ZIFs zinc finger families MBV 4230 Zinc finger

ZIFs: zinc finger families

MBV 4230 Zinc finger proteins n n n Zinc finger proteins were first discovered as transcription factors. Zinc finger proteins are among the most abundant proteins in eukaryotic genomes. Their functions are extraordinarily diverse ¨ ¨ ¨ n include DNA recognition, RNA packaging, transcriptional activation, regulation of apoptosis, protein folding and assembly, and lipid binding. Zinc finger structures are as diverse as their functions.

MBV 4230 Zinc finger family: subfamilies with common DBD-structure n n TFIIIA - prototype and founder member Zinc fingers = Zn-structued domains binding DNA ¨ ¨ ¨ ¨ ¨ classical C 2 H 2 -fingers Nuclear receptors GATA-factors LIM domains GAL 4 -related factors Nucleocapsid proteins TFIIS RING finger PKC CRD
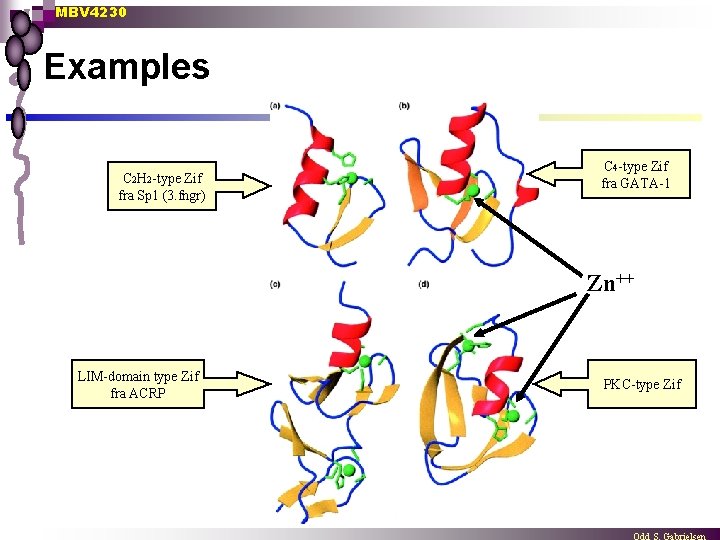
MBV 4230 Examples C 2 H 2 -type Zif fra Sp 1 (3. fngr) C 4 -type Zif fra GATA-1 Zn++ LIM-domain type Zif fra ACRP PKC-type Zif

MBV 4230 Alignments C-C-H-H C-C-C-C

The C 2 H 2 subfamily
![MBV 4230 Classical TFIIIA-related zinc fingers: n x [Zn-C 2 H 2] n History: MBV 4230 Classical TFIIIA-related zinc fingers: n x [Zn-C 2 H 2] n History:](http://slidetodoc.com/presentation_image/0fc976b0962f7422a1e1e9272a9b1760/image-7.jpg)
MBV 4230 Classical TFIIIA-related zinc fingers: n x [Zn-C 2 H 2] n History: Xenopus TFIIIA the first isolated and cloned eukaryotic TF Function: activation of 5 S RNA transcription (RNAPIII) ¨ Rich source : accumulated in immature Xenopus oocyttes as “storage particles” = TFIIIA+5 S RNA (≈ 15% of total soluble protein) ¨ Purified 1980, cloned in 1984 ¨ Mr= 38 600, 344 aa ¨ n Primary structure TFIIIA Composed of repeats: 9 x 30 aa minidomains + 70 aa unique region C-trm ¨ Each minidomain conserved pattern of 2 Cys+2 His ¨ Hypothesis: each minidomain structured around a coordinated zinc ion (senere bekreftet) ¨ ++ Zn++ Zn++ Zn. Zn
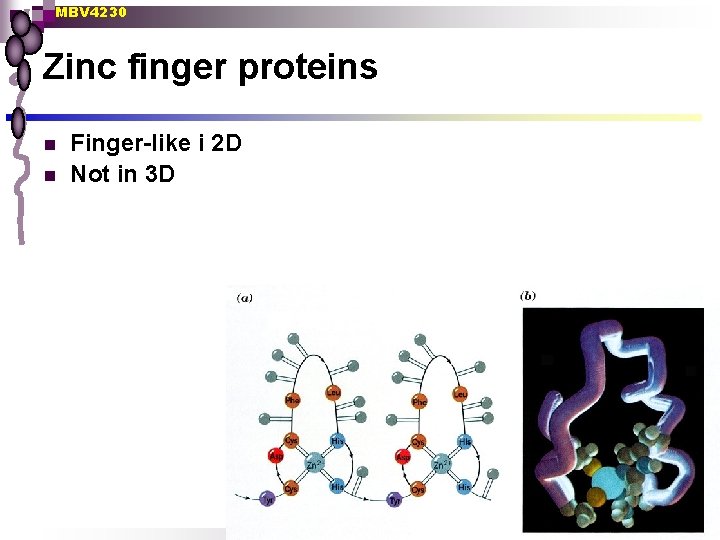
MBV 4230 Zinc finger proteins n n Finger-like i 2 D Not in 3 D

