U S Food and Drug Administration Office of


























- Slides: 28

U. S. Food and Drug Administration Office of Regulatory Affairs Manufactured Food Regulatory Program Standards (MFRPS-ISO Sampling Agreements) Michelle D. Motsinger MFRPS Specialist Office of Partnerships 1

Objectives • Provide background on ISO Agreements & MFRPS • Provide an overview of the requirement for sampling agreements • Discuss the importance of these agreements • Detail some of the requirements • Provide resources • Provide an update & next steps 2

ISO/IEC 17025: 2005 State Laboratory Accreditation • Purpose: Microbiological and chemical food analyses performed on behalf of State manufactured food regulatory programs conducted within the scope of an ISO/IEC 17025: 2005 accredited laboratory • 30 programs participating • Lab programs must: – Submit a self assessment of laboratory accreditation progress/status, and a cost proposal – Provide quality management personnel to ensure accreditation success – Develop an Action Plan – Develop a Proficiency Testing Plan – Obtain training necessary to obtain accreditation 3 – Obtain and maintain accreditation

ISO Funding Background • 30 State Labs currently have the funding • Requirement for sampling agreement deliverable was added to the NGAs in 2013 in year 2 of the funding • This new deliverable will be required moving forward 4

Manufactured Food Regulatory Program Standards 5

MFRPS Background • Manufactured Food Regulatory Program Standards • Arose from FDA OIG Audit in 2000 • 10 Standards for Improvement of State Manufactured food programs • 40 State Programs enrolled/participating • FDA supports via staff assistance and funding 6

Manufactured Food Regulatory Program Standards (MFRPS) • 10 Standards to establish a uniform foundation for the design and management of state programs responsible for regulating food plants • Institute a holistic quality assurance and standardization program • FDA Program-Assessment Validation Audits (PAVAs) will be conducted at 18, 36, and 60 months Program Standards 1. 2. 3. 4. 5. Regulatory Foundation Training Inspection Program Inspection Audit Program Food-related Illness and Outbreaks and Response 6. Compliance and Enforcement 7. Industry and Community Relations 8. Program Resources 9. Program Assessment 10. Laboratory Support 7

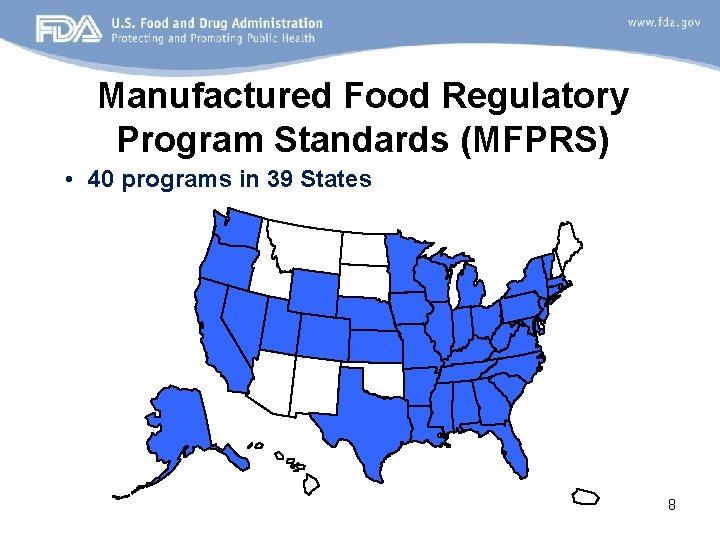
Manufactured Food Regulatory Program Standards (MFPRS) • 40 programs in 39 States 8

Why sample food? • It is the only way to: – isolate/identify contaminants in food – determine the safety of the food – ensure the food is what the product label declares • Our public health system demands a safe food supply • Food may be sampled due to consumer complaint, reported illness, routine inspection/surveillance 9

Why ISO/MFRPS and Sampling? • Propels State food laboratory programs forward • Enhanced communications amongst state agencies • Integrated planning • Advance MFRPS and ISO Sampling • Standardized food sampling methods • An Integrated Food Safety System needs to include sampling and State laboratories • Is a great tool for those with and without ISO funding 10

Importance of Sampling Agreement • Supports laboratory in obtaining or expanding scope of ISO 17025 accreditation • Supports manufactured food program in achieving conformance with MFRPS • Meets the goals of the RFA – Lab commitment to analyze surveillance and emergency samples – Sharing lab results through e. LEXNET – Increase national lab capacity and enhance efforts to protect the food supply 11

How are the ISO Sampling Agreements & MFRPS Connected? • ISO standards (Sampling in particular) relate to several MFRPS Standards - Specifically • Standard 3 – Inspections Program • Standard 4 – Inspection Audit Program • Standard 5 – FBI and Outbreak Response • Standard 8 – State Resources • Standard 10 – Laboratory Support 12

