The Mole and Avogadros Number The Mole Avogadros

The Mole and Avogadro’s Number • The Mole • Avogadro’s Number • Molar Mass • Finding n • Finding the # of atoms • Working with formulas Practice

We sometimes give a name to a specific amount Can you think of any more? ? ?

The Mole… • We need a relationship between the mass of a sample of an element/compound and the number of atoms/molecules in that sample • For this, we use the MOLE 1 mole (mol) = 6. 02 x 1023 particles = Avogadro’s Number

Interesting Facts…. • If one mole of pennies were distributed equally among 6 billion people, each person would get 1 x 1014 pennies or 1 x 1012 dollars (1, 000, 000 = 1 trillion dollars)! • If Avogadro’s number of sheets of paper were divided into a million equal piles, each pile would be so tall that it would stretch from Earth to the Sun and beyond. • In order to obtain Avogadro’s number of grains of sand, you would have to dig the entire surface of the Sahara desert to a depth of 2 m. The Sahara desert has an area of 8 x 106 km 2.

Molar Mass • The molar mass of anything is the mass of 1 mole of that thing in grams – Coins, chickens, computers, students, etc. • 1 mole 12 C atoms = 6. 022 x 1023 atoms = 12. 01 g • 1 mole 15 Mg atoms = 6. 022 x 1023 atoms = 24. 305 g For any element The atomic mass (amu) = molar mass (grams/mol)

What is the mass of one mole of: S C Hg Cu Fe
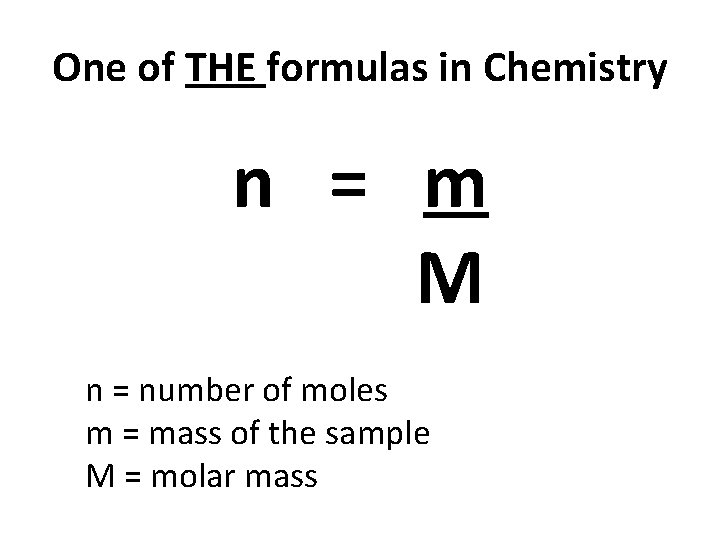
One of THE formulas in Chemistry n = m M n = number of moles m = mass of the sample M = molar mass

Check Your Understanding • Determine the amount of substance in a mass of 10. 0 g of iron? • Given: m. Fe = 10. 0 g • Required: amount of substance, n = ? mol MFe = 55. 85 g/mol n = m/M

Practice Questions 1. 2. 3. 4. How many moles of Na are in 42 g of Na? How many moles of O are in 8. 25 g of O? How much does 2. 18 mol of Cu weigh? What is the mass of 0. 28 mol of iron? n=m M Answers 1. 1. 8 mol Na 2. 0. 516 mol O 3. 139 g Cu 4. 16 g Fe

The Number of Atoms • How do you find the number of atoms? n=m/M • What does n represent? – The number of moles of a substance • What is a mole? – 1 mole = 6. 02 x 1023 particles (Avogadro's constant)

Easier • Remember the mole represents a number, a really big number, but it’s just a number. The same as a dozen = 12 or a pair = 2. Therefore, for now, let’s let n = a dozen 1) How many dozen eggs do you have if you have 4 eggs? • n = 4/12 = 0. 33 dozen • We can say that n = number of eggs/total needed for a dozen 2) How many dozen eggs do you have if you have 18 eggs? • n = 18/12 = 1. 5 dozen 3) How many dozen eggs do you have if you have 12 eggs? • n = 12/12 = 1 dozen

n = # of things total needed • When looking at moles, we do the same thing, only – The # of things = the # of particles – The total needed = Avogadro’s constant = 6. 02 x 1023 – How many particles do you need to get 1 dozen? • 12 – How many particles do you need to get 1 mole? • 6. 02 x 1023

Check Your Understanding: Find the # of particles/atoms 1. How many atoms are in 7. 2 mol of chlorine? 2. How many atoms are in 36 g of bromine? 3. How many moles are in 1. 0 x 109 atoms? n = # of particles 6. 02 x 1023 Answers: 1. 4. 3 x 1024 Cl atoms 2. 2. 7 x 1023 Br atoms 3. 1. 7 x 10 -15 mol

Two Step Calculations • Given the number of particles, the mass of the substance can be determined. Particles moles Mass • Given the mass, the number of particles can be determined Mass moles Particles