MBV 4230 Common features of TFIIIA-related zinc fingers n n n Consensus for each finger: FXCX 2 -5 CX 3 FX 5 FX 2 HX 2 -5 H Number of fingers in related factors varies: 2 -37 Number of members exceptionally high S. cerevisiae genome: 34 C 2 H 2 zinc fingers ¨ C. elegans genome 68 C 2 H 2 zinc fingers ¨ Drosophila genome 234 C 2 H 2 zinc fingers ¨ Humane genom 564 C 2 H 2 zinc fingers, (135 C 3 HC 4 zinc finger) ¨ n We now recognize the classical C 2 H 2 zinc finger as the first member of a rapidly expanding family of zinc-binding modules.

MBV 4230 3 D structure of the classical C 2 H 2 -type of zinc fingers n Each finger = a minidomain with -structure each finger an independent module ¨ Several fingers linked together by fleksible linkers ¨ First 3 D structure: the 3 -finger Zif 268 (mouse) ¨ n DNA interaction in Zif 268 major groove contact through -helix in ¨ recognition of base triplets ¨ aa in three positions responsible for sequence recognition: -1, 3 and 6 (rel. til -helix) ¨ Simpel one-to-one pattern (contact aa - baser) can a recognition code be defined ? ? ¨ n DNA interaction in GLI and TTK differs different phosphate contact ¨ distortion of DNA ¨ finger 1 without DNA contact ¨ D N A
