Biopharmacy drug absorption mechanisms of absorption physicochemical factors

Biopharmacy drug absorption; mechanisms of absorption; physicochemical factors related 10 -10 -2018

• The rate of transfer is called Flux (J) which is a net diffusion from side of high concentration side to the low concentration side. • Passive diffusion is the major absorption process for most drugs

According to Ficks first law of diffusion, drug molecules diffuse from region of high drug concentration to region of low drug concentration.
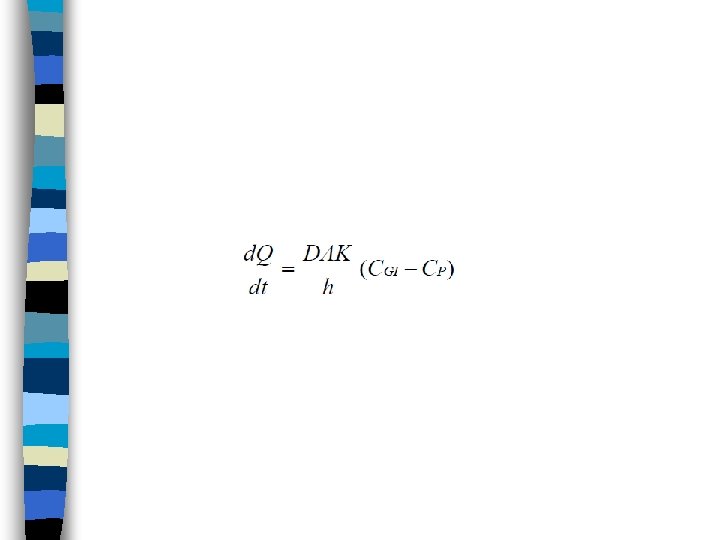


• Because of drug distribute rapidly into large volume after entering the blood, the concentration of the drug in the blood will be quite low with respect to the concentration of the drug at the site of absorption (blood act like a "sink" for drug absorption). • For example drug usually given in milligram doses whereas plasma concentrations are often in micrograms or nanograms per milliliter range.
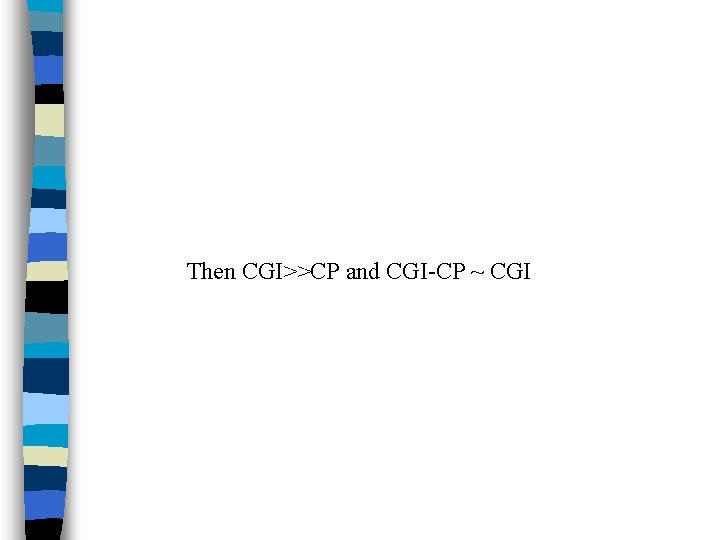
Then CGI>>CP and CGI-CP ~ CGI

• The degree of lipid solubility of the drug influences the rate of drug absorption. • The partition coefficient (K) represents the lipid-water partitioning of the drug across the hypothetical membrane in the mucosa. • Drug that are more lipid soluble have greater value of (K).

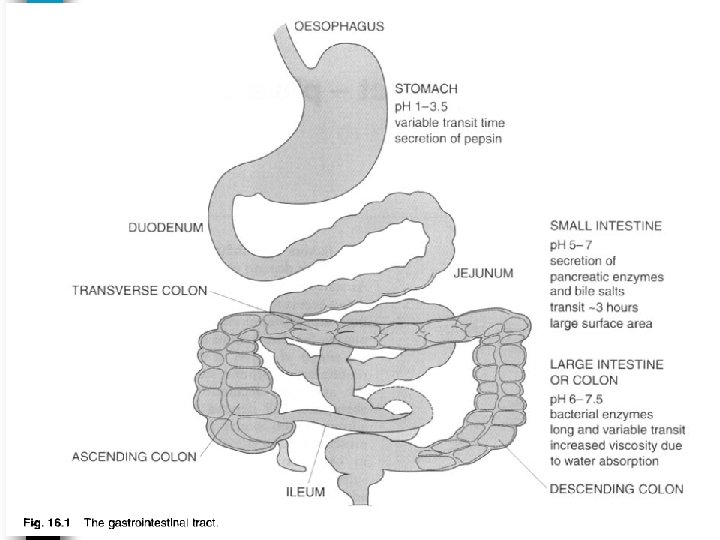
• The surface area (A) of the membrane also influence the rate of absorption. • Drug may be absorbed from most areas of the GIT. • However, the duodenum area of the small intestine shows the most rapid drug absorption, due to such anatomic features as villi and microvilli which provide large surface area and they are less abundant in other area of GIT.

