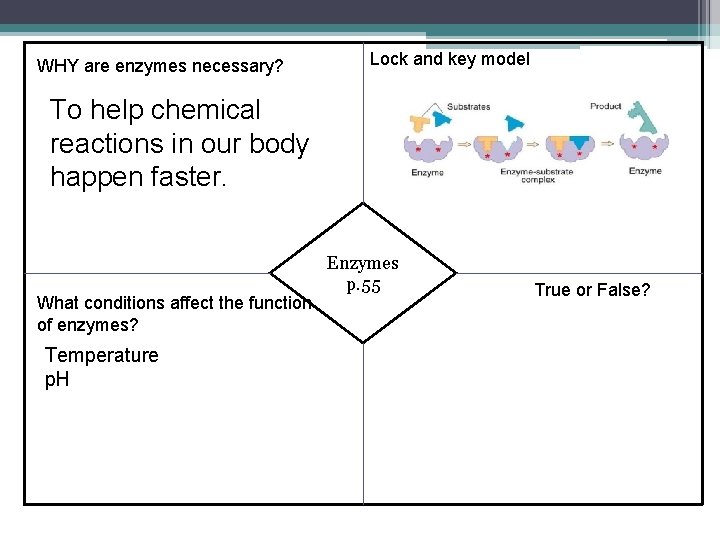
Definitions Word Catalyst Definition A substance that decreases or lowers the activation energy needed to start a chemical reaction; it increase the rate of the reaction Enzyme A protein that acts as a catalyst for chemical reactions Substrate Specific reactants that an enzyme binds with


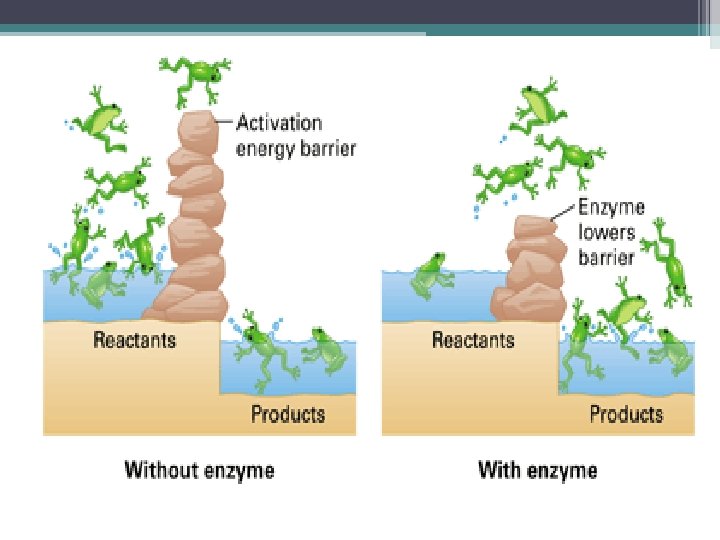
WHY are enzymes necessary? Lock and key model To help chemical reactions in our body happen faster. What conditions affect the function of enzymes? Temperature p. H Enzymes p. 55 True or False?

True or False 1. Enzymes are involved in almost every chemical process in an organism True 2. Enzymes are catalysts True 3. All enzymes are proteins True 4. The shape of the enzyme is critical to its function True 5. Enzymes are specific True 6. Enzymes are used up during a chemical reaction. False! 7. Enzymes remain unchanged after a chemical reaction. True 8. Enzyme names end in “ade” False! “ase”

Video • http: //highered. mheducation. com/sites/007249 5855/student_view 0/chapter 2/animation__ho w_enzymes_work. html

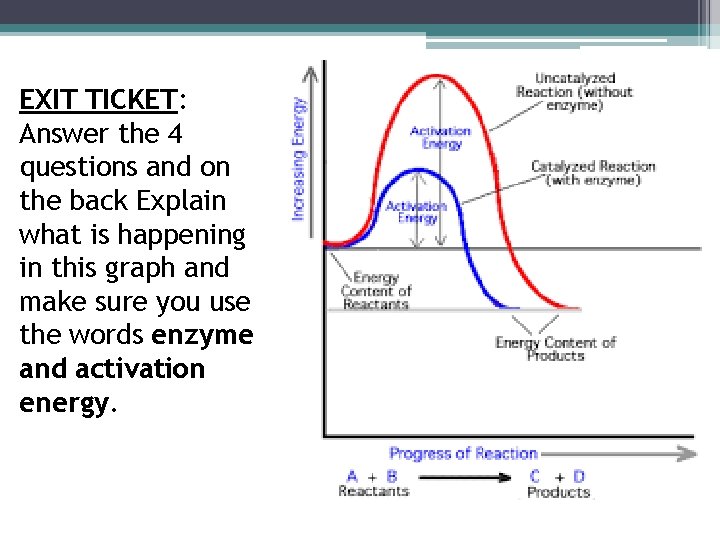
EXIT TICKET: Answer the 4 questions and on the back Explain what is happening in this graph and make sure you use the words enzyme and activation energy.

Enzymes- Day 3 9/24 and 9/25

Bellwork • If you were absent come see me • Turn in your concept Maps-get a yellow sheet, fill it out and turn it in if you don’t have them completed. Bring them competed next class. • Complete bellwork #3. • Study for your TEST

Agenda • Quiz Trade • Chapter 2 TEST • Toothpickase Enzyme Lab • HW: NONE-go enjoy your weekend

Enzymes Essential Question: • How do enzymes impact living things and what affects the way enzyme work? Learning Goal: • Explain how enzymes work by lowering the activation energy of a reaction. • Identify and explain how the factors like p. H and temperature affect enzymes.

Toothpickase Lab Complete the lab and answer the questions

Exit Ticket • How does a change in the concentration of substrates and the change in temperature affect and enzymes ability to function?