EFFECT OF PHYSICOCHEMICAL PROPERTIES OF DRUG ON ABSORPTION

EFFECT OF PHYSICOCHEMICAL PROPERTIES OF DRUG ON ABSORPTION By Dr. A. S. Adebayo 21 February 2021 1

Effect of drug dissolution n n Factors affecting rate of release/dissolution and hence, bioavailability from solid dosage forms: The rate and extent at which the drug in solution reaches the site (s) of absorption in absorbable form The rate and extent of absorption across the gastro-intestinal barrier The extent to which the drug is metabolized during passage through the g. i. t. and/or liver. 21 February 2021 2
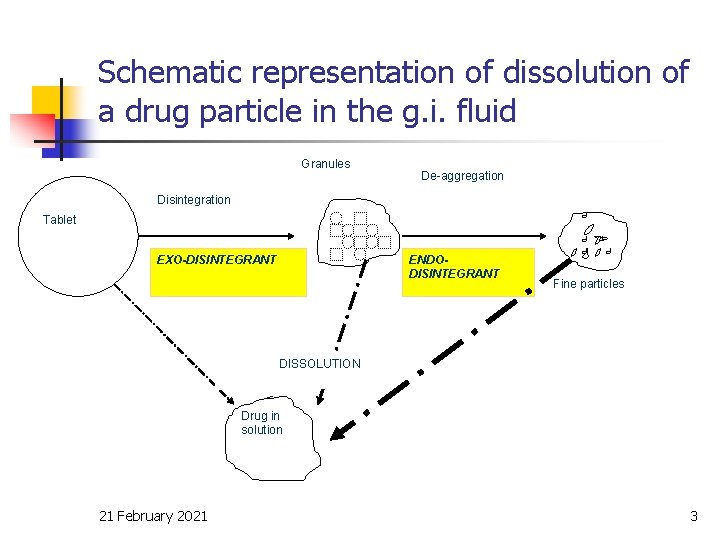
Schematic representation of dissolution of a drug particle in the g. i. fluid Granules De-aggregation Disintegration Tablet ENDODISINTEGRANT EXO-DISINTEGRANT Fine particles DISSOLUTION Drug in solution 21 February 2021 3

Noyes-Whitney equation n n 21 February 2021 where d. C/dt is the rate of dissolution D is the diffusion coefficient of the drug in solution in g. i. fluid S is the effective surface area of drug particle in contact with the g. i. fluid, Cs is the saturation solubility of the drug in the diffusion layer and Ct is the concentration of drug in solution in the bulk medium (g. i. fluid). 4

Factors affecting drug dissolution n Physiological conditions – n The diffusion coefficient, D, of a drug in the g. i. fluid may be decreased by presence of substances which increase the viscosity of the fluids such as food. n The thickness of the diffusion layer, h, will be influenced by the agitation experienced by drug particles due to gastric and/or intestinal motility. 21 February 2021 5

Factors affecting drug dissolution (Cont. ) n The concentration of drug, C, n n n will be influenced by the rate of removal of dissolved drug by absorption through the g. i. /blood barrier and the volume of fluid available for dissolution (fluid intake). A low value of C will increase the concentration gradient and this forms the basis for the dissolution under the so called “sink” condition. 21 February 2021 6

Sink condition n n 21 February 2021 Continuous, unidirectional flow from g. i. t. to blood First order kinetic process. 7

Physico-chemical properties of drug n Particle size Crystal form n Polymorphism –Drugs exhibiting polymorphism n include chloramphenicol palmitate, cortisone acetate, tetracyclines, sulphathiazole and paracetamol. n Armophous form-. Armophous form of novobiocin is effective while its crystalline forms are ineffective. 21 February 2021 8

Physico-chemical properties of drug (Cont) n Solvates and hydrates – n n For instance, the anhydrous form of ampicillin showed greater extent of absorption from hard gelatin capsule or aqueous suspension dosage forms than the less soluble, slower dissolving crystalline form. Salt form– n n For example, sodium salt of tolbutamide gave in vitro dissolution rate significantly greater than the acid form. Other examples are salt forms of penicillin, novobiocin and barbiturates. 21 February 2021 9

