Ch 2 Math Review Scientific Notation Scientific Notation














- Slides: 15

Ch. 2 Math Review

Scientific Notation � Scientific Notation: a method for _____________________. �Moving the decimal to the ________exponent, to the _____________________. Ex. 0. 00527 g = 5. 27 x 10 -3 g 52700000 m = 5. 27 x 107 m 3. 25 x 10 -6 cm 3 = 0. 00000325 cm 3 3. 25 x 104 cm 3 = 32500 cm 3 2 -2

Scientific Notation �Practice: 1) 0. 000060731 ml = 2) 2. 970 x 10 -5 mg = 3) 546901000 kg = 4) 6. 3 x 107 s = 2 -3

Rounding Atomic Masses �Rounding Atomic Masses: we will be rounding the atomic masses off the periodic table to the _____________________ Ex. C = 12. 011 g/mol = 12. 01 g/mol Bi = 208. 98038 g/mol = 208. 98 g/mol K = 39. 0983 g/mol = 39. 10 g/mol

Rounding Atomic Masses �Practice: 5) N = 14. 00674 g/mol = 6) Si = 28. 0855 g/mol = 7) O = 15. 9994 g/mol = 8) Ar = 39. 948 g/mol = 2 -5

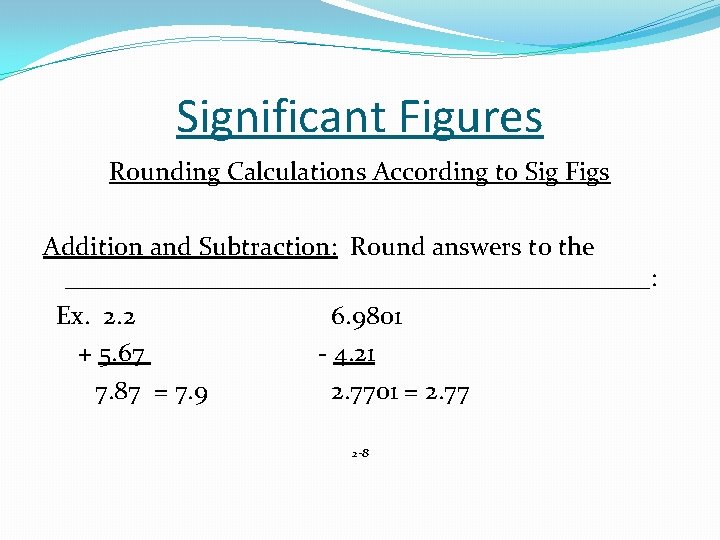
Significant Figures � Non zeros are always __________. � Zeros ___________are significant. � Leading and trailing zeros __________________. Atlantic/Pacific Rule: Pacific: Point Present, start from the left, first nonzero and everything after is significant Atlantic: Point Absent, start from the right, first nonzero and everything after is significant.

Significant Figures Ex. 1200 = 2 sig figs 1. 200 = 4 sig figs 0. 003 = 1 sig fig 0. 0030 = 2 sig figs Practice: 9) 1. 2650 = 10) 325000 = 11) 1. 280 x 10 -1 = 12) 31006 = 2 -7

Significant Figures Rounding Calculations According to Sig Figs Addition and Subtraction: Round answers to the _______________________: Ex. 2. 2 6. 9801 + 5. 67 - 4. 21 7. 87 = 7. 9 2. 7701 = 2. 77 2 -8

Significant Figures Practice: 13) 4. 71 - 1. 1 14) 5. 367 + 1. 2 2 -9

Significant Figures �Multiplication and Division: Round answers to the ________________________: Ex. 2. 60 x 300 = 780 = 800 52. 31 x 2600 = 136006 = 140000 2000000 ÷ 5. 63 = 355239. 7869 = 4000000 78 ÷ 6 =13 = 10 2 -10

Significant Figures Practice: 15) 240. ÷ 6. 0 = 16) 365 ÷ 5 = 17) 32 ÷ 3 = 18) 32 x 3. 0 = 2 -11

Dimensional Analysis �Used to convert between units. Take an equivalent (12 in. = 1 ft. ) and make it into a _________: 1 ft. or 12 in. 1 ft. Use conversion factors to convert between units. ________________________ and multiply by the conversion factor with _______________________. (so they cancel) 2 -12

Dimensional Analysis � Ex. 5. 36 ft. = ? in. 5. 36 ft. x 12 in. = 64. 3 in. 1 ft. � 32 in. = ? ft. 32 in. x 1 ft. = 2. 666 ft. = 2. 7 ft. 12 in. 12 -13

Dimensional Analysis � Ex. 3. 26 in. = ? cm 3. 26 in. x 1 cm= 8. 280416561 cm = 8. 28 cm 0. 3970 in. � 25 g = ? kg just use your handout, move your decimal 3 places to the left 25 g = 0. 025 kg � 5. 69 oz. = ? g 5. 69 oz. x 28. 3 g= 161. 027 g = 161 g 1 oz.

Dimensional Analysis � Ex. 1 year = ? S 1 year x 365 days x 24 hours x 60 min. X 60 s = 1 yr. 1 day 1 hr. 1 min. = 31536000 s = 30000000 s 12 -15