Dyeing of Wool Silk Nylon and Acrylic Faiza

















- Slides: 18

Dyeing of Wool, Silk, Nylon and Acrylic Faiza Anwar

Acid Dyes • So called because these are usually applied under acidic conditions. • Dyes are normally very large aromatic molecules consisting of many linked rings. • Acid dyes usually have a carboxyl or amino group on the molecule. • General formula DSO 3 -Na+ i-e sodium salt of sulphonic acid

Acid Dyes • Attached to fiber by ionic or salt linkage at “dye site”. • Dye site in wool is amino group (NH 2); • Applied under acidic conditions, their types are: – Good leveling characteristics – Average leveling characteristics – Poor leveling characteristics

Types of Acid Dyes • Good leveling – They have poor substantivity that’s why they have good levelling property. – applied in 3. 5 -4. 5 ph. • Average leveling – They have average substantivity that’s why they have average levelling property. – applied in 5 -6 p. H, • Poor leveling characteristics • They have very good substantivity that’s why they have poor levelling property. – applied in 6 -7 p. H.


Acid Dyes • Dark shades achievable on wool b/c of its amorphous nature and plenty of amino groups. • Same is the mechanism for silk and synthetics like polyamide • Synthetic fibers may form an ionic or salt linkage. • They are highly substantive dyes so uneven dyeing may result • Evenness achieved by use of retarders such as Na 2 SO 4.

Dyeing with Acid Dyes • Applied under acidic conditions maintained by the addition of – sulphuric acid in dye solution for good leveling dyes – Acetic Acid in dye solution for average and poor leveling dyes • Good substantivity (so chances of unlevelness) • Sodium Sulphate is used as a Retarder for level dyeing.

Addition of Acid • Addition of acid acts as an exhausting agent, because strongly acidic conditions makes more cationic sites available and thus available dye anions got combined with these.

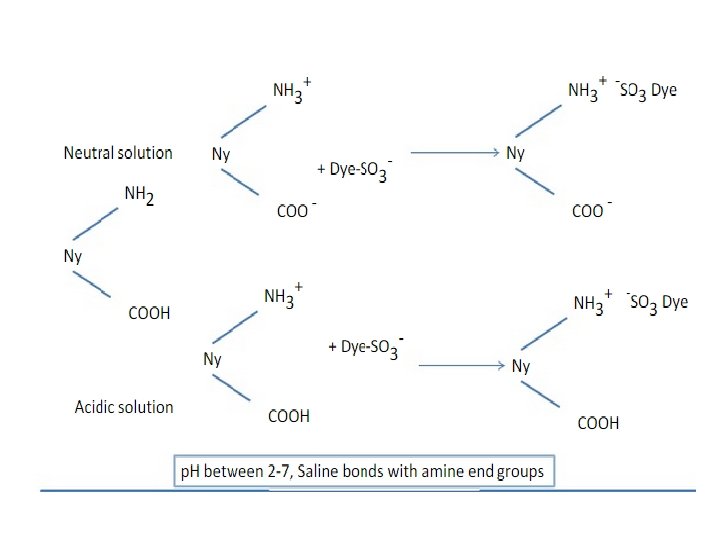
Mechanism of Acid Dyes • Fixation mechanism • Wool-NH 2 + H+ Wool-NH 3 + – Chromophore is -ively charged – Amino group of Wool is made +ively charged so dyes attaches to it by the addition of acid which liberates H+ – DSO 3 - + 3 HN-wool

Addition of Salt • Electrolyte in the acid dye bath act as a retarding agent because of chlorides ions attracted by the positive sites at the fiber. • Anions of the retarders (chloride or sulphate radicals) are negatively charged and smaller than the dye anion and can move more rapidly in the dye liquor. • Dye sites of the fiber polymer are rapidly occupied by the chloride/sulphate radical and in effect compete with the acid dye anion for the dye sites.

Effect of Heat • Dye molecules have the greater affinity for the dye sites but the sulphate radicals retards the rate at which the dye molecules occupies the dye sites results in uniform dyeing. • The application of heat assists the dyeing process by increasing the kinetic energy of the dye molecules which are slowly overcoming the retarding effect of the sulphate radicals. • Thus, the dye anion will gradually replace the sulphate radical that has been attached to the dye sites.

Properties of Acid Dyes • Light fastness is good being rating of about 5. – Since the chromophore of acid dyes are stable and can bear the UV exposure present in sunlight. – • Wash fastness is poor to good depending upon substantivity of the type of dye used. – W. F is 2 -3 with good leveling characterstics – 3 -4 for those whose average leveling – 4 -5 for poor leveling characterstics.

Reasons for Low wash fastness – Firstly, This is because the ionic and hydrogen bonds are hydrolyzed in water. – Secondly these dyes are acidic in nature so can not bear alkaline treatments during washing and laundering.

Basic Dyes • Can be used for wool, silk and acrylic and modacrylic • But poor properties for fibers other than acrylic • Also called cationic dyes b/c they acquire +ive charge in solution • Are MOST BRILLIANT dyes

Dyeing with Basic Dyes • Applied under slight acidic solution normally 6 -7. • Good substantivity (so chances of unlevelness) • Retarder used for level dyeing

Fixation mechanism – Chromophore is +ively charged – Acrylic has –ively charged dyesites normally sulphonate groups so dyes attaches to it – D+ + Ac. SO 3 -+ D

Properties of Basic Dyes • Light fastness is excellent 6 -7 • Wash fastness is very good 4 -5 – Due to high substantivity and hydrophobicity of acrylic • Brightness – Excellent brilliance and color intensity

References • Textile Science, by: E. P. G Gohl