Aim What factors affect rate of reactions DO








- Slides: 14

Aim: What factors affect rate of reactions? DO NOW: 1. What does the word “rate” mean? 2. What would affect the rate of reaction? What would speed up a reaction?

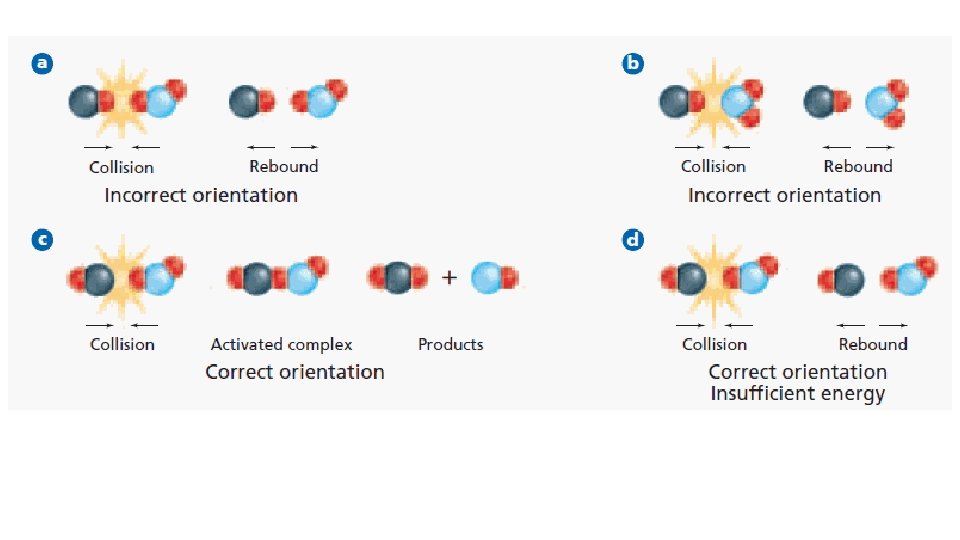
Terminology • Rate of reaction- how fast, how many moles are produced or consumed in a unit of time • Collision Theory- rate of reaction depends on how often collisions occur and how many of them are effective • Activation Energy- smallest amount of energy needed to start a reaction

Collision Theory For a reaction to happen, the particles must have effective collision; particles must have enough energy and collide at the correct angles. The collision theory explains the factors that affect the rate of reactions.



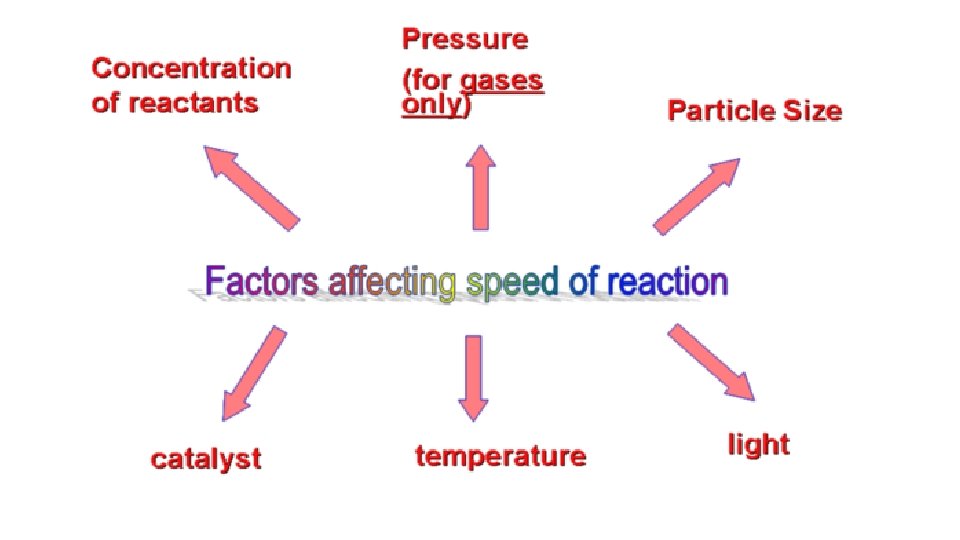
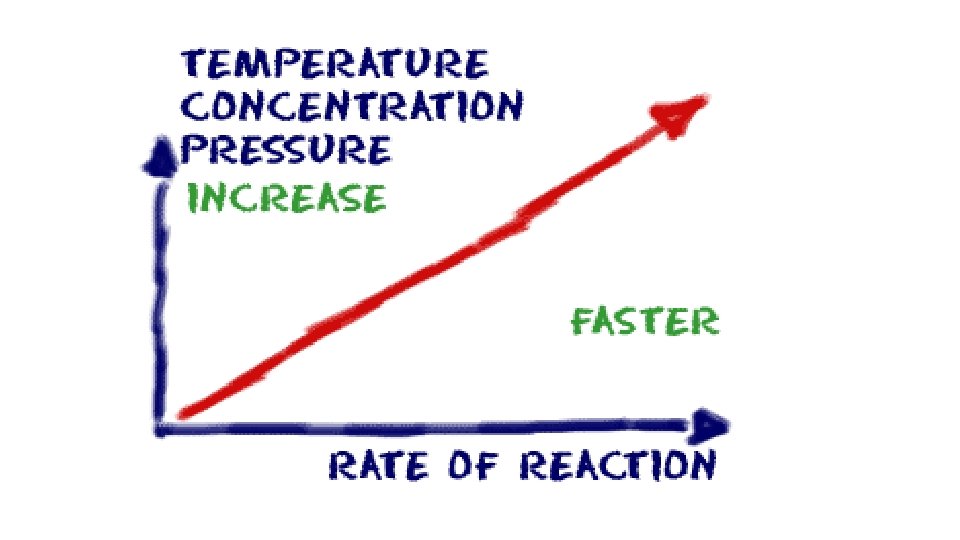
Factors that Affect Rate of Reaction 1. Concentration Increase concentration more collisions For gases increase in pressure increases concentration increases rate of reaction

