Mineral Weathering and Secondary Mineral Formation weathering chemical







- Slides: 17

Mineral Weathering and Secondary Mineral Formation weathering: chemical alteration of minerals (in soils, involves water, gases, acids, etc). Parent material soil Desilication via weathering Parent Material=primary silicates formed from igneous/metamorphic processes Soil= secondary silicates, oxides, carbonates, etc. formed from weathering processes

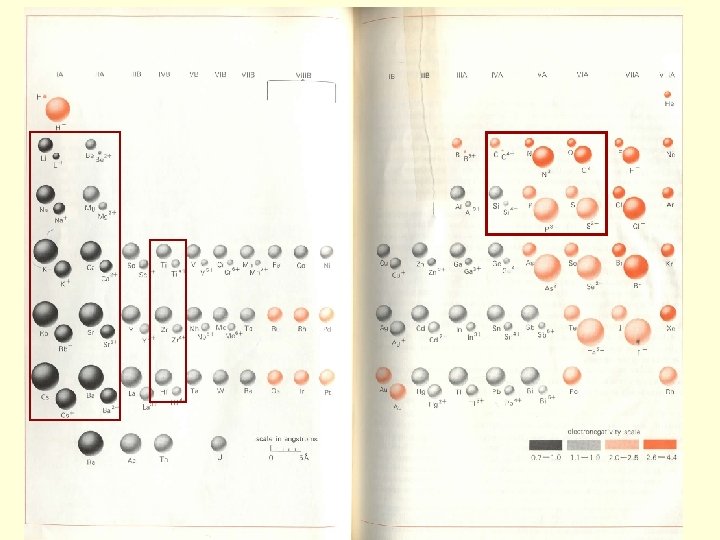
Behavior of Elements During Chemical Weathering • Soils are depleted in elements relative to parent material • Element loss/depletion is determined by elements position on periodic table (which column or group of columns) AND the element’s ionic potential Z/R = ionic potential z=charge, r=radius Classes: Z/R= 0 -3 ion surrounded by H 2 O shell, soluble in H 2 O (Na, Ca, etc) Z/R=3 -~9. 5 ion so strongly attracts H 2 O that insoluble oxides/hydroxides form (Al, Fe) Z/R=>~9. 5 soluble oxyanions form (S, C, etc. )


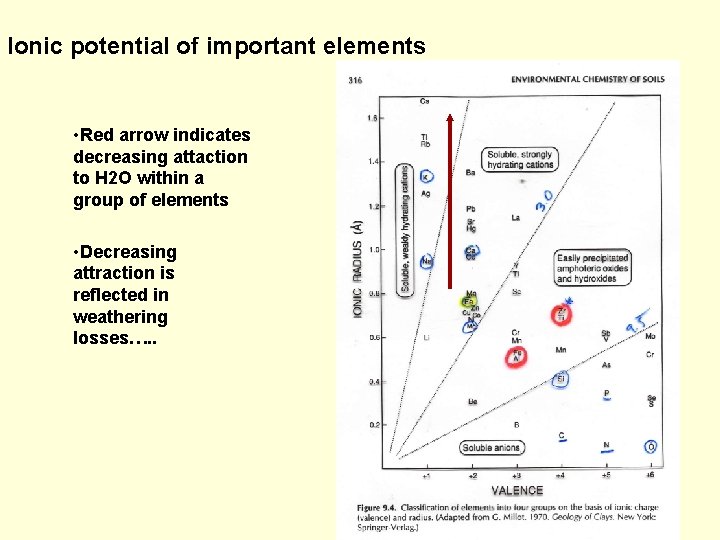
Ionic potential of important elements • Red arrow indicates decreasing attaction to H 2 O within a group of elements • Decreasing attraction is reflected in weathering losses…. .

Element loss varies with ionic potential Ti group Alkali metals and alkaline earths

Mineral Particle Size and Mineralogy Gravel > 2 mm (primary) Sand >= 0. 05 to 2. 0 (primary) Silt <0. 05 to 0. 002 (primary + secondary) Clay < 0. 002 (secondary) Most secondary mineral are silicates, and most secondary silicates are phyllosilicates.



