es da l Cy ca Sharanya K P





























![• Later archegonial initials [2 -10] appear at the micropylar region of female • Later archegonial initials [2 -10] appear at the micropylar region of female](https://slidetodoc.com/presentation_image_h/1ae80083f067199f430e925a74e4bb5d/image-39.jpg)








- Slides: 54

es da l Cy ca Sharanya K P Department of Botany NSS College Pandalam

• Cycadales include both living (extant) and extinct genera • Originated in the Upper Triassic period of early Mesozoic era abt 20000 yrs ago • Flourished in Jurassic period

Cycads Appeared on Earth 250 MYA • Reached their greatest abundance and diversity during the Jurassic • Declined sharply during the Cretaceous radiation of the angiosperms.

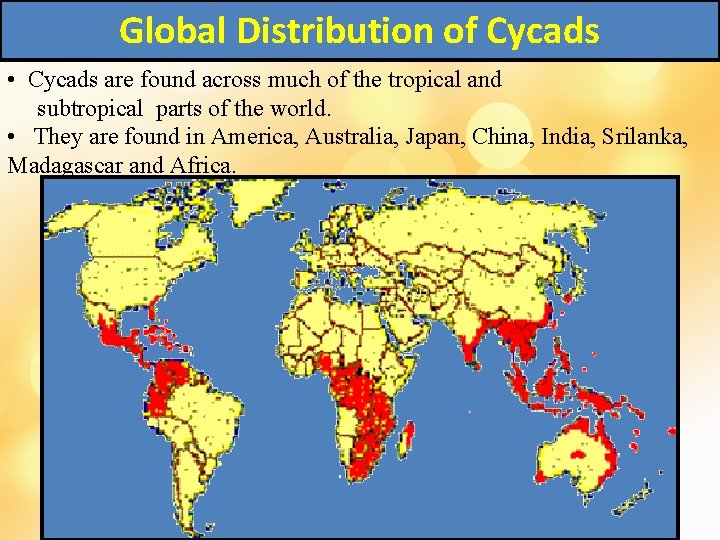
Global Distribution of Cycads • Cycads are found across much of the tropical and subtropical parts of the world. • They are found in America, Australia, Japan, China, India, Srilanka, Madagascar and Africa.

• Members show difft habitats : xerophytes/ halophytes, wet rain forest, sand/rock, O 2 poor swampy bog-like soils rich in org. materials, in full sun/ shade/both. • Fossils include organ bearing generas : Nilssonia – leaves, Bennia – seed, Androstrobus - pollen Classification • Variously classified • Sporn – 1965 – 2 families – Nilssoniaceae (fossils), Cycadaceae (10 living genera) • Bierhorst – 1971 – 3 extant families • Cycadaceae – Cycas 1 ly • Stangeriaceae – Stangeria 1 ly and • Zamiaceae – 8 genera ; Zamia, Dioon, Macrozamia etc

Cycadaceae • 10 extant , 1 extinct (Paleocycas) genera • Living genera include Cycas – 20 sp, Zamia ; 20 -40 sp, Dioon; 3 -5 sp etc • At present there are only 10 genera with 107 -117 sps • Living genera- man’s attention-save fr extinction • Their superficial resemblance- sometimes confused with and mistaken for palms or ferns but -distantly related to either. • Also resemble with Bennettitales, Pentoxylales, Cordaitales etc