ISO Sampling Plan Requirement • Requirements are written into the ISO Cooperative Agreement – Charges the program to develop & execute a detailed sampling agreement – Requires the plan to be developed in conjunction with the State MFRPS supported by the lab and FDA – Recommends agreements should outline the minimum # of samples, types of analysis and frequency of testing annually, including 13 sample collection methods

Getting Started… • Requires proactive dialogue, communication & planning between the food safety program and the laboratory • Requires agencies working together to develop the plan which will outline what, when, who, why where, how much, etc. in a mutually acceptable manner to create the foundation of their sampling agreement 14

Getting Started- Sampling Webinar • December 2013 – provided a MFRPS national webinar on the topic of Sampling Agreements • Attendees were State MFRPS and ISO lab grant enrollees, FDA and related association participants 15

ISO-MFRPS Sampling Protocol Guidance 16

Acceptable Samples • Manufactured Foods under the purview of FDA • Manufactured Foods under the purview of State Inspection, if also under FDA jurisdiction • Environmental Samples • Routine Manufactured Food samples • Seafood samples • Processed Shellfish • Bottled Water only (not municipal water) • Samples collected in FBI Outbreaks and on Complaints • *Produce, if it is washed and processed 17

Unacceptable Samples • • Grade A Dairy products (Milk) Food produced in a retail setting Environmental Samples from retail settings Municipal Water USDA regulated products Foods intended for animals Raw shellfish 18

Types of Acceptable Analysis • Include, but not limited to: – – – – – Chemical Pesticides Biological Physical (foreign matter)-filth Allergens Metals Toxins Contaminants Specie Substitution/confirmation Confirmation of product identity - example (Honey). 19

Sampling Agreement Example 20

Sampling Resources 21

Updates • • • April 1, 2014: Sampling Agreements due All 30 were submitted Review Feedback Commonalities 22

Next Steps • Laboratory and Manufactured food programs to finalize their Implementation Strategy by Sept. 1, 2014 • Agreements cover 1 year and are reviewed & revised in the following year • Practice should be sustained when funding is no longer available • Future funding 23

Additional Resources • Office of Regulatory Science assistance Ruiquing Pamboukian, FDA ORS Ruiquing. pamboukian@fda. hhs. gov 301 -7968157 • Food. SHIELD, MFRPS Workgroup Contact your State’s SIS representative • AFDO Portal 24

AFDO Portal • Interested in becoming accredited? http: //www. aphl. org/aphlprograms/food/laborat ory-accrediation/pages/default. aspx • Contact: Dare Akingbade, APHL for access to the Discussion Board dare. akingbade@aphl. org 25

Question and Answers I nailed that presentation!!! 26

Contacts Cooperative Agreement Funding Erin Woodom-Coleman, Contracts Project Officer Erin. Woodom-Coleman@fda. hhs. gov 240 -205 -1606 Food Program SIS Representative Guy Delius Angela Kohls Tressa Madden Michelle Motsinger Priscilla Neves Laboratory Sampling Plan Office of Regulatory Science CFSAN-ORS@fda. hhs. gov 27

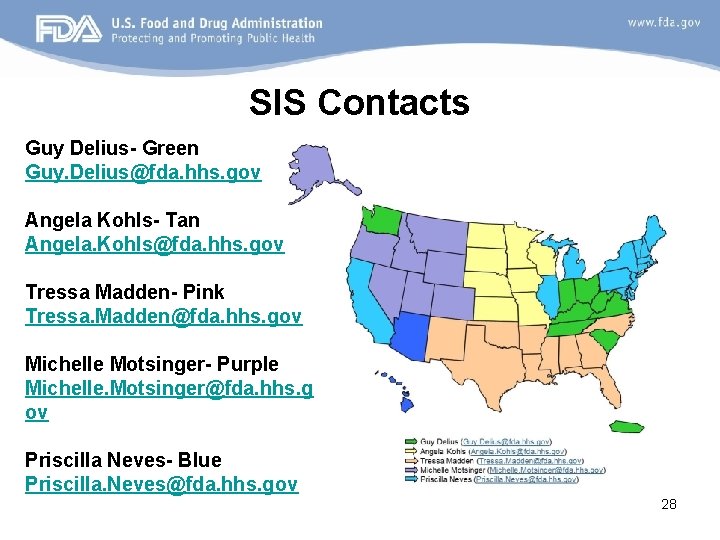
SIS Contacts Guy Delius- Green Guy. Delius@fda. hhs. gov Angela Kohls- Tan Angela. Kohls@fda. hhs. gov Tressa Madden- Pink Tressa. Madden@fda. hhs. gov Michelle Motsinger- Purple Michelle. Motsinger@fda. hhs. g ov Priscilla Neves- Blue Priscilla. Neves@fda. hhs. gov 28