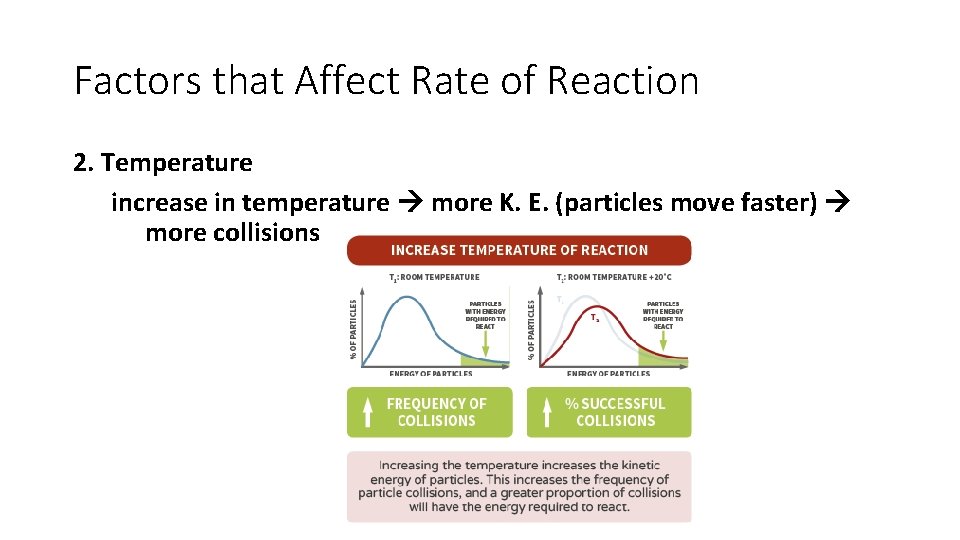
Factors that Affect Rate of Reaction 2. Temperature increase in temperature more K. E. (particles move faster) more collisions

Factors that Affect Rate of Reaction 3. Surface Area increases surface area more exposed particles more collisions

Factors that Affect Rate of Reaction 4. Nature of Reactants Ionic substances react faster than molecules; involve less rearrangement during a reaction.