How many atoms are in 0. 551 g of potassium (K)? • Write down what you’re given and what you need and include the units Given: mk = 0. 551 g, Mk = 39. 10 g/mol Avogadro’s constant (AC) = 6. 02 x 1023 atoms/mol Needed: Number of atoms Equations: n = m / M n = # atoms/AC Solve for n in (1) and substitute it into (2) Substitute: (1) n = 0. 551 g 39. 10 g/mol (2) n = # atoms 6. 02 x 1023 Solve: n = 0. 01409 # atoms = 0. 01409 x 6. 02 x 1023 # atoms = 8. 4822 x 1021 atoms = 8. 48 x 1021 atoms

Molar Mass • The molar mass of a compound is found by adding together the molar masses of all of its elements, taking into account the number of moles of each element present. • Recall: atomic mass = molar mass if there is 1 mole of substance • Molecular mass is the addition of each atomic mass within a compound • e. g. H 2 = 1. 008 + 1. 008 (we don’t round!) = 2(1. 008) = 2. 016 amu • If there is 1 mole of H 2 (6. 02 x 1023 H 2) then the molar mass of the compound = 2. 016 g/mol

Check Your Understanding: What is the molar mass? 1) H 2 O 2) CO 2 3) CO 4) C 6 H 12 O 6 Answers: 1) 18. 015 g/mol 2) 44. 009 g/mol 3) 28. 0101 g/mol 4) 180. 152 g/mol
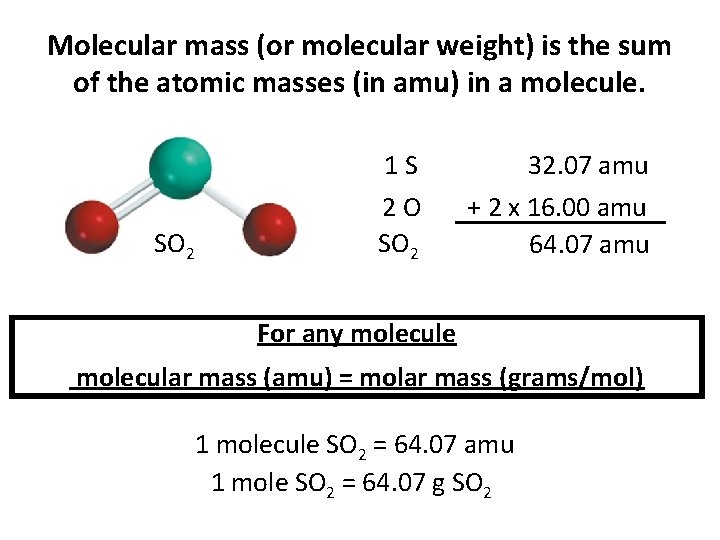
Molecular mass (or molecular weight) is the sum of the atomic masses (in amu) in a molecule. SO 2 1 S 32. 07 amu 2 O SO 2 + 2 x 16. 00 amu 64. 07 amu For any molecule molecular mass (amu) = molar mass (grams/mol) 1 molecule SO 2 = 64. 07 amu 1 mole SO 2 = 64. 07 g SO 2
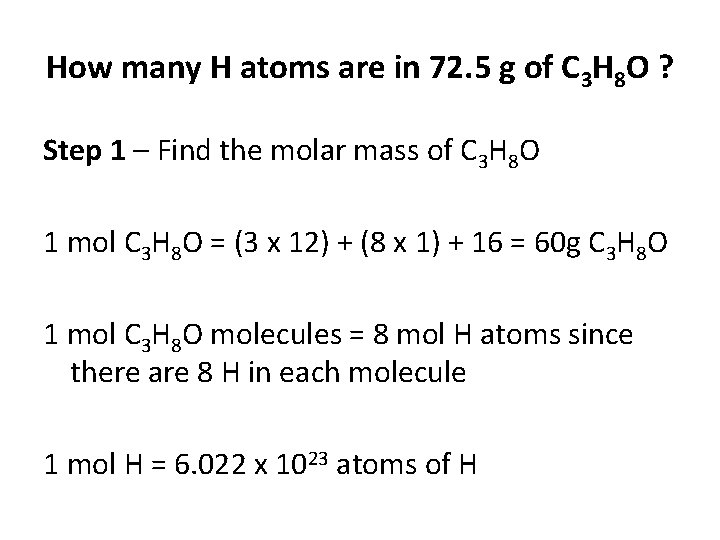
How many H atoms are in 72. 5 g of C 3 H 8 O ? Step 1 – Find the molar mass of C 3 H 8 O 1 mol C 3 H 8 O = (3 x 12) + (8 x 1) + 16 = 60 g C 3 H 8 O 1 mol C 3 H 8 O molecules = 8 mol H atoms since there are 8 H in each molecule 1 mol H = 6. 022 x 1023 atoms of H

How many H atoms are in 72. 5 g of C 3 H 8 O ? Step 2 – Find the number of moles (n) of H n = m/M n. C 3 H 8 O n = #atoms/6. 02 x 1023 atoms/mol = m/M = 72. 5 g / 60 g/mol = 1. 208 mol n. H = (1. 208 mol C 3 H 8 O)(8 mol H) = 9. 66 mol H

How many H atoms are in 72. 5 g of C 3 H 8 O ? n. H = 9. 66 mol H Step 3 – Find the number of atoms of H using n. H n = # atoms / 6. 02 x 1023 atoms/mol # atoms = n x 6. 02 x 1023 atoms/mol # atoms = (9. 66 mol)(6. 022 x 1023 atoms/mol) # atoms = 5. 81 x 1024 H atoms

The Mole
- Slides: 22