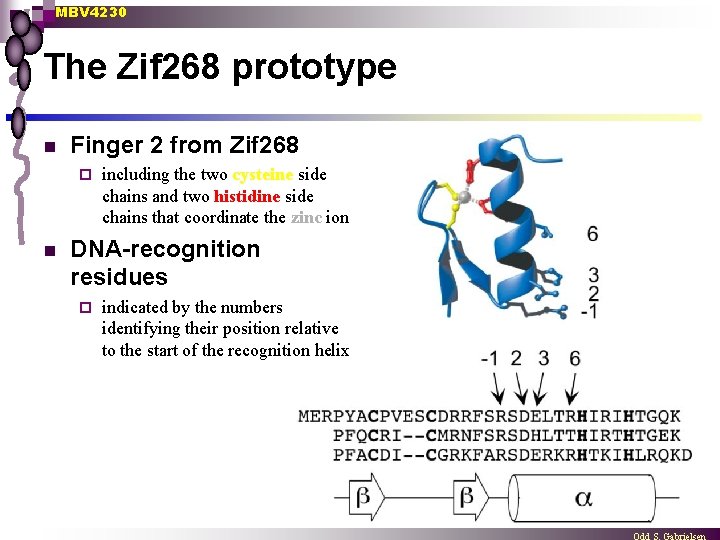
MBV 4230 The Zif 268 prototype n Finger 2 from Zif 268 ¨ n including the two cysteine side chains and two histidine side chains that coordinate the zinc ion DNA-recognition residues ¨ indicated by the numbers identifying their position relative to the start of the recognition helix
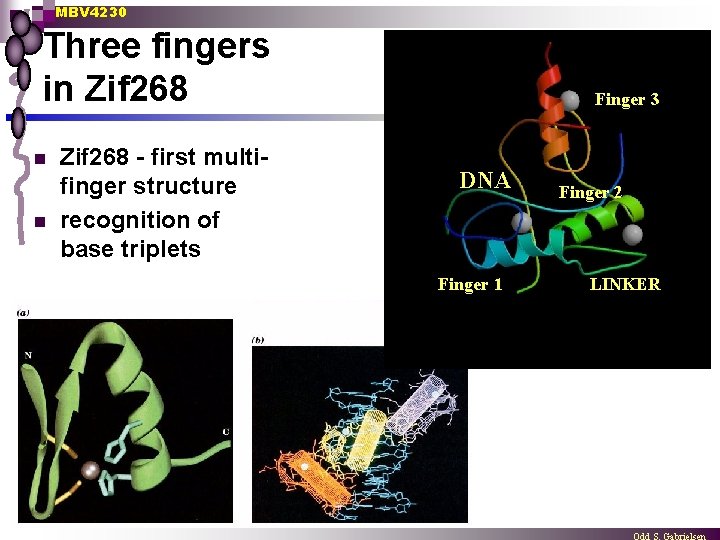
MBV 4230 Three fingers in Zif 268 n n Zif 268 - first multifinger structure recognition of base triplets Finger 3 DNA Finger 1 Finger 2 LINKER
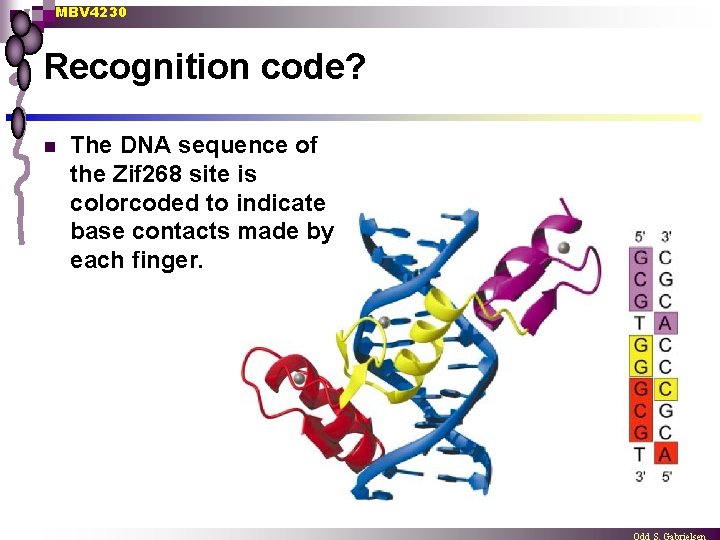
MBV 4230 Recognition code? n The DNA sequence of the Zif 268 site is colorcoded to indicate base contacts made by each finger.
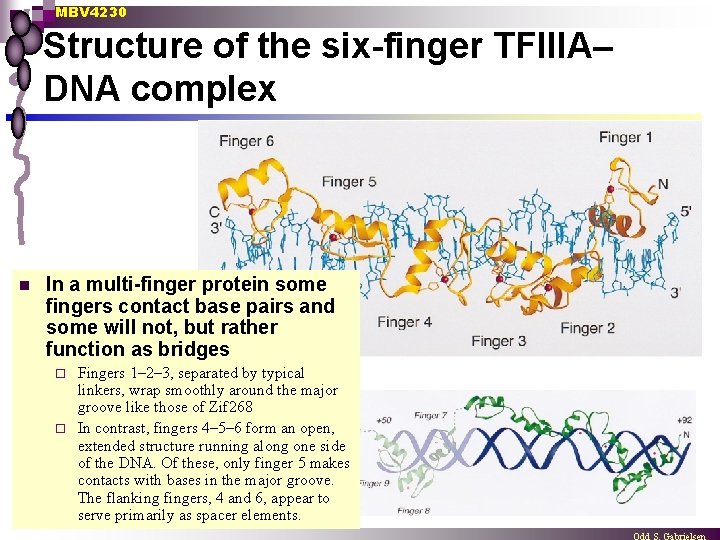
MBV 4230 Structure of the six-finger TFIIIA– DNA complex n In a multi-finger protein some fingers contact base pairs and some will not, but rather function as bridges Fingers 1– 2– 3, separated by typical linkers, wrap smoothly around the major groove like those of Zif 268 ¨ In contrast, fingers 4– 5– 6 form an open, extended structure running along one side of the DNA. Of these, only finger 5 makes contacts with bases in the major groove. The flanking fingers, 4 and 6, appear to serve primarily as spacer elements. ¨

