Compound composed of two different nonmetallic elements STEPS













- Slides: 15

- Compound composed of two different non-metallic elements.

STEPS IN NAMING BINARY MOLECULAR COVALENT COMPOUND 1. The more electronegative non-metallic element is written or named last. This is usually the element present on the right side of the periodic table.

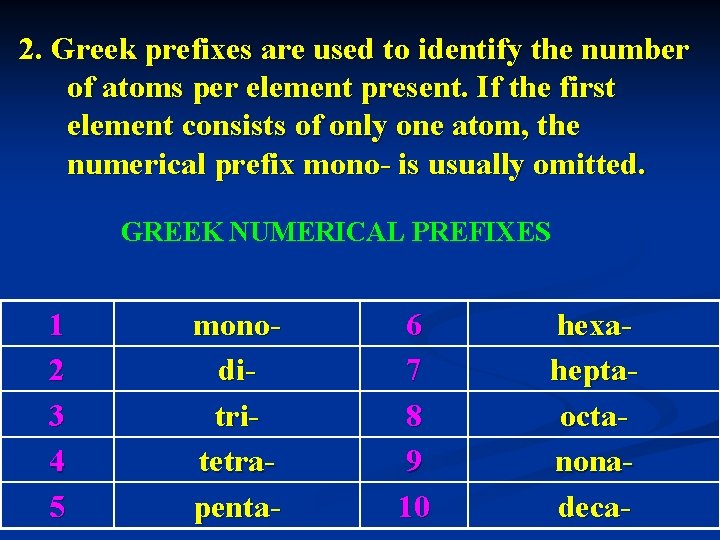
2. Greek prefixes are used to identify the number of atoms per element present. If the first element consists of only one atom, the numerical prefix mono- is usually omitted. GREEK NUMERICAL PREFIXES 1 2 3 4 5 monoditritetrapenta- 6 7 8 9 10 hexaheptaoctanonadeca-

3. For the second element, the stem name is retained and the suffix –ide is added to the it. +6 -2 S O 2 6 S O 3 Sulfur trioxide

CO 2 Carbon dioxide CCl 4 Carbon tetrachloride N 2 O Dinitrogen monoxide N 2 O 4 Dinitrogen tetroxide P 2 O 5 Diphosphorus pentoxide

EXCEPTIONS TO THE RULE H 2 O - water NH 3 - ammonia PH 3 - phosphine CH 4 - methane

- contains polyatomic (more than one type of atom) ions - Naming follows the same step as binary ionic compounds, except that the name of the metal (using Roman numeral if necessary) is named or written first followed by the name of the polyatomic ion

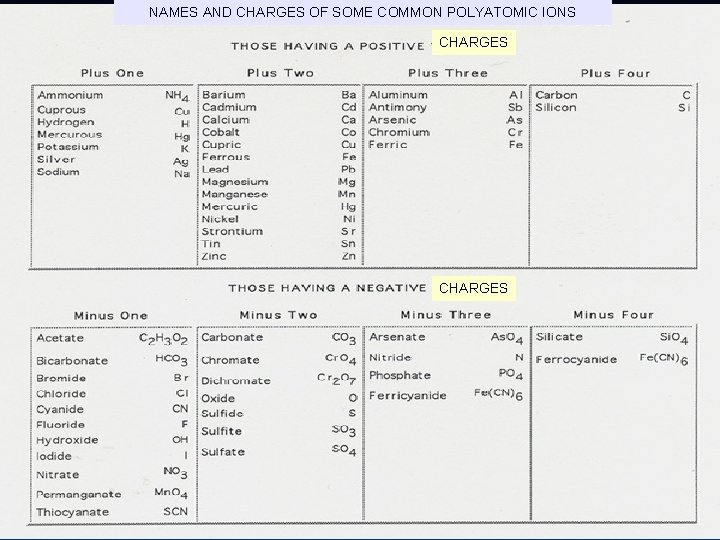
NAMES AND CHARGES OF SOME COMMON POLYATOMIC IONS CHARGES

Ca. CO 3 calcium carbonate KNO 3 potassium nitrate Cu(OH)2 copper (II) hydroxide Cu(OH)3 copper (III) hydroxide Hg. NO 3 mercury (I) nitrate (NH 4)2 SO 4 ammonium sulfate


Non-metallic elements combined with oxygen to form non-metallic oxide. The non-metallic oxide reacts with water to form an acid. NM + O 2 = NMO + H 2 O ACID

TWO TYPES OF ACIDS BINARY ACIDS – compounds of hydrogen plus non-metal. 1. Add hydro- to the name of the NM. 2. Remove –ide and replace with –ic acid. Ex. HF – hydrogen fluoride (binary compound) Hydrofluoric acid HCl – hydrogen chloride Hydrochloric acid

OXYACID – contains hydrogen, oxygen and another element. 1. Polyatomic ion ends in –ate, replace – ate with –ic acid. Ex. H 2 SO 4 – hydrogen sulfate sulfuric acid 2. Polyatomic ion ends in –ite, replace – ite with –ous acid. Ex. H 2 SO 3 – hydrogen sulfite sulfurous acid

Metallic elements combined with oxygen to form metallic oxide. The metallic oxide reacts with water to form a base. M + O 2 = MO MO + H 2 O BASE

BASE – identified by the presence of the hydroxide (–OH) group Write the name of the metal followed by the word hydroxide. Ex. Na. OH – sodium hydroxide Al(OH)3 – aluminum hydroxide Fe(OH)2 – iron (II) hydroxide Fe(OH)3 – iron (III) hydroxide