Physico-chemical properties of drug (Cont. ) n Ester form – Chloramphenicol, erythromycin & Pivaloyloxymethylester of ampicillin (Pivampicillin). 21 February 2021 10

Pro-drugs n n Rationale: I. A drug may be too water insoluble for i. v. dosage form. n n n Chemical modification may produce significant water solubility for its i. v. formulation II. A drug required to alter some CNS function may be too polar and therefore not well absorbed across the lipoidal blood-brain-barrier. III. Rapid metabolism of a drug at the site of absorption leading to a decrease in systemic bioavailability after oral dosing. 21 February 2021 11

Complex form n n Molecular complex consists of components held together by weak forces such as hydrogen bond Bonding interaction between the two molecules is rapidly reversible, provided the complex is soluble in biological fluids. 21 February 2021 12

Complex form (Cont. ) n n Properties of drug complexes such as solubility, molecular size and lipid-water partition coefficient differ significantly from those of the respective free drugs. Complexation is often a deliberate attempt in dosage form design to increase solubility or stability of the drug e. g. solid-in-solid complex. 21 February 2021 13

Clathrate form n n n Clathrates are formed if a substance is capable of forming channels or cages which can take up another substance into the intra-space of the structure. Clathrate forming substances are gallic acid urea, thiourea, aminos and zeolites. Clathrates are formed by crystallization of an organic solution of clathrate forming substance with the drug. T n he drug normally exists as monomolecular dispersion in the clathrate complex. 21 February 2021 14

Clathrate form (Cont. ) n On exposure to water or dissolution medium, clathrate-forming vehicle dissolves rapidly and exposes the drug molecule to dissolution medium. n n Drugs that have been presented in clathrate forms include Vitamine A, sulphathiazole, chloramphenicol and reserpine. Clathrates are stable in the dry form. 21 February 2021 15

Adsorption n Concurrent administration of drugs and medicinal substances containing solid adsorbents (e. g. antidiarrhoeal mixtures) may result in interference with the absorption of drugs in the git. Drug may be adsorbed onto kaolin, attapulgite or charcoal with consequent decrease in the rate and extent of its absorption. Examples of documented interactions are promazine/charcoal, lincomycin/kaopectate, talc/cyanocobolamin. 21 February 2021 16

Physical-Chemical Factors Affecting Oral Absorption n o o o Objectives: To understand the physical-chemical factors which affect the oral absorption of drug products To understand the p. H-partition theory and Fick’s law as they apply to drug absorption Apply p. H-partition principle to predict drug absorption along git 21 February 2021 17

p. H - partition theory n For weak acid or basic drug, the solubility of the drug and the rate of absorption through the membranes (lining the GI tract) is controlled by: q the p. Ka of the drug q the p. H of the fluid in the GI tract q the p. H of the blood stream 21 February 2021 18

p. H of git & plasma fluid q q q control the process of its transfer across biomembrane. This can be explained by the p. H partition theory of Brodie (1957). The theory is based on the assumption that only unionized drug moiety can cross biomembrane. 21 February 2021 19

Distribution coefficient 21 February 2021 20
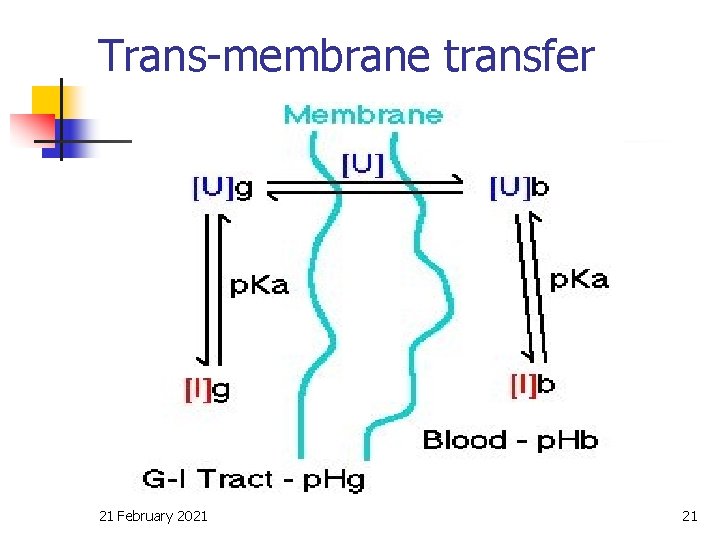
Trans-membrane transfer 21 February 2021 21