• The thickness of hypothetical model membrane (h) is constant for any absorption site (70 -100 Ao in GIT). • The diffusion coefficient (D) is s constant for each drug and defined as the amount of the drug that diffuses across a membrane of a given unite area per unit time when concentration gradient is unity

• Because (D), (A), (K) and (h) are constant under usual conditions for absorption, • a combined constant (P) or permeability coefficient may be defined:
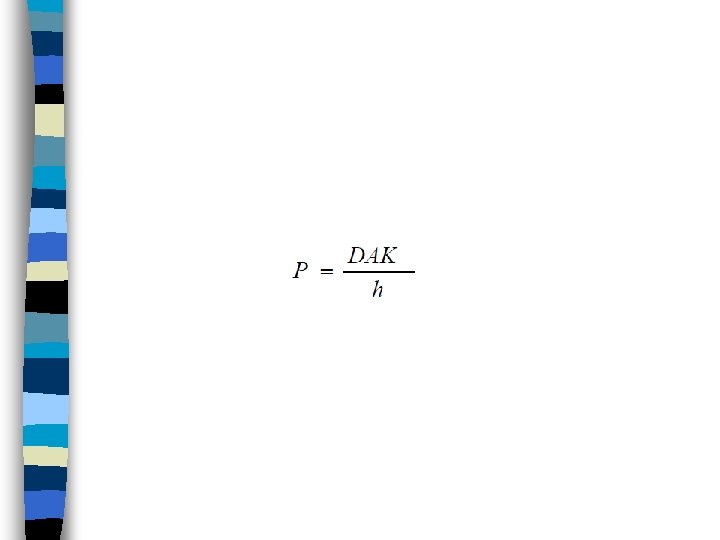

And CP is extremely small compared to CGI , so following relationship for Ficks law is obtained :


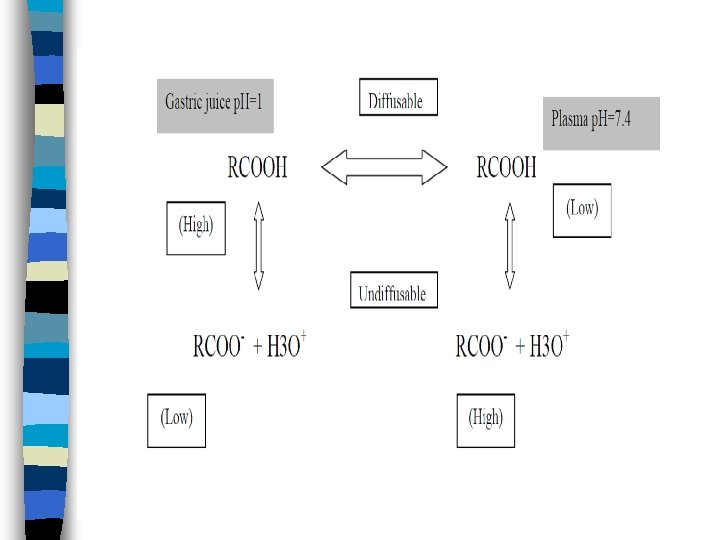
Cell membranes are more permeable to the unionized forms of the drugs than to their ionized forms, mainly because of • the greater lipid solubility of the unionized forms • the highly charged nature of the cell membrane which results in binding or repelling of the ionized drug and thereby decrease cell penetration. • Also ions become hydrated through association with water molecules, resulting in larger molecules than the undissociated molecules and again decreased penetration capability.

Most drugs today are weak organic acids or weak organic bases. Knowledge of their individual ionization or dissociation characteristic is important, because their absorption is governed to a large extent by their degree of ionization as they presented to the membrane barriers. The degree of drugs ionization depends both on the (p. H) of the solution in which it is presented to biological membrane and on the dissociation constant (p. Ka) of the drug. (whether an acid or base).

The concept of p. Ka is derived from Henderson-Hasselbalch equation :

Since the p. H of the body fluids varies (stomach p. H=1, lumen of the intestine p. H=6. 6, plasma p. H=7. 4), the absorption of the drug from various body fluids will differ.


2 - Active Transport:

Thank you
- Slides: 23