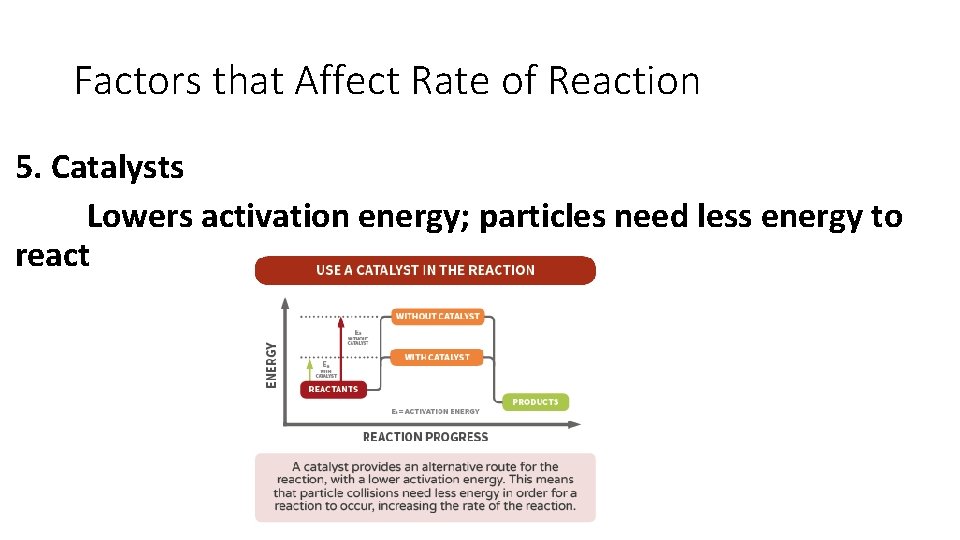
Factors that Affect Rate of Reaction 5. Catalysts Lowers activation energy; particles need less energy to react


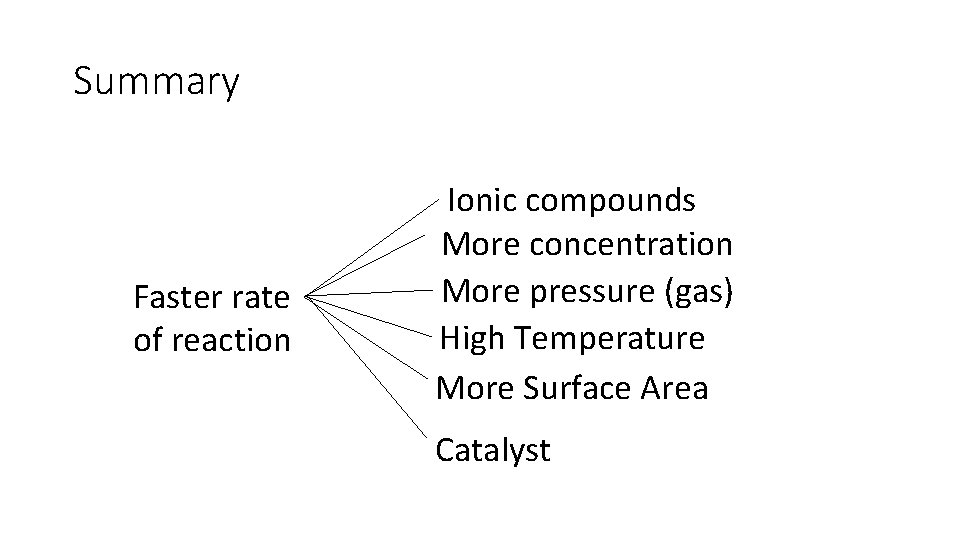
Summary Faster rate of reaction Ionic compounds More concentration More pressure (gas) High Temperature More Surface Area Catalyst

1. Consider the following reaction. Mg(s) + 2 H 2 O (l) --> Mg(OH)2 + H 2(g) For the reaction to occur at the fastest rate, 1 g of Mg (s) should be added in the form of 1) large chunks 3) a ribbon 2) small chunks 4) a powder 2. Raising the temperature speeds up the rate of chemical reaction by increasing 1) the effectiveness of the collisions only 2) the frequency of the collisions only 3) both the effectiveness and frequency of the collisions 4) neither the effectiveness nor frequency of the collisions

3. Consider the following equation. Fe(s) + Cu. SO 4(aq) --> Cu(s) + Fe. SO 4(aq) The Fe reacts more rapidly when it is powdered because the increased surface due to powdering permits 1) increased reactant contact 2) decreased reactant contact 3) pressure to affect reaction rate 4) warmer solution to be used 4. If the pressure on gaseous reactants is increased the rate of the reaction is increased because there is an increase in the 1) temperature 2) volume 3) concentration 4) heat of reaction