Zero and FirstOrder Rate Reactions Samir Kumar Khanal











- Slides: 21

Zero and First-Order Rate Reactions Samir Kumar Khanal, Ph. D. Department of Civil, Construction and Environmental Engineering Iowa State University

Question!! Assume you are a process engineer (biological), newly recruited by P & G. In your first week of job, you have been assigned to assist in the design of a bioreactor for growing edible fungi on synthetic growth medium to produce light-weight protein diet for astronauts. How are you going to design? Specifically what data do you need? Call your professor? Ask the senior engineer? Surf the internet? Consult BSE 482: lecture notes

Before you start designing a bioreactor (fermentor), you must have clear understanding of the followings: How fast the fungi are able to convert the organics into protein? That is bioconversion rate or reaction rate or kinetics. (C 2 -C 1)/(t 2 -t 1) = d. C/dt When is the reaction going to be over? “t” Which bioreactor configuration would be ideal? Suspended growth Attached growth

At the end of this class, you should be able to q define biochemical kinetics, reaction rate and order q derive the rate of a reaction in terms of the appearance of products or disappearance of reactants q describe the basic factors that influence the rate of a reaction q integrate the rate laws for 0, and 1 st order reactions q determine the rates and orders of the biochemical reactions q explain the practical significance of reaction rate and order

Reaction Rates • Definition – change in concentration of a reactant or product with time • The rate will be negative (-) for reactants • The rate will be positive (+) for products Reactants: mostly pollutants (we want to get rid of), e. g. nitrate, phosphate, organics, pesticides, etc. Products: mostly value-added commodities (we want to produce), e. g. protein, lactic acid, enzymes, nisin, yeast, etc.

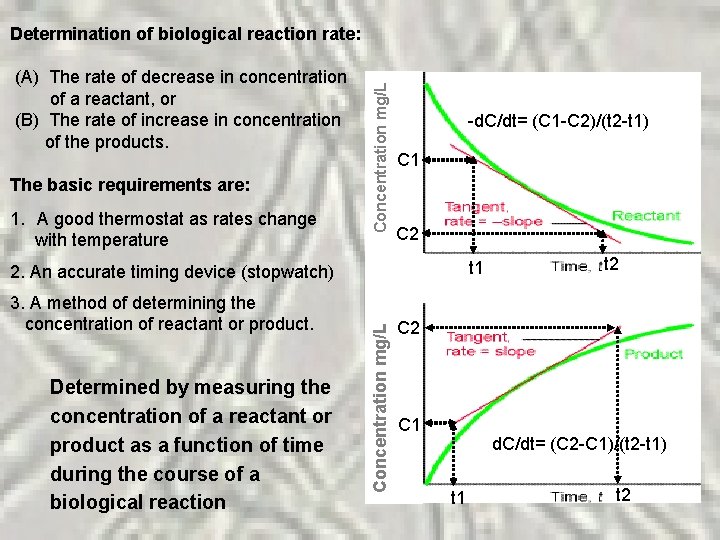
1. A good thermostat as rates change with temperature 2. An accurate timing device (stopwatch) 3. A method of determining the concentration of reactant or product. Determined by measuring the concentration of a reactant or product as a function of time during the course of a biological reaction -d. C/dt= (C 1 -C 2)/(t 2 -t 1) C 2 C 1 (A) The rate of decrease in concentration of a reactant, or (B) The rate of increase in concentration of the products. The basic requirements are: Concentration mg/L Determination of biological reaction rate: C 1 C 2 t 1 t 2 C 1 d. C/dt= (C 2 -C 1)/(t 2 -t 1) t 1 t 2

Factors affecting the speed or rate of a biological reaction q Concentration q Temperature q Presence of a macro/micro-nutrients q Physical state of reactants

