ISPE 2017 Europe Annual conference 3 5 April












- Slides: 19


ISPE 2017 Europe Annual conference 3 -5 April 2017 Barocelona, Spain Sep. 2017

1. ISPE 2017 Europe Annual conference 2. Data Integrity

Track 1 Track 2 Ø Factories of the Future Ø Innovation Ø Serialisation Ø Advanced Aseptic Processing Track 3 Track 4 Ø Clinical and ATMP Supply chain Ø Data integrity and GAMP Ø GDP & GMP Annex 15 Risk Manag… Ø Patient Centricity and innovation supp. . Paperless…

2. Data Integrity

Dr. Nuala Calnan, Pharmaceutical Regulatory Science Team , Dublin Institute of Technology, Ireland Barocelona 5 th April 2017 Brian Ducan Vice President, Engagement Operations, Quality Executive Partners, Inc. Barocelona 5 th April 2017

Chris Clark Director & Principal consultant , Ten consulting Limited, 2017 EU conference, Barocelona 4 -5 April, 2017

Dr. Nuala Calnan, Pharmaceutical Regulatory Science Team , Dublin Institute of Technology, Ireland Barocelona 5 th April 2017

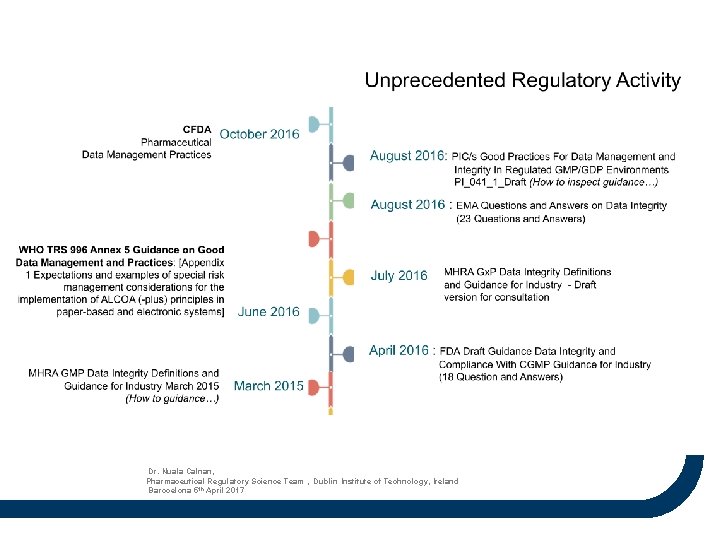
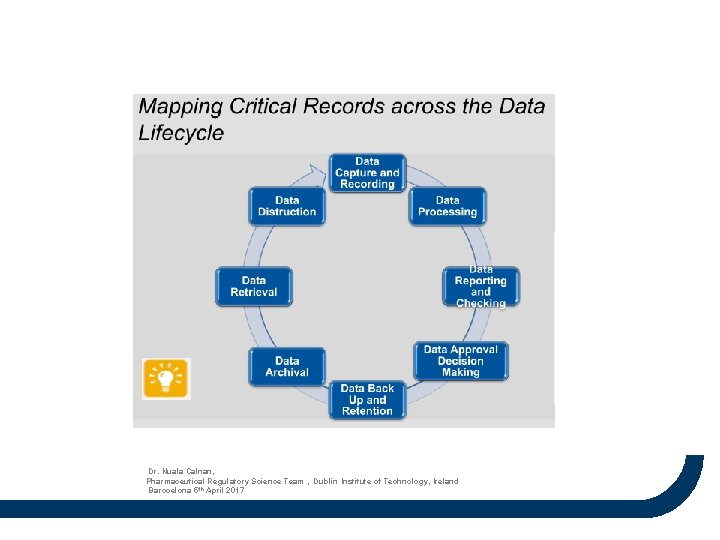
Dr. Nuala Calnan, Pharmaceutical Regulatory Science Team , Dublin Institute of Technology, Ireland Barocelona 5 th April 2017

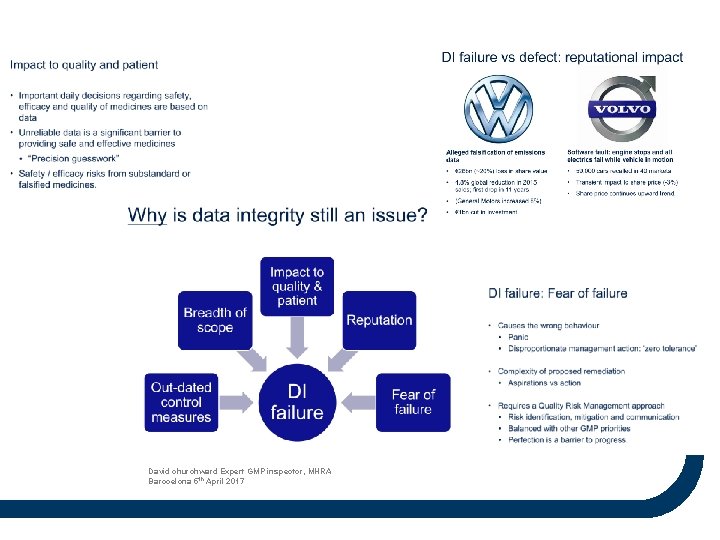
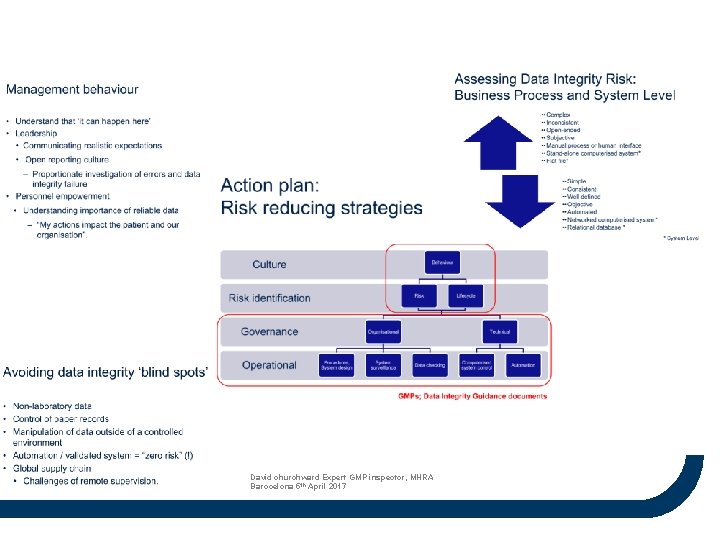
David churchward Expert GMP inspector, MHRA Barocelona 5 th April 2017

