Metallic and NonMetallic Elements 04 December 2020 Learning

Metallic and Non-Metallic Elements 04 December 2020 Learning question: What is the difference between elements, compounds and mixtures?

Learning objective: • Recall the properties of metallic and nonmetallic elements and locate these elements on the periodic table.

Key words: • Lustre • Conductor • Insulator • Malleable • Ductile • Brittle

Key words – can you find the definition to these words? • Lustre • A material or an object that conducts heat or electricity. • Conductor • Ability to be hammered thin or stretched into wire without breaking. • Insulator • Malleable • Ductile • Brittle • A substance that does not bend, but breaks or shatters. • Able to be hammered or pressed into shape without breaking or cracking. • A material or an object that does not easily allow heat or electricity to pass through it. • When an object is shiny
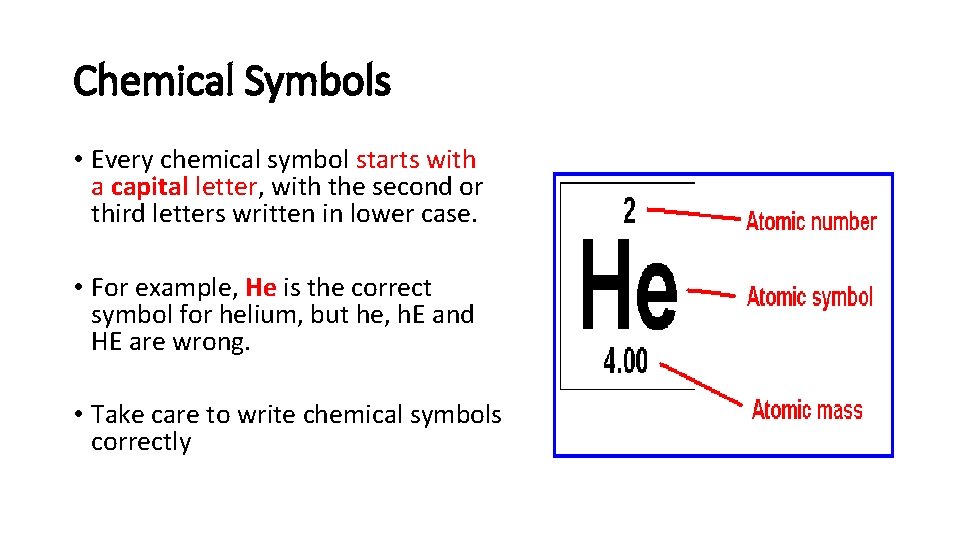
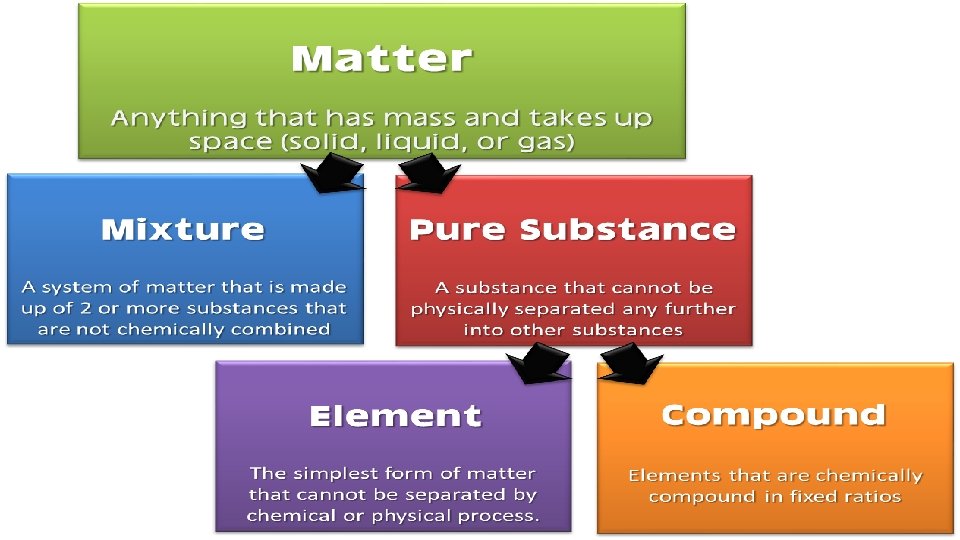
Chemical Symbols • Every chemical symbol starts with a capital letter, with the second or third letters written in lower case. • For example, He is the correct symbol for helium, but he, h. E and HE are wrong. • Take care to write chemical symbols correctly