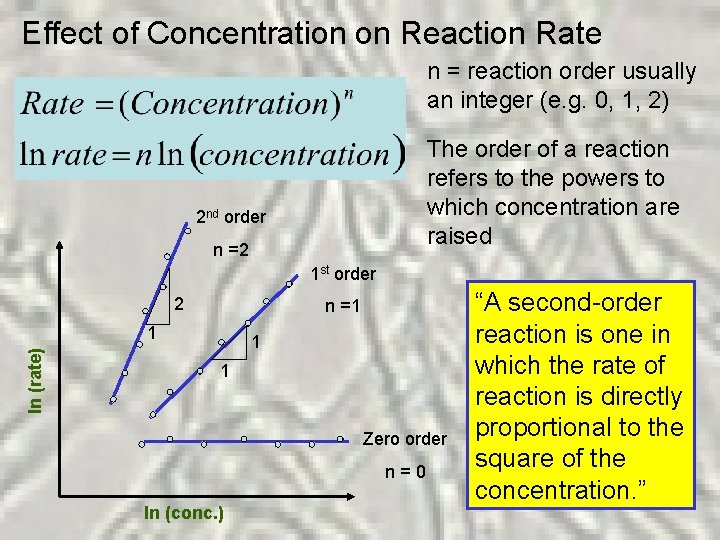
Effect of Concentration on Reaction Rate n = reaction order usually an integer (e. g. 0, 1, 2) The order of a reaction refers to the powers to which concentration are raised 2 nd order n =2 1 st order 2 n =1 ln (rate) 1 1 1 Zero order n = 0 ln (conc. ) “A second-order “A zero-order “A first-order reaction is one in which the rate of reaction is directly proportional to the independent of proportional to square of the concentration. ”

What does reaction order tell us? ? Relationship between rate and concentration! How the amount of compound speeds up or retards the reaction rate!

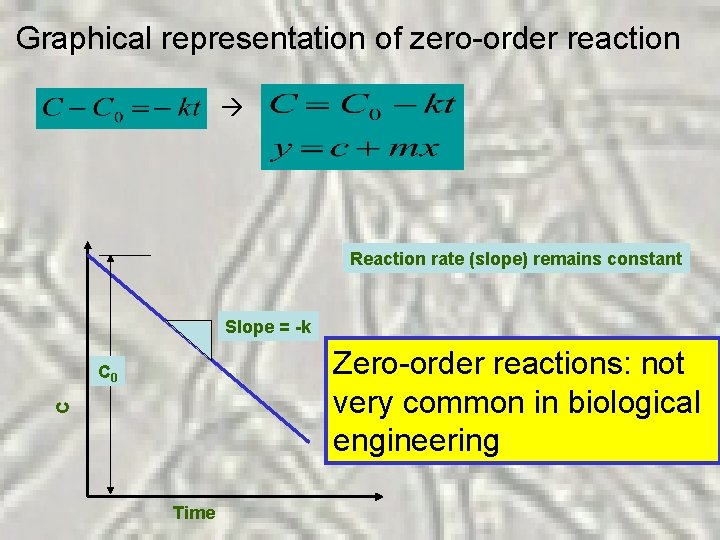
Zero-Order Reactions k = rate constant For zero-order reaction, n = 0 Negative means, [C] decreases with time unit of k is mass volume-1 time-1 If [C] increases with time (for product formation)

Graphical representation of zero-order reaction Reaction rate (slope) remains constant Slope = -k Zero-order reactions: not very common in biological engineering C C 0 Time

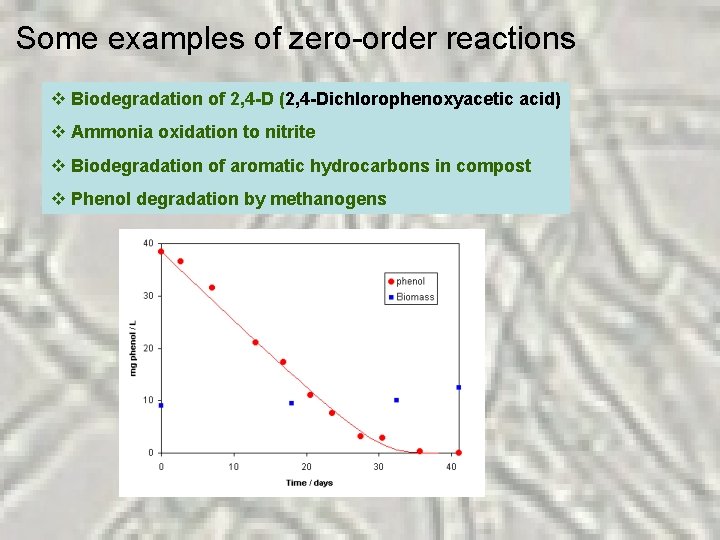
Some examples of zero-order reactions v Biodegradation of 2, 4 -D (2, 4 -Dichlorophenoxyacetic acid) v Ammonia oxidation to nitrite v Biodegradation of aromatic hydrocarbons in compost v Phenol degradation by methanogens