MBV 4230 Linker connecting the fingers also important n Linker between fingers ¨ n Half of the known C 2 H 2 zinc finger proteins contain a highly conserved linker of sequence TGEKP that connects adjacent fingers. Function of linker The linker is dynamically disordered in the free protein, but adopts well-defined structure with restricted backbone flexibility upon binding to DNA. ¨ DNA-induced helix capping - diffusing Zif ≠ docked Zif ¨ n WT 1 - variant linker forms Zifs with different function WT 1 two splice variants: (+KTS) with an insertion or (–KTS) without in the TGEKP linker between the 3. and 4. zinc fingers. ¨ Modification of linker in vivo can have profound physiological consequences. ¨ n Frasier syndrome is caused by mutation that prevents the +KTS isoform. The –KTS isoform binds DNA with high affinity and regulates transcription; in contrast, the +KTS variant binds DNA weakly and associates preferentially with the splicing machinery, where it may interact with RNA. ¨ KTS insertion increases linker flexibility, abrogates binding of 4. finger to DNA. ¨
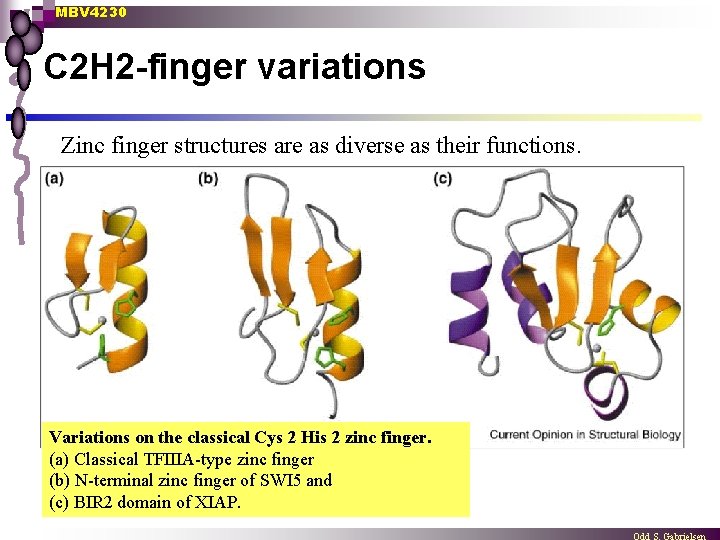
MBV 4230 C 2 H 2 -finger variations Zinc finger structures are as diverse as their functions. Variations on the classical Cys 2 His 2 zinc finger. (a) Classical TFIIIA-type zinc finger (b) N-terminal zinc finger of SWI 5 and (c) BIR 2 domain of XIAP.