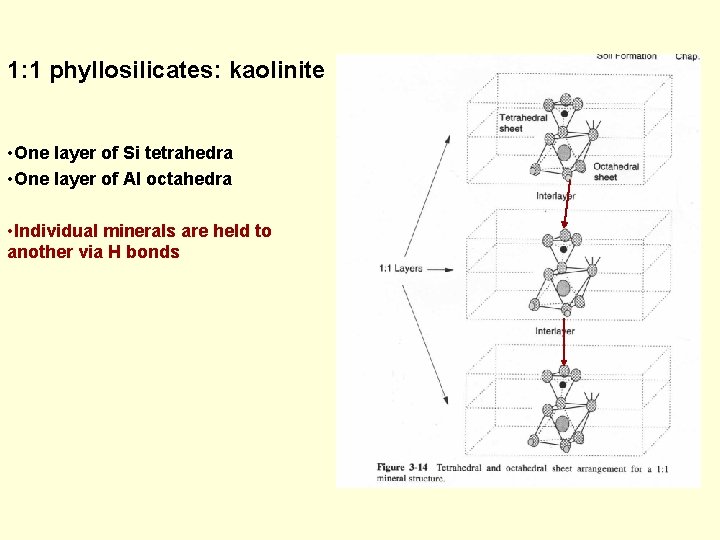
1: 1 phyllosilicates: kaolinite • One layer of Si tetrahedra • One layer of Al octahedra • Individual minerals are held to another via H bonds

2: 1 Phyllosilicates: di and trioctahedral Dioctahedral (smectites) • Substitution of +2 for +3 in octahedral layer (called isomorphous substitution) • Creates a net negative charge (and property of cation exchange capacity) • Results in expandable layers Trioctahedral (vermiculite) • Substitution of +3 for +4 in tetrahedral layer • Also has CEC, but little or no expansion

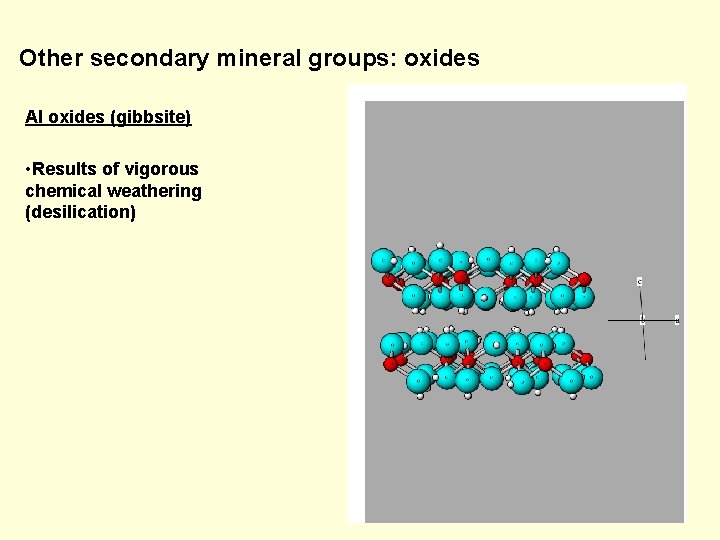
Other secondary mineral groups: oxides Al oxides (gibbsite) • Results of vigorous chemical weathering (desilication)

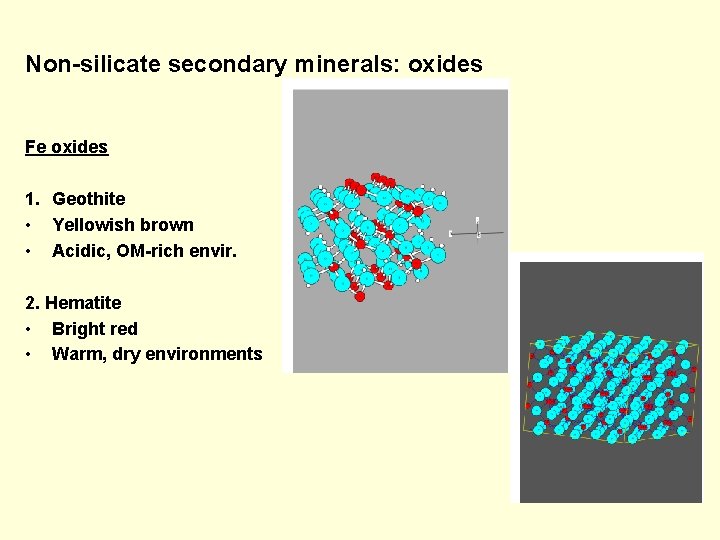
Non-silicate secondary minerals: oxides Fe oxides 1. Geothite • Yellowish brown • Acidic, OM-rich envir. 2. Hematite • Bright red • Warm, dry environments