Mexican Cycads Dioon Ceratozamia

Australian Cycads Macrozamia

African Cycads Encephalartos Stangeria

Encephalartos in Africa

Only Cycad Native to the US Zamia pumila


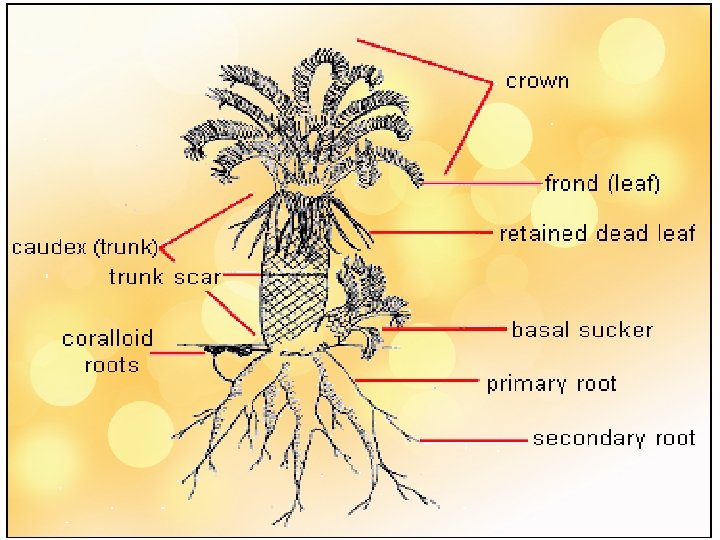
External Features • The basic growth form of a cycad is a central “trunk” with leaves spirally arranged at the top • STEM : thick, slow growing, unbranched/ branched • LEAVES : spirally arranged, foliage leaves, pinnate/ bipinnate, circinate vernation, leaflet – dstinct midrib (Cycas, stangeria), persist for 3 -10 yrs – drop; base persist - persist longer helios. bto. ed. ac. uk/bto/microbes/nitrogen. htm http: //www. damer. com/pictures/travels/southafrica/transvaal 2. jpg

Cycad Leaves • Produced in crowns at tip of stem • Pinnately compound

Cycas revoluta In Cycas, Dioon leaves – dimorphic, within a single phyllotactic series there are zones of green pinnate veg leaves + zone of brown cataphylls (simple, hairy, long, sharply pointed scale leaves)

New flush of leaves


Cycad Stems • Columnar • Little branching • Not very woody

Certain roots in Cycads grow toward the soil surface Corraloid Roots

Corraloid root Cycas

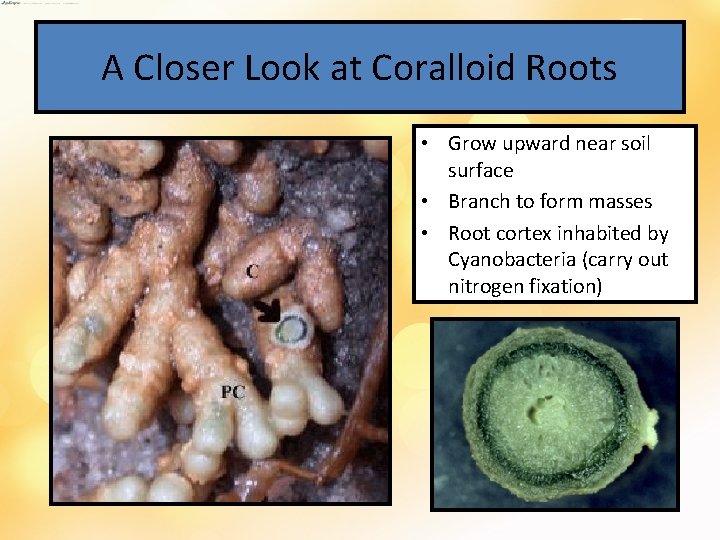
A Closer Look at Coralloid Roots • Grow upward near soil surface • Branch to form masses • Root cortex inhabited by Cyanobacteria (carry out nitrogen fixation)

Coralloid roots in Cycads (a synapomorphy of cycads) • These roots grow up rather than down and can be seen above groun They have symbiotic blue-green algae Cross-section of coralloid root showing layer of blue-green algae (a nitrogen fixer). (Dioon spinulosum)