MBV 4230 Zinc finger engineering n Effort focused on the design of novel C 2 H 2 zinc finger proteins that can specifically target unique binding sites within the human genome. ¨ applications as probes and may ultimately prove valuable for human gene therapy. ¨ Challenge: to achieve the high binding affinity and specificity ¨ 1 -2 -3 Zif increase binding affinity 1000 -fold for each additional finger. Only modest improvements in affinity occur >3 Zif ¨ n Design of dimerized Zif-dimers or triplets that recognize 10 bp with high affinity. ¨ Wolffe´s company

MBV 4230 Unsolved problems n Topological problem with a factor that is wrapped around DNA 3 fingers dekker en full turn of DNA ¨ krysning of minor groove nødvendig når Number of fingers >3 ¨ n RNA and DNA binding ¨ i TFIIIA: finger 1 -3 DNA-binding, 4 -6 RNA-binding

Kjernereceptors 2 x. C 4
![MBV 4230 Nuclear receptors: 2 x[Zn-C 4]: n n n Large family where DBD MBV 4230 Nuclear receptors: 2 x[Zn-C 4]: n n n Large family where DBD](http://slidetodoc.com/presentation_image/0fc976b0962f7422a1e1e9272a9b1760/image-20.jpg)
MBV 4230 Nuclear receptors: 2 x[Zn-C 4]: n n n Large family where DBD binds two Zn++ through a tetraedrical pattern of Cys conserved DBD 70 -80 aa Protein structure Two “zinc fingers” constitute one separate domain ¨ Two -helices with C 3 -Zn-C 4 N-terminally ¨ Disse vinkelrett på hverandre with hydrophobic overkrysning ¨ n n Mediates trx response to complex ekstracellular signals Evolutionary coupled to multicellular organisms Yeast = 0 but C. elegans 233 eller 1. 5% of genes !! ¨ Sequence prediksjon: 90% with nuclear receptor DBD has potential ligand-BD ¨ Implies that lipophilic signal molecules have been important to establish communication between cells ¨
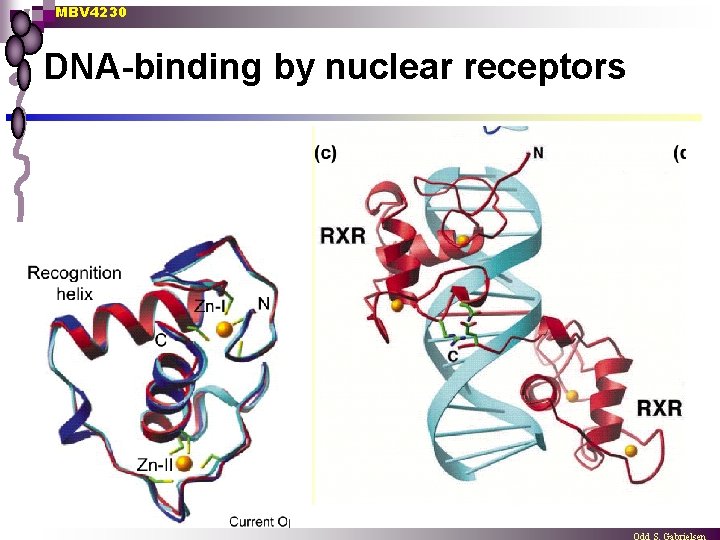
MBV 4230 DNA-binding by nuclear receptors
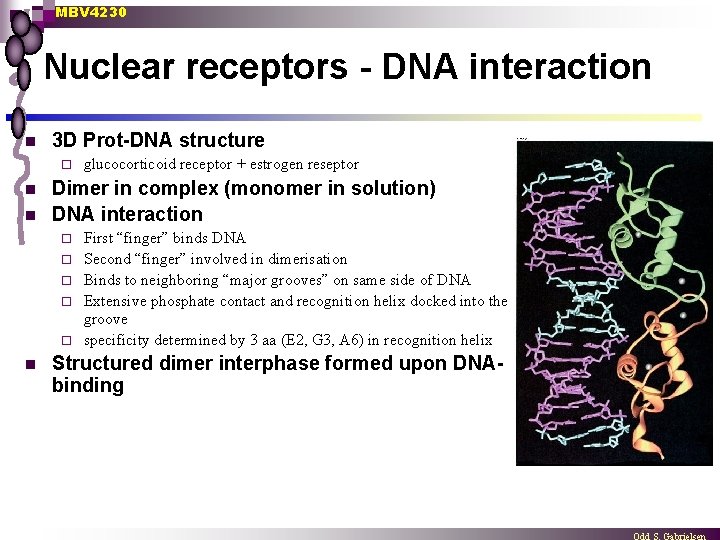
MBV 4230 Nuclear receptors - DNA interaction n 3 D Prot-DNA structure ¨ n n Dimer in complex (monomer in solution) DNA interaction ¨ ¨ ¨ n glucocorticoid receptor + estrogen reseptor First “finger” binds DNA Second “finger” involved in dimerisation Binds to neighboring “major grooves” on same side of DNA Extensive phosphate contact and recognition helix docked into the groove specificity determined by 3 aa (E 2, G 3, A 6) in recognition helix Structured dimer interphase formed upon DNAbinding