First-Order Reactions For first-order reaction, n = 1 k = rate constant Negative means, [C] decreases with time unit of k is time-1 Which is similar to a straight line equation

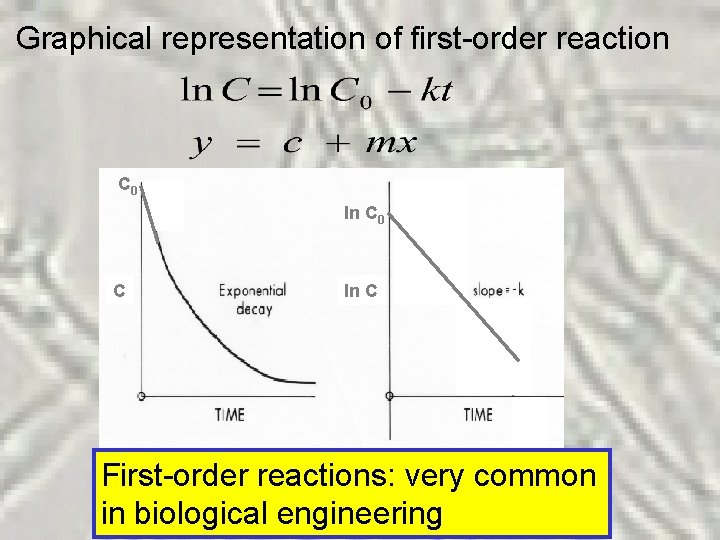
Graphical representation of first-order reaction C 0 ln C 0 C ln C First-order reactions: very common in biological engineering

Some examples of first-order reactions v Degradation of chlorinated compounds v Microbial growth (bacteria/fungi) v Oxidation of organic matter

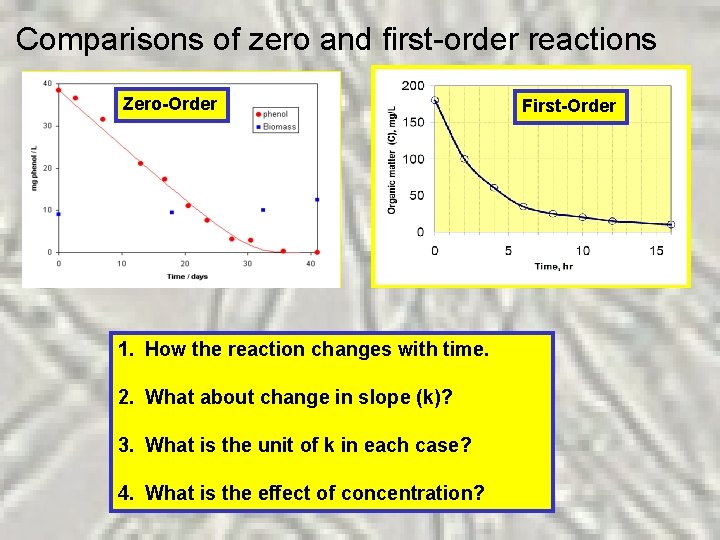
Comparisons of zero and first-order reactions Zero-Order 1. How the reaction changes with time. 2. What about change in slope (k)? 3. What is the unit of k in each case? 4. What is the effect of concentration? First-Order

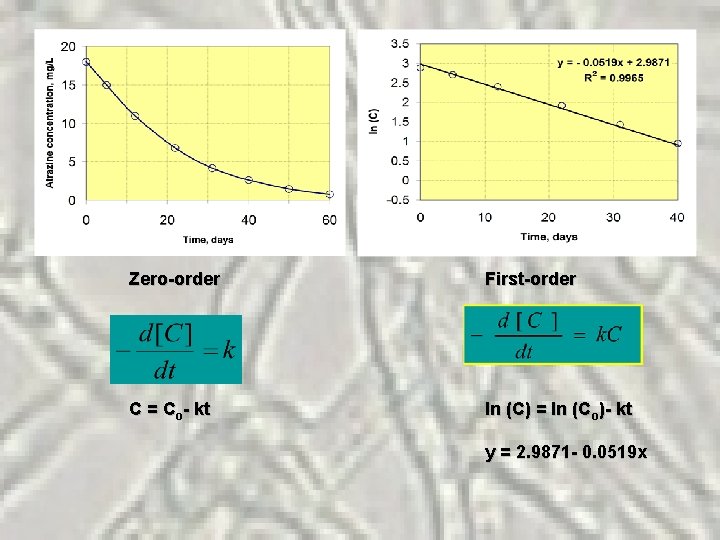
Example: An engineering student was interested in the biodegradation of atrazine in an aqueous environment, its reaction rate and order. She went to the lab and conducted a series of batch tests in shaker flasks at 25 o. C using an enriched microbial culture of Pseudomonas. During her experiments, she collected data every alternative day. The data are shown in the table below. Time, days Atrazine, mg/L 0 18 ln (C) 2. 890 5 15 2. 708 12 11 2. 398 22 6. 8 1. 917 31 4. 2 1. 435 40 2. 6 0. 956 50 1. 5 0. 405 60 0. 8 - 0. 223