p. H-p. Ka Relationship with proportion unionized. For weak acidic drugs: For weak basic drugs: 21 February 2021 22
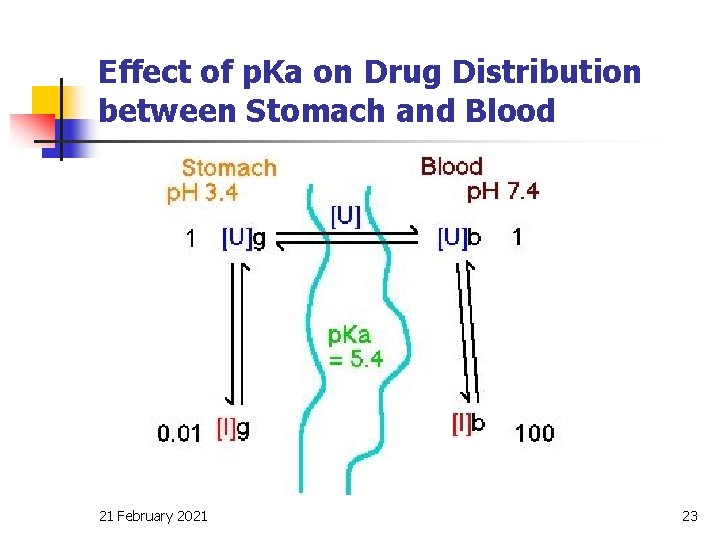
Effect of p. Ka on Drug Distribution between Stomach and Blood 21 February 2021 23

Illustrative example n Compare D for a weak acid (p. Ka = 5. 4) from the stomach (p. H 3. 4) or intestine (p. H 6. 4), with blood p. H = 7. 4 ? ? 21 February 2021 24
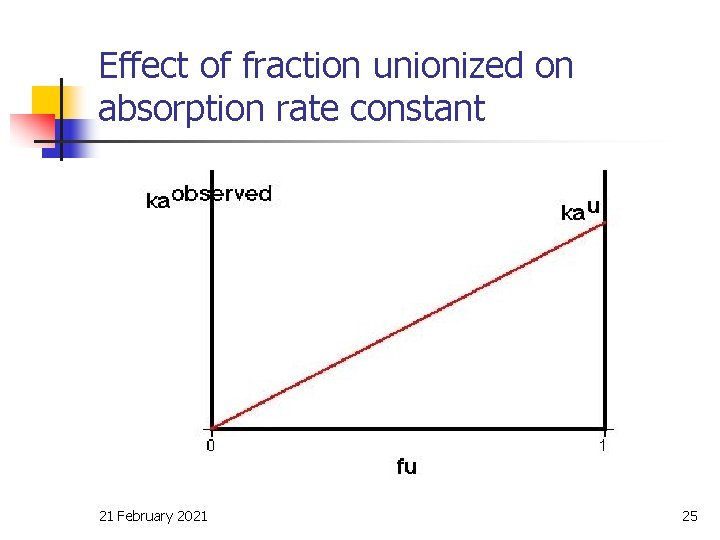
Effect of fraction unionized on absorption rate constant 21 February 2021 25
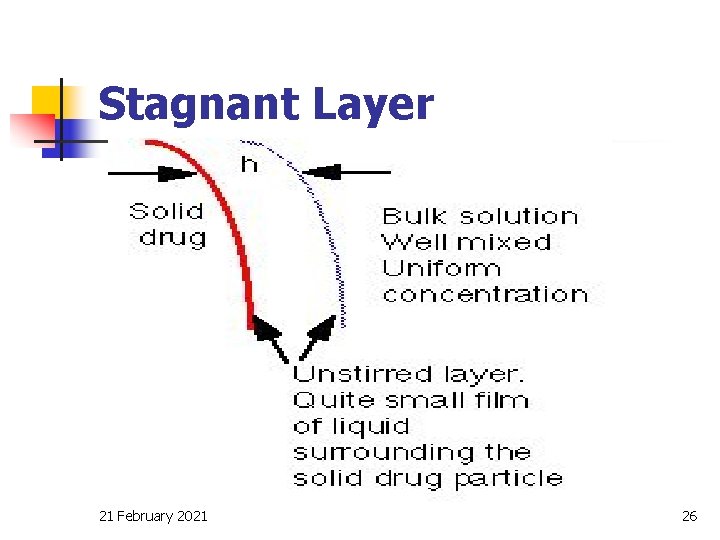
Stagnant Layer 21 February 2021 26

