TEKNIK MESIN FAKULTAS TEKNOLOGI INDUSTRI UNIVERSITAS MERCU BUANA

TEKNIK MESIN FAKULTAS TEKNOLOGI INDUSTRI UNIVERSITAS MERCU BUANA MODUL KE SEMBILAN THERMODINAMIKA DASAR NANANG RUHYAT CARNOT CYCLE The Carnot cycle is a particular thermodynamic cycle, modeled on the Carnot heat engine, studied by Nicolas Léonard Sadi Carnot in the 1820 s and expanded upon by Benoit Paul Émile Clapeyron in the 1830 s and 40 s. Every thermodynamic system exists in a particular state. A thermodynamic cycle occurs when a system is taken through a series of different states, and finally returned to its initial state. In the process of going through this cycle, the system may perform work on its surroundings, thereby acting as PUSAT PENGEMBANGAN BAHAN AJAR-UMB MT. http: //www. mercubuana. ac. id Ir. Nanang Ruhyat THERMODINAMIKA DASAR 1

open systems: exchanging energy (heat and work) and matter with their environment. A boundary allowing matter exchange is called permeable. The ocean would be an example of an open system. It is a fact that, for isolated systems, as time goes by, internal differences in the system tend to even out. Pressures and temperatures tend to equalize, as do density differences. A system in which all these equalizing processes have gone practically to completion, is considered to be in a state of thermodynamic equilibrium. Its thermodynamic properties are, by definition, unchanging in time. Systems in equilibrium are much simpler and easier to understand than systems which are not in equilibrium. Often, when analysing a thermodynamic process, it can be assumed that each intermediate state in the process is at equilibrium. This will also considerably simplify the situation. Thermodynamic processes which develop so slowly as to allow each intermediate step to be an equilibrium state are said to be reversible processes. In open systems, matter may flow in and out of the system boundaries. The first law of thermodynamics for open systems states: the increase in the internal energy of a system is equal to the amount of energy added to the system by matter flowing in and by heating, minus the amount lost by matter flowing out and in the form of work done by the system. The first law for open systems is given by: During steady, continuous operation, an energy balance applied to an open system equates shaft work performed by the system to heat added plus net enthalpy added. where Uin is the average internal energy entering the system and Uout is the average internal energy leaving the system Open systems PUSAT PENGEMBANGAN BAHAN AJAR-UMB MT. http: //www. mercubuana. ac. id Ir. Nanang Ruhyat THERMODINAMIKA DASAR 3
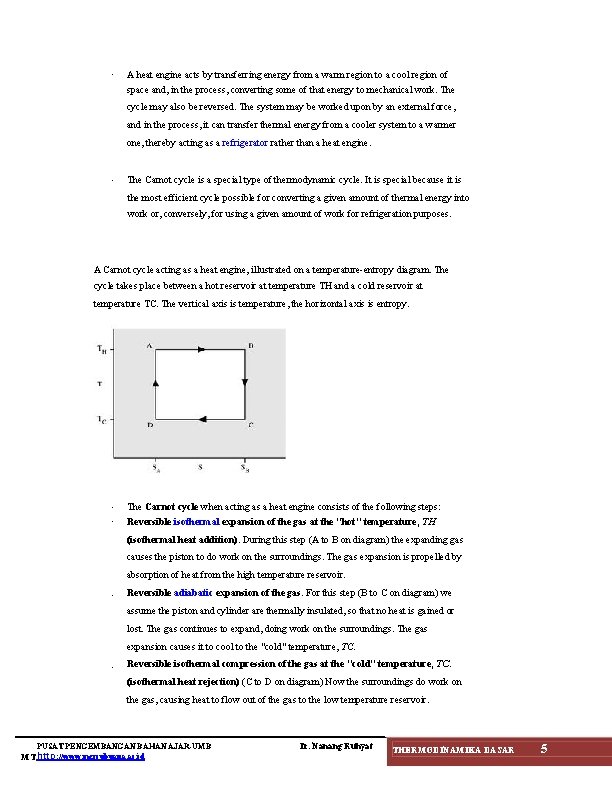
· A heat engine acts by transferring energy from a warm region to a cool region of space and, in the process, converting some of that energy to mechanical work. The cycle may also be reversed. The system may be worked upon by an external force, and in the process, it can transfer thermal energy from a cooler system to a warmer one, thereby acting as a refrigerator rather than a heat engine. · The Carnot cycle is a special type of thermodynamic cycle. It is special because it is the most efficient cycle possible for converting a given amount of thermal energy into work or, conversely, for using a given amount of work for refrigeration purposes. A Carnot cycle acting as a heat engine, illustrated on a temperature-entropy diagram. The cycle takes place between a hot reservoir at temperature TH and a cold reservoir at temperature TC. The vertical axis is temperature, the horizontal axis is entropy. · · The Carnot cycle when acting as a heat engine consists of the following steps: Reversible isothermal expansion of the gas at the "hot" temperature, TH (isothermal heat addition). During this step (A to B on diagram) the expanding gas causes the piston to do work on the surroundings. The gas expansion is propelled by absorption of heat from the high temperature reservoir. · Reversible adiabatic expansion of the gas. For this step (B to C on diagram) we assume the piston and cylinder are thermally insulated, so that no heat is gained or lost. The gas continues to expand, doing work on the surroundings. The gas expansion causes it to cool to the "cold" temperature, TC. · Reversible isothermal compression of the gas at the "cold" temperature, TC. (isothermal heat rejection) (C to D on diagram) Now the surroundings do work on the gas, causing heat to flow out of the gas to the low temperature reservoir. PUSAT PENGEMBANGAN BAHAN AJAR-UMB MT. http: //www. mercubuana. ac. id Ir. Nanang Ruhyat THERMODINAMIKA DASAR 5
- Slides: 3