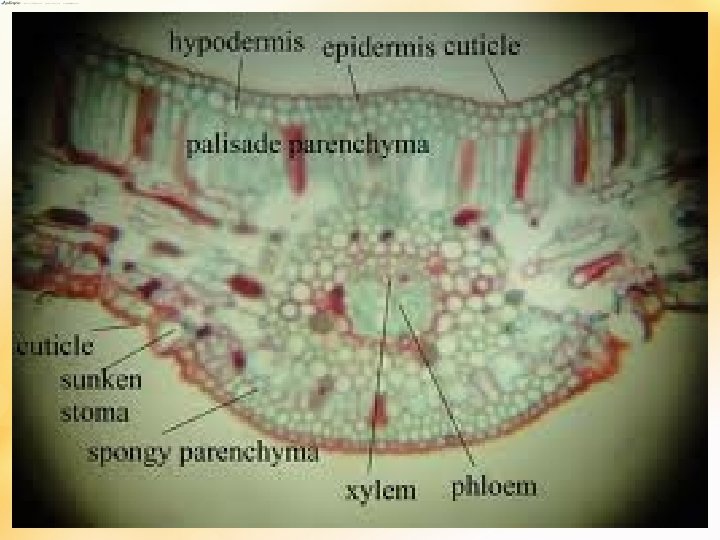
ANATOMY STEM C S • Roughly circular in outline • Massive peripheral cortex and central pith • Both traversed by mucilage canals • VB conjoint, collateral, open, endarch • Girdling Leaf traces are present • 2 o X amount less – compensate by persistent leaf base- manoxylic wood • Monoxylic 2 o growth – single cambial ring ROOT C S • Roots – very thick at base • Polyarch – 8 X and P bundles • 1 o X exarch

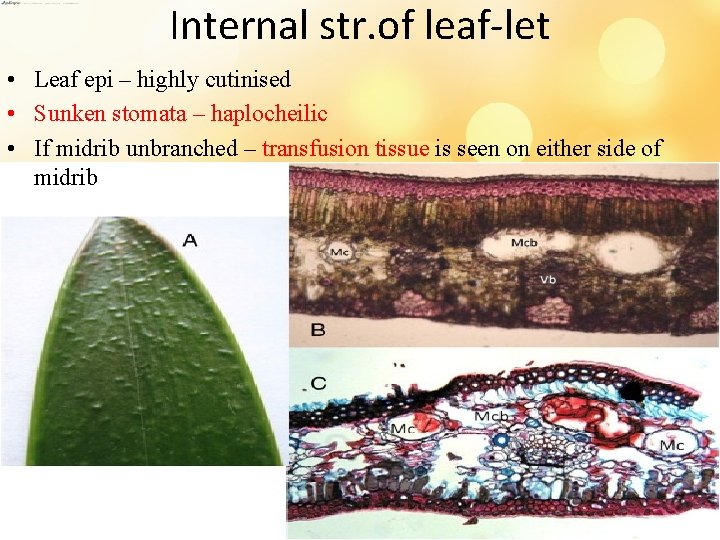
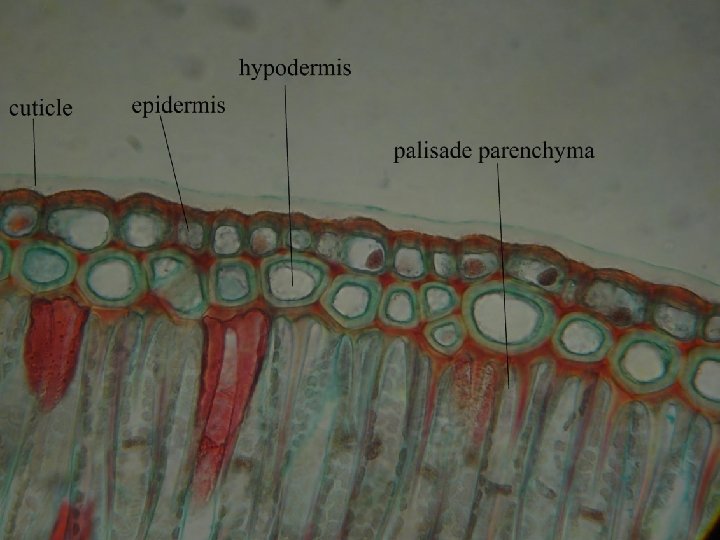
Internal str. of leaf-let • Leaf epi – highly cutinised • Sunken stomata – haplocheilic • If midrib unbranched – transfusion tissue is seen on either side of midrib



Cycads – Reproductive Structures

Cycads produce pollen and seeds in cones • Cones develop at apex of stem

All Cycads are Dioecious (The individual plants are either all male or all female ) • Individual plants produce either pollen cones or seed cones Pollen Cone Cycas revoluta Seed Cone

