AVOGADROS LAW WHAT IS AVOGADROS LAW v Avogadros









- Slides: 10

AVOGADRO’S LAW

WHAT IS AVOGADRO’S LAW v Avogadro’s Principle – equal volumes of gases at the same temperature and pressure contain equal numbers of particles

AVOGADRO’S FORMULA * n represents the amount of gas V 1= V 2 n 1 n 2 This is a direct relationship! v So if the amount of gas increases, then the volume will increase ______. If the amount of gas decreases, then the decreas e volume will _____.

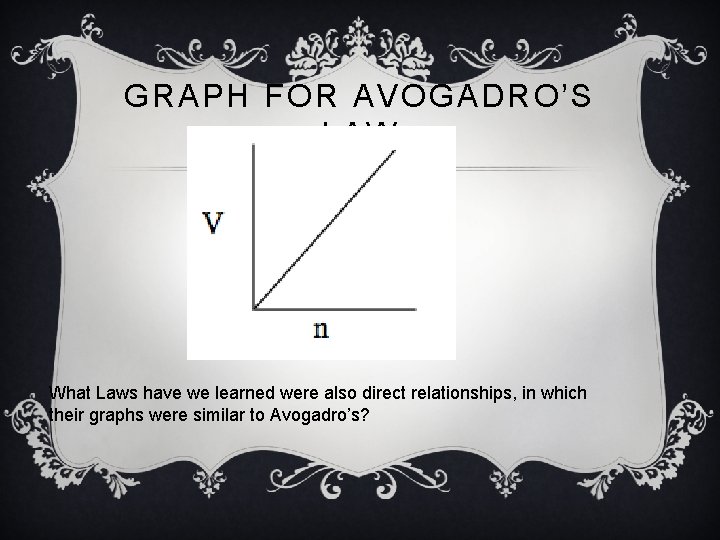
GRAPH FOR AVOGADRO’S LAW What Laws have we learned were also direct relationships, in which their graphs were similar to Avogadro’s?


AVOGADRO’S LAW v. Molar Volume – for a gas is the volume that one mole of that gas occupies at STP Avogadro showed experimentally that 1 mole of any gas will occupy a volume of 22. 4 L at STP **Conversion Factor: 1 mol (any gas) = 22. 4 L at STP **

AVOGADRO’S LAW: EXAMPLE 1 Calculate the volume that 0. 881 moles of oxygen gas at STP will occupy.

AVOGADRO’S LAW: EXAMPLE 1 ANSWER v Formula: V 1 = V 2 n 1 V 1 = ? L n 1 = 0. 881 mol O 2 V 2 = 22. 4 L n 2 = 1 mol O 2 n 2 Remember, at STP: 22. 4 L/mol V 1. 881 mol O 2 = 22. 4 L 1 mol O 2 After cross multiplying you end up with ---------V 1(1 mol O 2) = (22. 4 L)(. 881 -----mol O 2) -----1 mol O 2 19. 7 L 1 mol O 2 V 1 = ______

AVOGADRO’S LAW: EXAMPLE 2 How many grams of N 2 will be contained in a 2. 0 L flask at STP? (Remember- one mole WEIGHS the formula mass in grams)

AVOGADRO’S LAW: EXAMPLE 2 ANSWERRemember, at STP: 22. 4 L/mol v Formula: V 1 = V 2 n 1 First , solve for the number of moles of N 2: 2 L N 2 | 1 mol N 2 | 22. 4 L N 2 =. 089 n 2 V 1 = 2. 0 L n 1 = ? g N 2 V 2 = 22. 4 L n 2 = 1 mol N 2 Then, use dimensional analysis to convert from moles of N 2 to grams of N 2: . 089 mol N 2 | 28. 014 g N 2 = | 1 mol N 2 2. 5 g N 2