Diffusion gradient/Concentration Gradient 21 February 2021 27

Noyes-Whitney Equation for particle dissolution when Cs » Cb, the equation reduces to: ØDissolution under sink condition is a 1 st order process 21 February 2021 28

Surface area, A n A is the surface area per gram (or per dose) of a n A can be changed by altering the particle size. n n solid drug Generally as A increases the dissolution rate will also increase. Improved bioavailability has been observed with griseofulvin, digoxin, etc. 21 February 2021 29

Diffusion layer thickness, h n n This thickness is determined by the agitation in the bulk solution. In vivo we usually have very little control over this parameter, however factors affecting g. i. t motility/transit time can be important. n Affected by agitation rate which must be controlled when we perform in vitro dissolution studies 21 February 2021 30
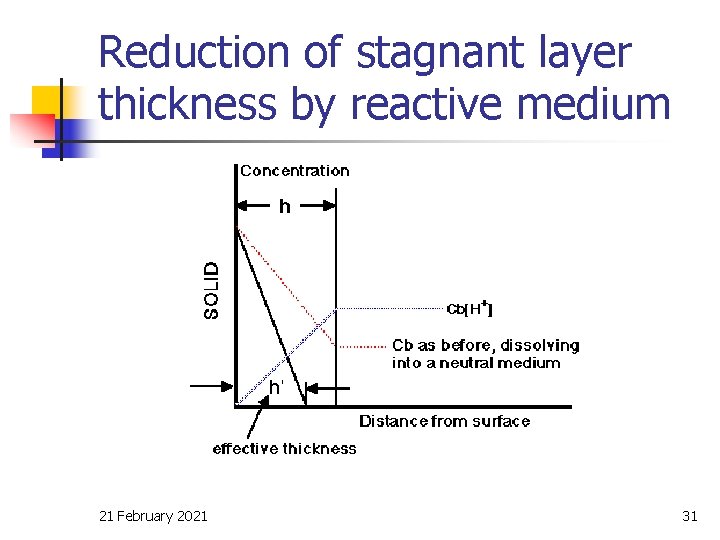
Reduction of stagnant layer thickness by reactive medium 21 February 2021 31

Diffusion coefficient, D n The value of D depends on the: n size of the molecule n viscosity of the dissolution medium 21 February 2021 32