Female cone and Male cone

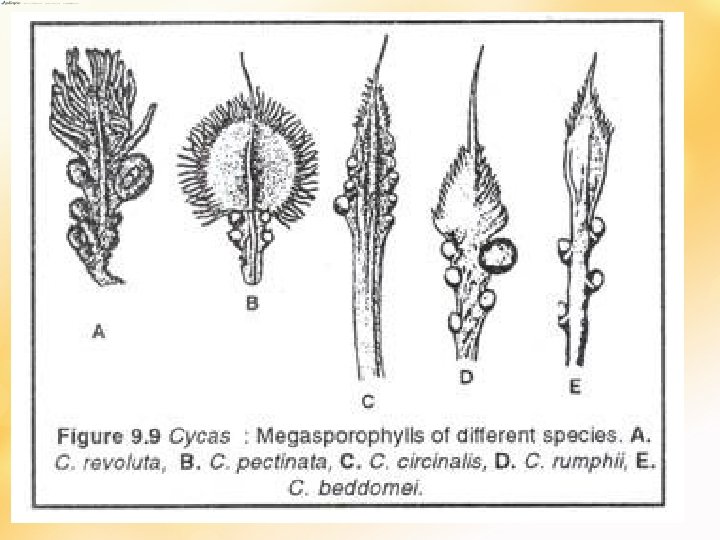
• All living genera except Cycas bear compact female cones (megasporophylls arrange compactly around a central axis) – terminate fem pls • Growth continue - another bud near base of fem cone – wh pushes cone to 1 side – sympodial growth • In Cycas fem pls –spirally arranged megasporophylls- alternate with cataphylls and foliage leaves- loose megasporangiate zone (very primitive) – monopodial growth (apical growth continues) • Usually 1 cone at the apex, sometimes 2 may appear • Cone size varies – Dioon (2 feet), Macrozamia (75 cm long), Z. pygmea (2 cm)- smallest • Megasporophyll (Ms) have distal sterile laminate portion + stalk like portion – bear 2 -3 ovules- micropyles towards cone axis.

The megasporophylls for Cycas is leaf-like with ovules at the base. The enlarged one has been fertilized and is developing into a seed

• • • 2 views Cycas Ms – most primitive – leaf like nature Course of evolution – laminate distal part reduced + no. and size of ovule Stobiliferous cycads Ms – 2 -3 ovules – distal part laminate but not leaf like+ never peltate ( intermediate stage) Final stage – advanced – peltate Ms – Zamia, Macrozamia – 2 ovules Ovules foliar in origin – cycads are phyllospermous Another view – peltate primitive; -open fem organs of Cycas – peltate sporophylls regarded as sporangiophores instead of sporophylls - so ovules are cauline in origin and cycads are stachyospermous

Cycadaceae Megastrobilus / Female cone ♀ § megasporophyll § megasporangia Zamiaceae

Cycas circinalis



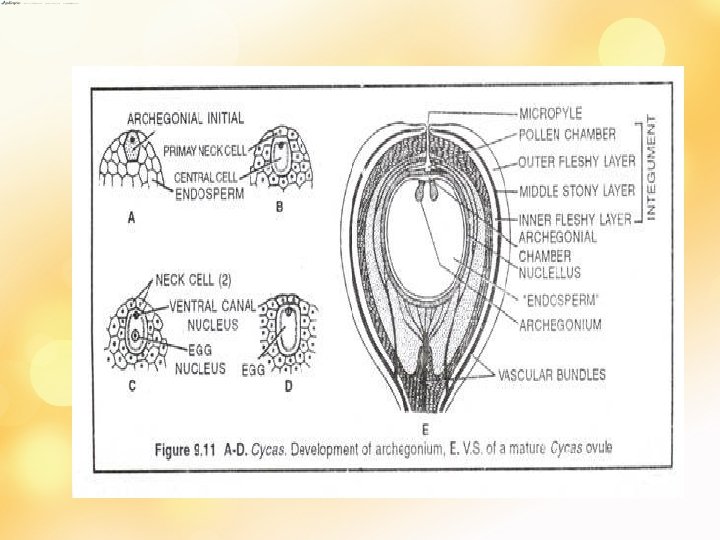
• Ovule – sessile, a nucellus + single integument with distinct micropyle • at maturity – 1/3 rd of nucellus 1 ly free fr Integument at micropylar region • Integument 3 regions – outer and inner fleshy, middle stony • Nucellus – parenchymatous – sub epidermal cell – MMC RD– 4 megaspores – 3 degenerate [micropylar region] - 1 functional – fn megaspore • Fn megaspore enlarge – nuclei divide [1000] – wall formation fr periphery [centipetal]– later cellular mass – wh is the female gmtophyte • As gmtphyte dvlp- nucellus extend apically forming solid nucellar beak – extend into micropyle
![Later archegonial initials 2 10 appear at the micropylar region of female • Later archegonial initials [2 -10] appear at the micropylar region of female](https://slidetodoc.com/presentation_image_h/1ae80083f067199f430e925a74e4bb5d/image-39.jpg)
• Later archegonial initials [2 -10] appear at the micropylar region of female prothallus • All dvlp to archegonia • Each initial –TD- outer 1 o neck cell [prd 2 neck cells by 1 more div] + an inner/ central cell. – enlarges – bfr ferti divides to prd small ventral canal cell + a massive egg cell. • Nucellar cells form a jacket layer • Bfr ferti – nucellar tissue separate pollen chamber nd fem gmtphyte break- disappear – 3 celled spore enter arche. Chamber – VCC degenerate- fertilisation – 2 n oospore – polyembryony [sev. Archegonia fertilised – but 1 get maturity]- others degenerate – seed form –integument ; seed coat