Zero-order First-order C = Co- kt ln (C) = ln (Co)- kt y = 2. 9871 - 0. 0519 x

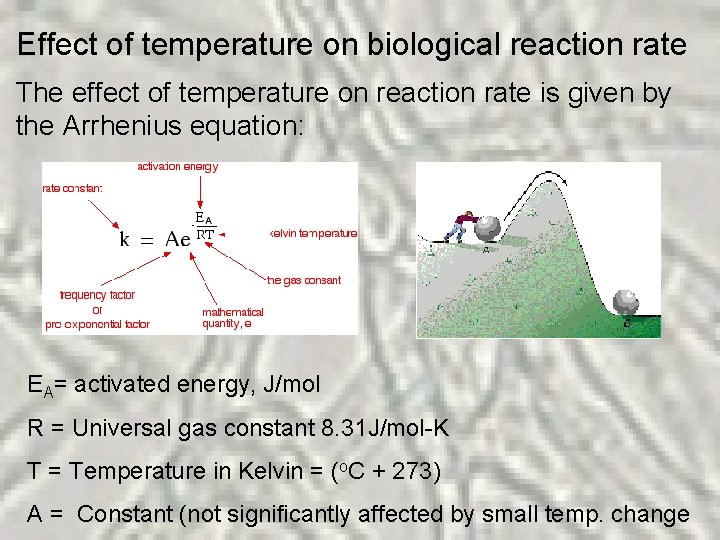
Effect of temperature on biological reaction rate The effect of temperature on reaction rate is given by the Arrhenius equation: EA= activated energy, J/mol R = Universal gas constant 8. 31 J/mol-K T = Temperature in Kelvin = (o. C + 273) A = Constant (not significantly affected by small temp. change

What happens if you increase the temperature by 10°C from, say, 20°C to 30°C (293 K to 303 K)? Let's assume an activation energy, EA of 50, 000 J mol-1. gas constant, R, is 8. 31 J K -1 mol-1. At 20°C (293 K), the value of the fraction: At 30°C (303 K), the fraction is: Rule of thumb: Rate of a reaction doubles for every 10 degree rise in temperature

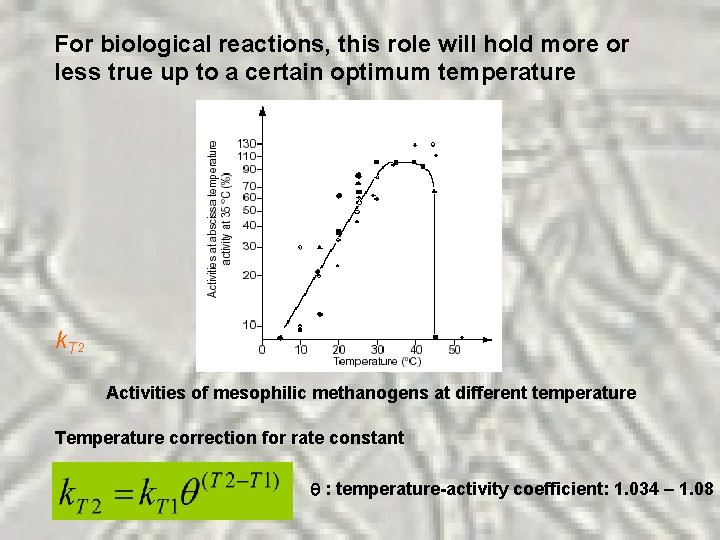
For biological reactions, this role will hold more or less true up to a certain optimum temperature k. T 2 Activities of mesophilic methanogens at different temperature Temperature correction for rate constant : temperature-activity coefficient: 1. 034 – 1. 08