Atomic Number • Elements are arranged in order of atomic number. • The atomic number identifies the element. • E. g. Helium always has the atomic number 2.
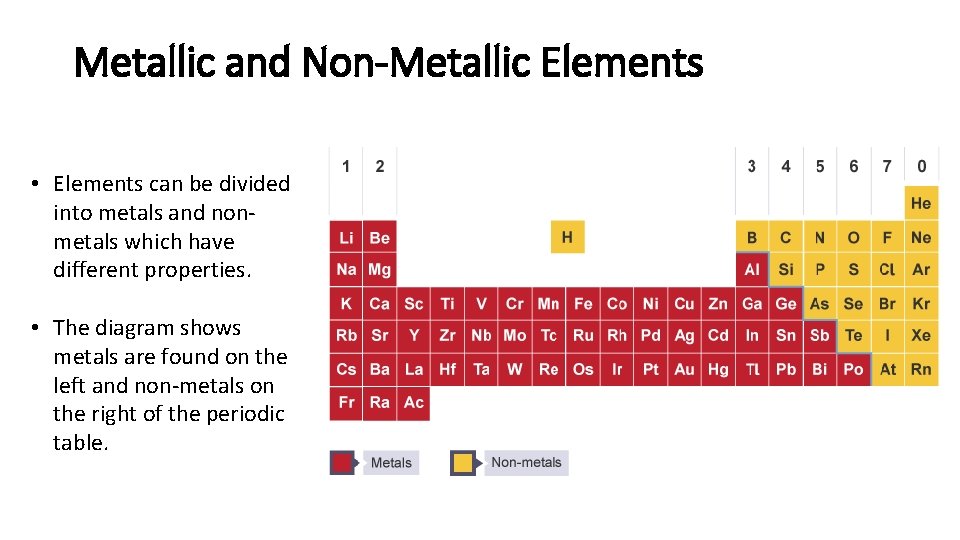
Metallic and Non-Metallic Elements • Elements can be divided into metals and nonmetals which have different properties. • The diagram shows metals are found on the left and non-metals on the right of the periodic table.

Your task • Collect a table from your teacher (or download it) and complete it using the different elements around the room.

Summary of metallic and non-metallic elements

metals are. . . non-metals are. . . • SHINY • usually DULL • MALLEABLE & DUCTILE • usually BRITTLE • GOOD conductors electricity • POOR conductors electricity in the SOLID STATE • usually have HIGH melting points • usually have LOW melting points • form OXIDES known as BASES • form OXIDES that are ACIDIC • Reactivity INCREASES down a group • Reactivity DECREASES down a group
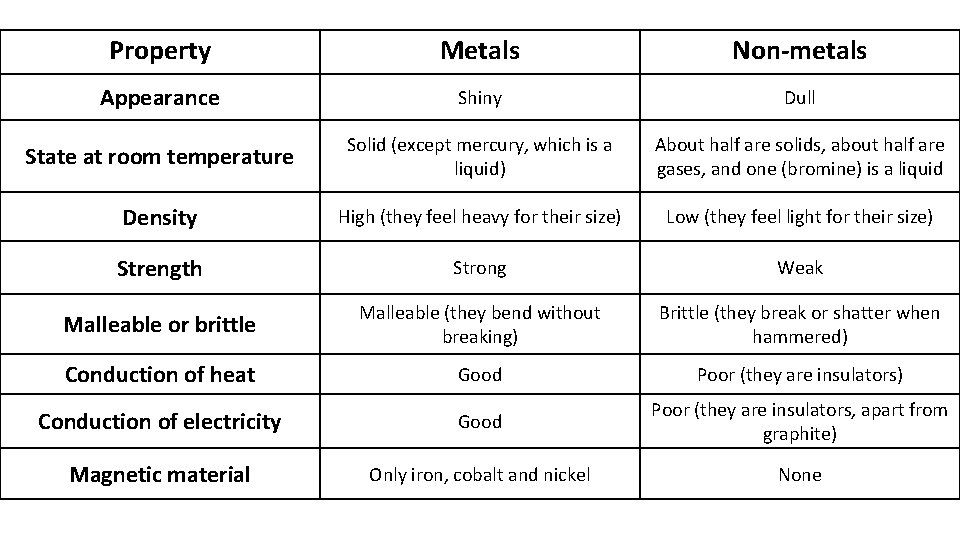
Property Metals Non-metals Appearance Shiny Dull State at room temperature Solid (except mercury, which is a liquid) About half are solids, about half are gases, and one (bromine) is a liquid Density High (they feel heavy for their size) Low (they feel light for their size) Strength Strong Weak Malleable or brittle Malleable (they bend without breaking) Brittle (they break or shatter when hammered) Conduction of heat Good Poor (they are insulators) Conduction of electricity Good Poor (they are insulators, apart from graphite) Magnetic material Only iron, cobalt and nickel None

Examples of Elements Gold is a yellow metal that is found in nuggets. It does not tarnish. Its atomic number is 79. Helium is a colourless, unreactive gas with a low density. Its atomic number is 2. Iodine (atomic number 53) sublimes. Oxygen is a colourless gas found in the air. It is used in respiration. Its atomic number is 8.
- Slides: 13