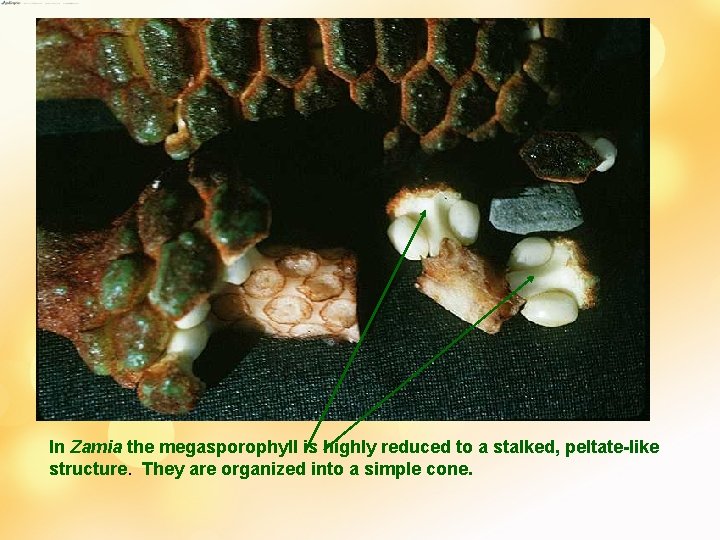
In Zamia the megasporophyll is highly reduced to a stalked, peltate-like structure. They are organized into a simple cone.

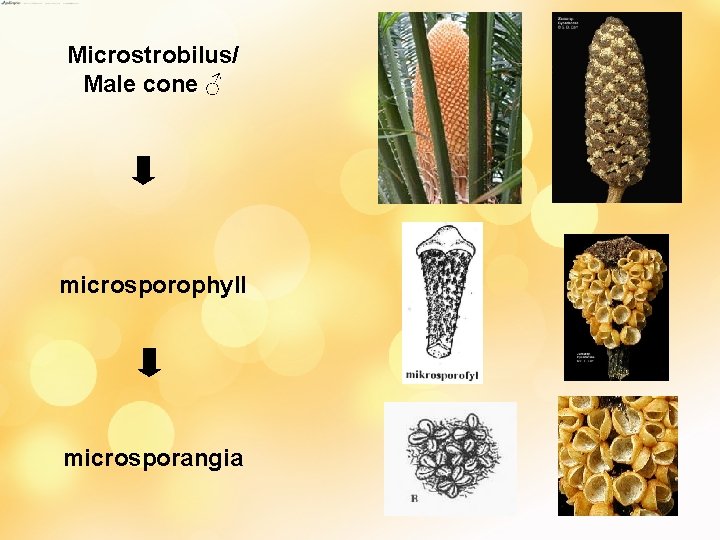
• Male cones are terminal except Macrozamia (arise in axil of leaves) • Compact, central axis bea numerous spirally arranged microsporophylls • Msp – simple conical strr. - distinct fertile and sterile regions (distal end produce to single spinous projection in Cycas, Dioon, Macrozamia), 2 in Ceratozamia, absent in Zamia • Microsporangia on abaxial/ lower surface – appear as mass but in distinct gps of 1/more. These gps are sori • No. varies; 1000 - lesser (50/5/6) • Sporangia dehisce – longitudinal slit • Eusporangiate dvpt