GATA factors
![MBV 4230 GATA-factors: 1 x [Zn-C 4]: n n n Small family Prototype erythroid MBV 4230 GATA-factors: 1 x [Zn-C 4]: n n n Small family Prototype erythroid](http://slidetodoc.com/presentation_image/0fc976b0962f7422a1e1e9272a9b1760/image-24.jpg)
MBV 4230 GATA-factors: 1 x [Zn-C 4]: n n n Small family Prototype erythroid TF: GATA-1 (2 fingers) From fungi to humans Structure ≈ 1. finger in nuclear receptors Hydrophobic DNA interphase Evolusjonary implicasjoner ¨ Early duplication of primitive finger divergent functions developed in NR

Gal 4 p factors
![MBV 4230 GAL 4 -related factors: 1 x [Zn 2 -C 6]: n GAL MBV 4230 GAL 4 -related factors: 1 x [Zn 2 -C 6]: n GAL](http://slidetodoc.com/presentation_image/0fc976b0962f7422a1e1e9272a9b1760/image-26.jpg)
MBV 4230 GAL 4 -related factors: 1 x [Zn 2 -C 6]: n GAL 4 -DBD = 28 aa cys-rich domain binds 2 Zn++ ¨ + 26 aa C-terminalt domain involv. in dimerization ¨ n Cys-rich domain consensus: CX 2 CX 6 CX 2 CX 6 C ¨ A Zn-Cys cluster with shared Cys (1. and 4. ) ¨ Two short -helicer with C-Zn-C N-terminalt ¨
![MBV 4230 GAL 4 -related factors: 1 x [Zn 2 -C 6]: n Dimerisation MBV 4230 GAL 4 -related factors: 1 x [Zn 2 -C 6]: n Dimerisation](http://slidetodoc.com/presentation_image/0fc976b0962f7422a1e1e9272a9b1760/image-27.jpg)
MBV 4230 GAL 4 -related factors: 1 x [Zn 2 -C 6]: n Dimerisation domain Monomer in solution, dimer in DNA-compleks ¨ In solution on ly Cys-rich motif structured ¨ In compleks forms two extended helix-strand motives ¨ Amfipathic helicer form a dimer-interphase in the complex ¨ n DNA interaction ¨ ¨ ¨ contacts CGG-triplets in major groove C-terminal of 1. -helix contacts bases Phosphate contact via helix-strand motif Coiled-coil dimer-interphase at right angle to DNA (≈b. ZIP) Linker determines spacing of CGG-tripletter: 11 bp in GAL 4, 6 bp in PPR 1