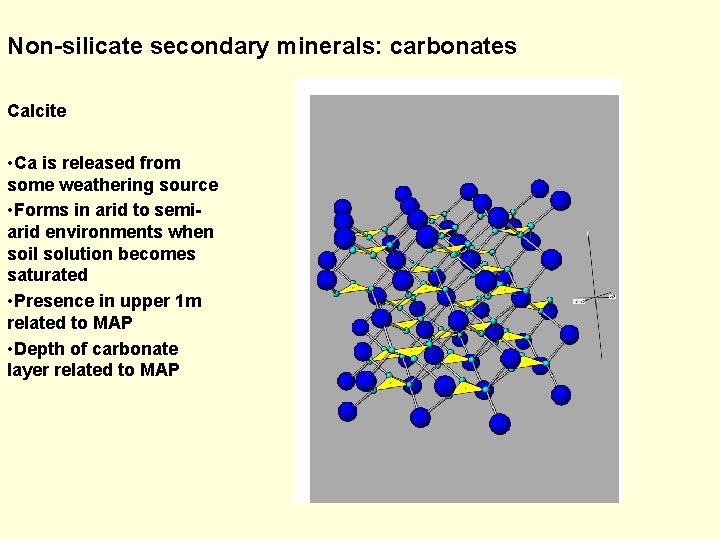
Non-silicate secondary minerals: carbonates Calcite • Ca is released from some weathering source • Forms in arid to semiarid environments when soil solution becomes saturated • Presence in upper 1 m related to MAP • Depth of carbonate layer related to MAP

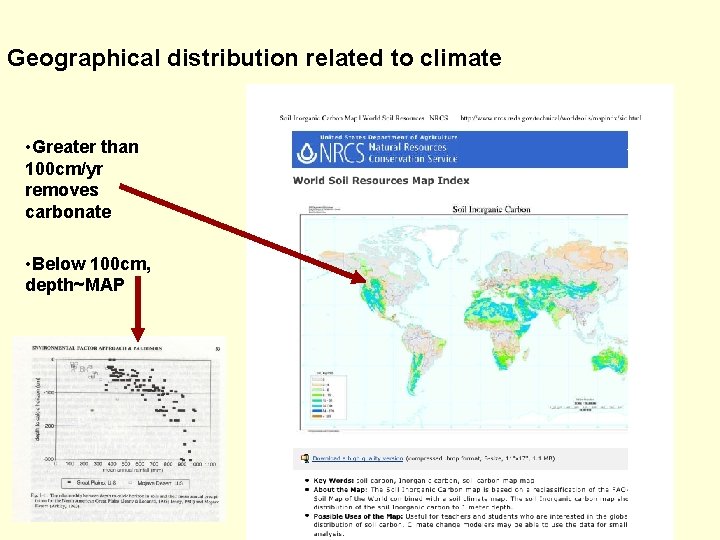
Geographical distribution related to climate • Greater than 100 cm/yr removes carbonate • Below 100 cm, depth~MAP

Non-silicates: sulfates (gypsum) • Presence of sulfates in soils usually occurs in hyperarid climates (or sites with high water table and evaporative enrichment of salts)

Secondary Minerals in California Soils: Sierra Nevada

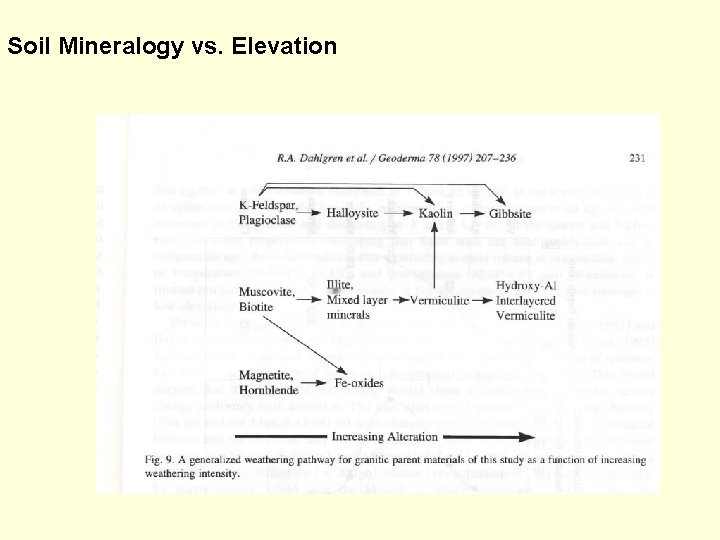
Soil Mineralogy vs. Elevation