Microstrobilus/ Male cone ♂ microsporophyll microsporangia

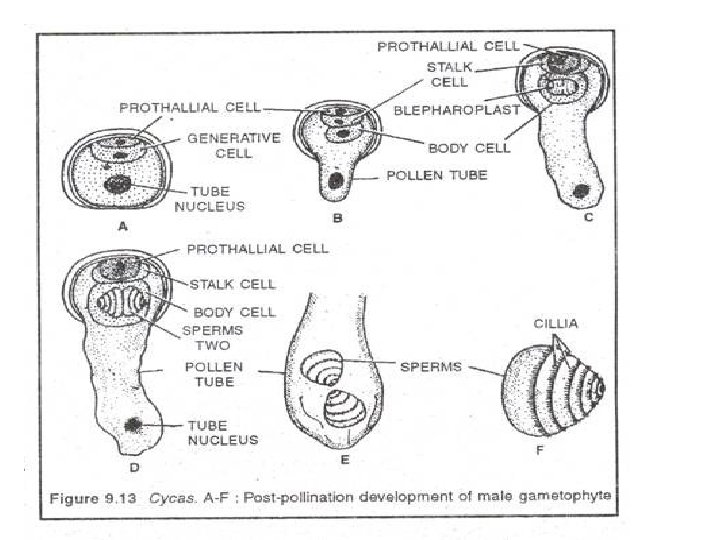
• A microsporangium has massive wall made up of several layers of cells • Hypodermal cell – microsporangial initial – 1 o wall cell (sporangial wall)+ 1 o sporogenous cell (sporogenous tissue – outer single layer – tapetum, rest – MMC – microspores/ pollen grains- arranged in tetrads) • Pollen grain – pioneer of male gametophyte • Each microspore with 2 spore coat – outer exine – thick, inner intine – thin along top and bottom of spore- thicker along sides of microspores, so spore has distinct upper and lower end • Big centrally located nucleus + dense cytoplasm • Microspores germinate within microsporangium [precocious germination] as in heterospory

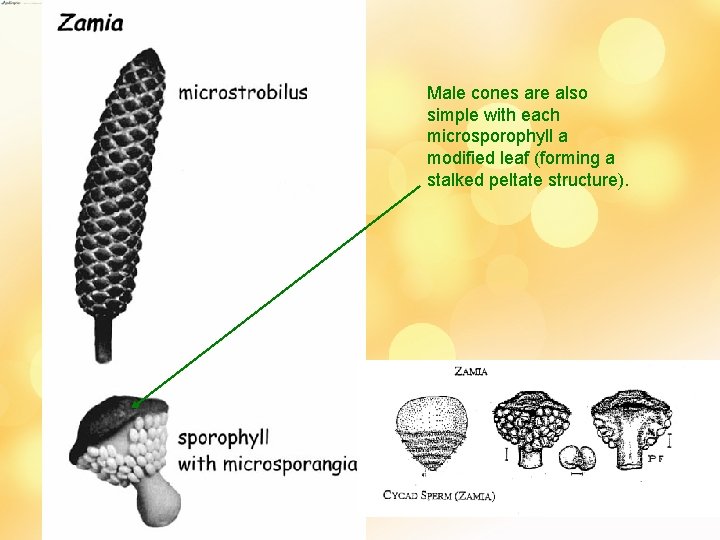
Male cones are also simple with each microsporophyll a modified leaf (forming a stalked peltate structure).