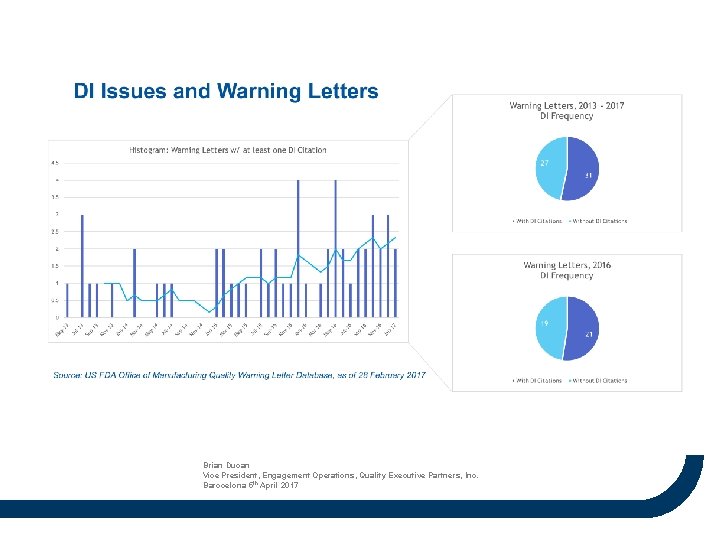
Brian Ducan Vice President, Engagement Operations, Quality Executive Partners, Inc. Barocelona 5 th April 2017

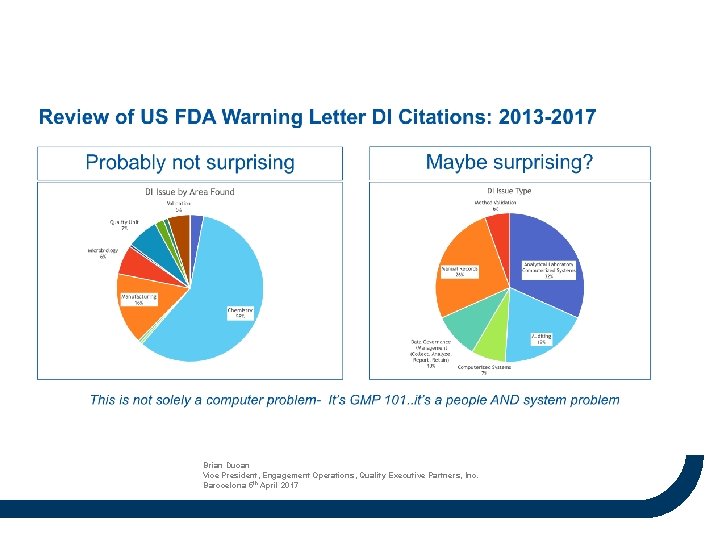
Brian Ducan Vice President, Engagement Operations, Quality Executive Partners, Inc. Barocelona 5 th April 2017

Dr. Nuala Calnan, Pharmaceutical Regulatory Science Team , Dublin Institute of Technology, Ireland Barocelona 5 th April 2017

Brian Ducan Vice President, Engagement Operations, Quality Executive Partners, Inc. Barocelona 5 th April 2017

Dr. Nuala Calnan, Pharmaceutical Regulatory Science Team , Dublin Institute of Technology, Ireland Barocelona 5 th April 2017

Chris Clark Director & Principal consultant, Ten consulting Limited Barocelona 5 th April 2017

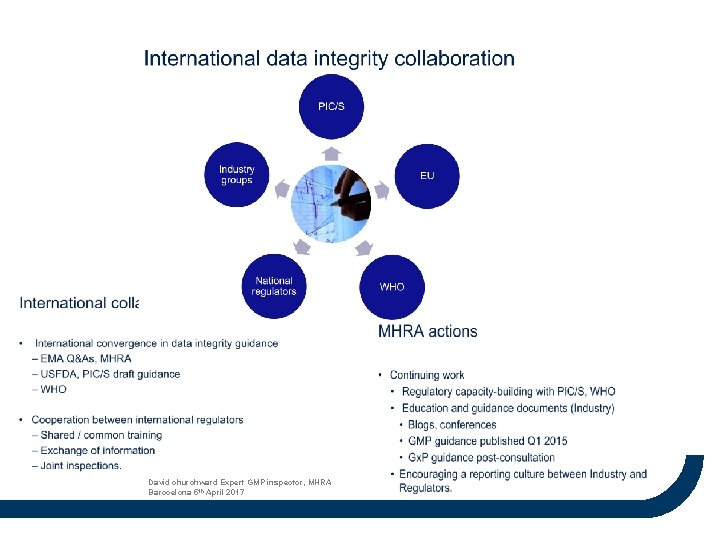
David churchward Expert GMP inspector, MHRA Barocelona 5 th April 2017

David churchward Expert GMP inspector, MHRA Barocelona 5 th April 2017