Drug solubility, Cs n n n Dissolution rate increases with Cs Salts of weak acids and weak bases generally have much higher aqueous solubility than the free acid or base If the drug can be given as a salt the solubility and dissolution rate can be increased (e. g. Penicillin V). 21 February 2021 33
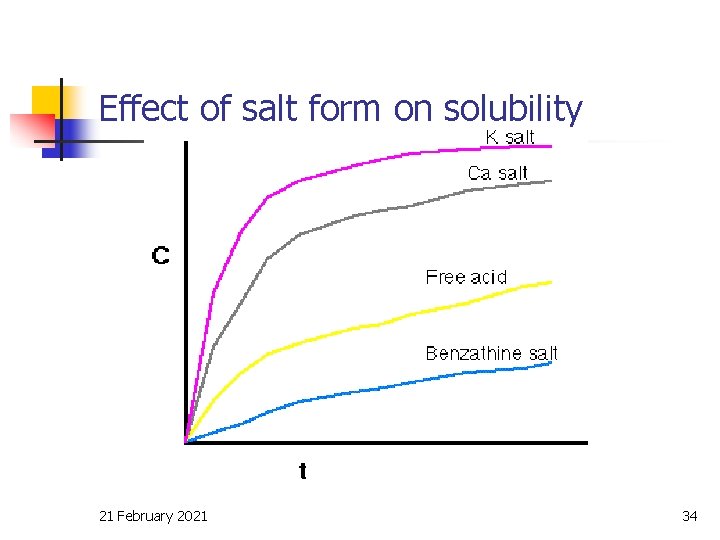
Effect of salt form on solubility 21 February 2021 34
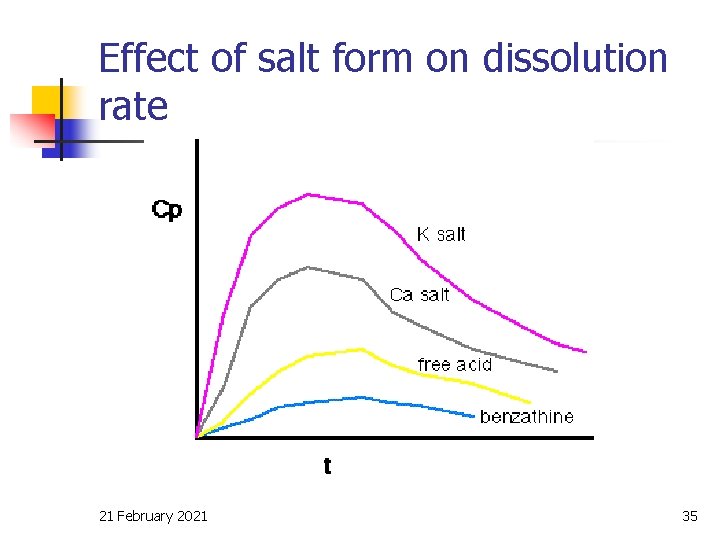
Effect of salt form on dissolution rate 21 February 2021 35
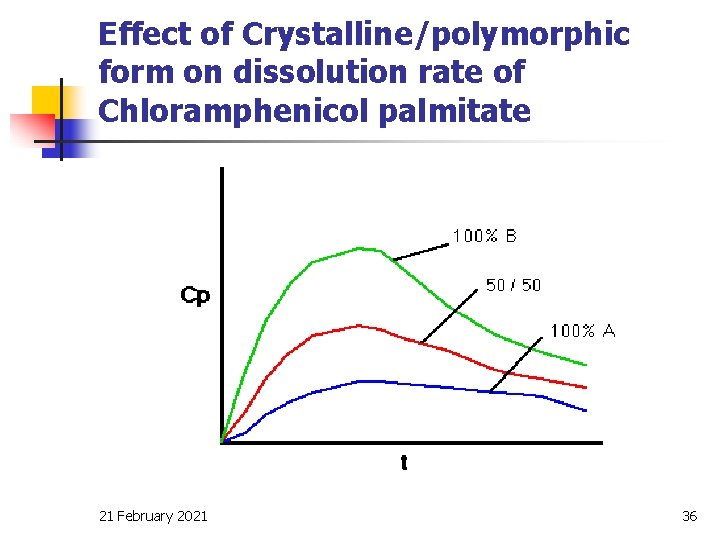
Effect of Crystalline/polymorphic form on dissolution rate of Chloramphenicol palmitate 21 February 2021 36

***END OF PRESENTATION*** QUESTIONS/DISCUSSION 21 February 2021 37
- Slides: 37