. . beyond DNA-binding

MBV 4230 >DNA-binding: A broader function for TFIIIA-type -folds n n Zif = Zinc sensors Zif = Protein-protein interaction domain ¨ Ikaros-homodimers n The zinc finger protein Ikaros, which plays a crucial role in lymphoid differentiation, forms homodimers through the association of the two C-terminal C 2 H 2 zinc finger motifs. Aiolos both homodimerizes and forms heterodimers with Ikaros through a two-zinc finger domain. ¨ GATA-1 interacts with FOG (”friend of GATA”) through a C 2 H 2 zinc finger in GATA-1 and CCHC-fingers in FOG ¨ n n n GATA-1 = 2 CCCC zinc fingers, N-terminal finger = both DNA- and FOG-binding FOG = 8 -9 zinc fingers, mix of CCHH and CCHC Zif = TAD (transactivation domain) N-terminal part = -folded C 2 H 2 Zif ¨ C-terminal part = unstructured ¨ n Zif in RNAPII (6 Zn-binding proteins)
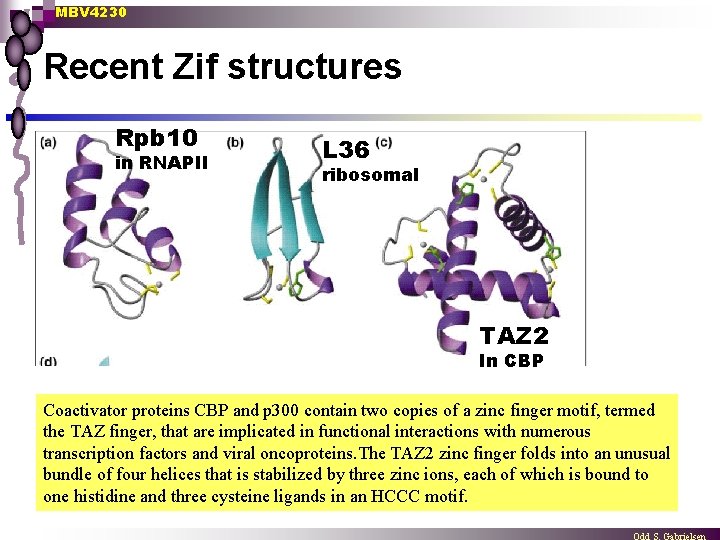
MBV 4230 Recent Zif structures Rpb 10 in RNAPII L 36 ribosomal TAZ 2 In CBP Coactivator proteins CBP and p 300 contain two copies of a zinc finger motif, termed the TAZ finger, that are implicated in functional interactions with numerous transcription factors and viral oncoproteins. The TAZ 2 zinc finger folds into an unusual bundle of four helices that is stabilized by three zinc ions, each of which is bound to one histidine and three cysteine ligands in an HCCC motif.
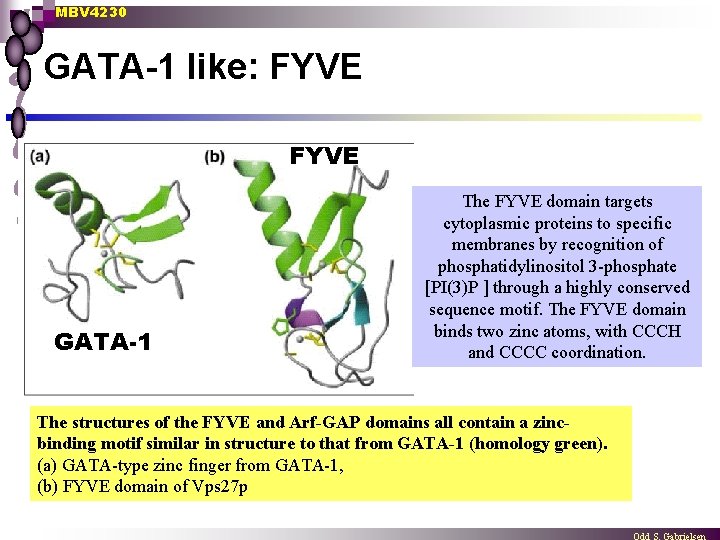
MBV 4230 GATA-1 like: FYVE GATA-1 The FYVE domain targets cytoplasmic proteins to specific membranes by recognition of phosphatidylinositol 3 -phosphate [PI(3)P ] through a highly conserved sequence motif. The FYVE domain binds two zinc atoms, with CCCH and CCCC coordination. The structures of the FYVE and Arf-GAP domains all contain a zincbinding motif similar in structure to that from GATA-1 (homology green). (a) GATA-type zinc finger from GATA-1, (b) FYVE domain of Vps 27 p
- Slides: 31