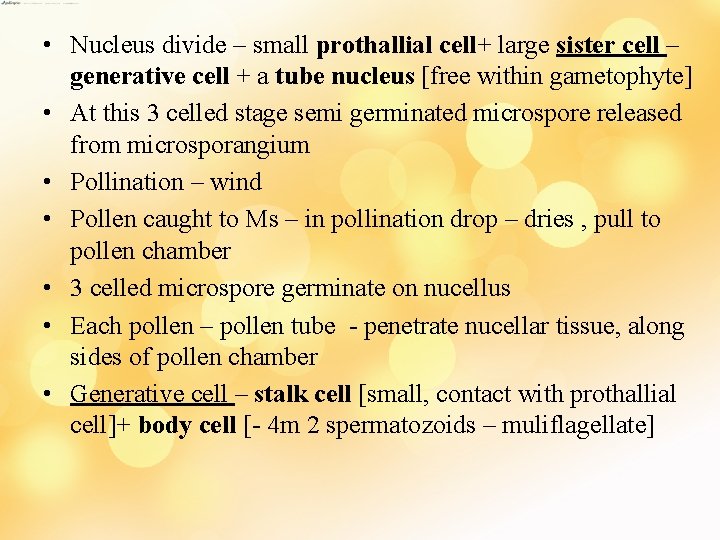
• Nucleus divide – small prothallial cell+ large sister cell – generative cell + a tube nucleus [free within gametophyte] • At this 3 celled stage semi germinated microspore released from microsporangium • Pollination – wind • Pollen caught to Ms – in pollination drop – dries , pull to pollen chamber • 3 celled microspore germinate on nucellus • Each pollen – pollen tube - penetrate nucellar tissue, along sides of pollen chamber • Generative cell – stalk cell [small, contact with prothallial cell]+ body cell [- 4 m 2 spermatozoids – muliflagellate]

Cycadaceae Male cone and Cataphylls (a synapomorphy) of Cycas taitungensis (native to Taiwan) www. pacsoa. org. au/cycads/Cycas/taitungcone. jpg

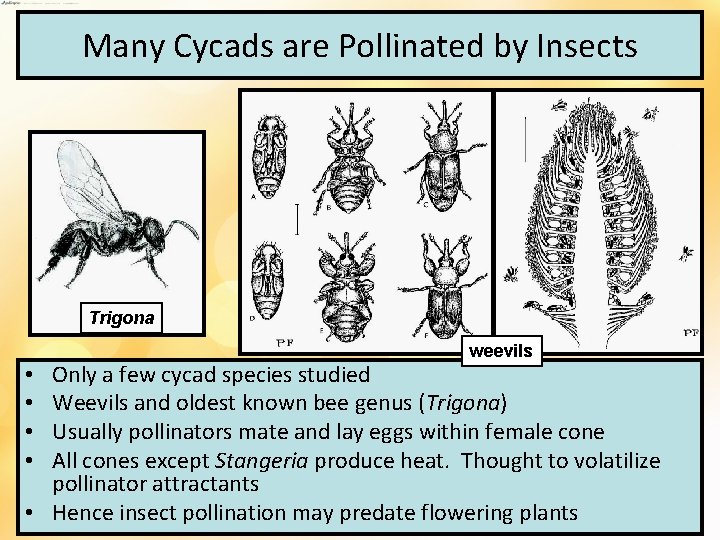
Many Cycads are Pollinated by Insects Trigona weevils Only a few cycad species studied Weevils and oldest known bee genus (Trigona) Usually pollinators mate and lay eggs within female cone All cones except Stangeria produce heat. Thought to volatilize pollinator attractants • Hence insect pollination may predate flowering plants • •


www 006. upp. so-net. ne. jp Zamia furfuracea Zamia pumila ♂ ♀ megasporofyly se 2 vajíčky www. botany. hawaii. edu http: //www. botany. hawaii. edu

Ceratozamia mexicana http: //en. academic. ru http: //bomennederland. wordpress. com Megastrobilus Megasporofyl Mikrostrobilus

Cycas revoluta mikrostrobilus

Cycads are gymnosperms (naked seeded), meaning that their unfertilized seeds (ovules) are open to the air to be directly fertilized by pollination, as contrasted with angiosperms, which have enclosed seeds with more complex fertilization arrangements. Cycads have very specialized pollinators, usually a specific kind of beetle. They have been reported to fix nitrogen in association with a cyanobacterium living in the roots. These blue-green algae produce a neurotoxin called BMAA that is found in the seeds of cycads. A neurotoxin cycling through the food chain from the cyanobacterium through cycads to its seeds, to bats eating the seeds, to humans consuming the bats, is hypothesized to be a source for some neurological diseases in humans