Zeinab Mokhtari 1 15 December2009 Two important fundamental

Zeinab Mokhtari 1 15 -December-2009

Two important fundamental molecular properties : - The ionization potential (IP) - The electron affinity (EA) The ionization potential (IP) is the energy required to remove an electron from a molecule. R → R+ + e H = IP Definitively can be measured, The electron affinity (EA) is the energy difference because it is large. between the anion and the neutral. R + e → R- H = -EA It has a small value. So difficult to be measured. 2
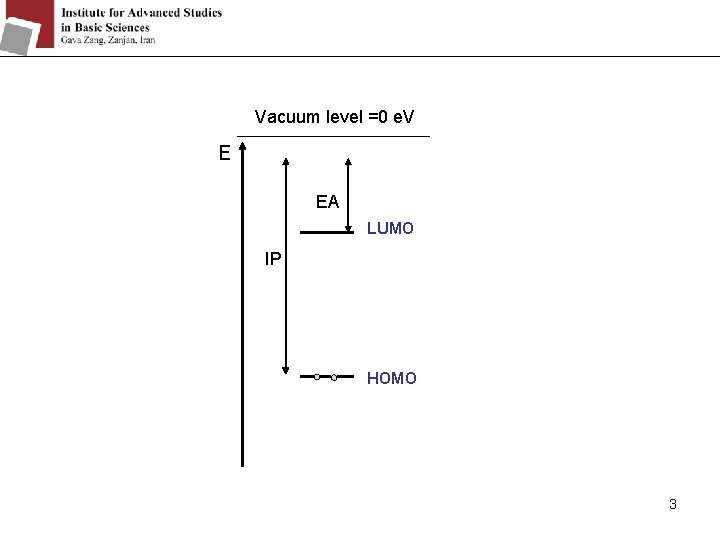
Vacuum level =0 e. V E EA LUMO IP HOMO 3
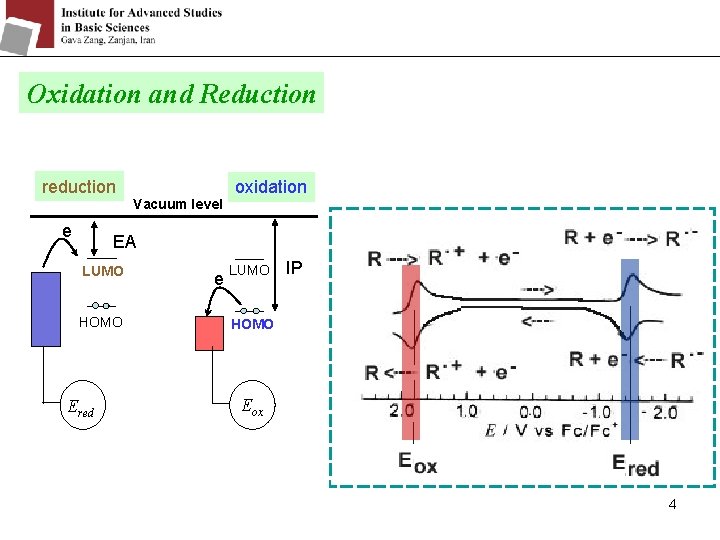
Oxidation and Reduction reduction e Vacuum level oxidation EA LUMO HOMO Ered e LUMO IP HOMO Eox 4

The major purpose : To obtain EA of aromatic hydrocarbons from half wave reduction potentials in aprotic solvents - Fullerenes - Organic molecules containing O, N, and halogen atoms - Nucleic acids and other biologically significant molecules A second objective: To calculate the electronegativities, (EN)=(IP+EA)/2 , of the aromatic hydrocarbons by using ionization potentials in the literature. 5
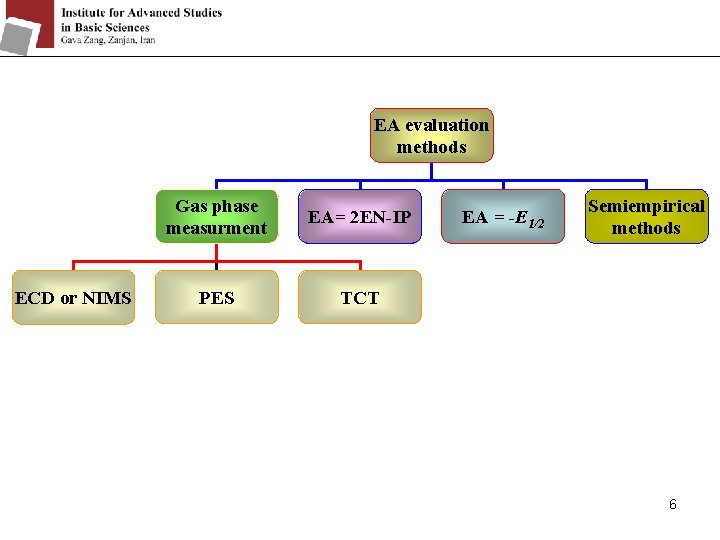
EA evaluation methods ECD or NIMS Gas phase measurment EA= 2 EN-IP PES TCT EA = -E 1/2 Semiempirical methods 6

THE METHOD OF CALCULATING EA’S FROM HALF WAVE REDUCTION POTENTIALS The first experimental method for obtaining relative electron affinities. - G is the solution energy difference for the reaction in the gas phase and in solution. mdd. G(- G) is a function of charge density. 7

Before the 1960’s: it was assumed that the mdd. G were the same for different molecules. By gas phase electron affinities measurement, the mdd. G values range from 1. 7 e. V for the large fullerenes to 2. 7 e. V for small anions. They make some different groups with different mdd. G: 8
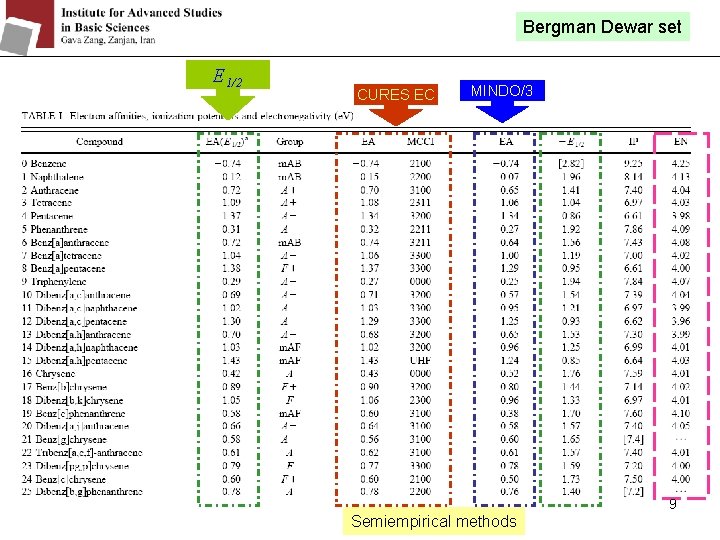
Bergman Dewar set E 1/2 CURES EC MINDO/3 9 Semiempirical methods
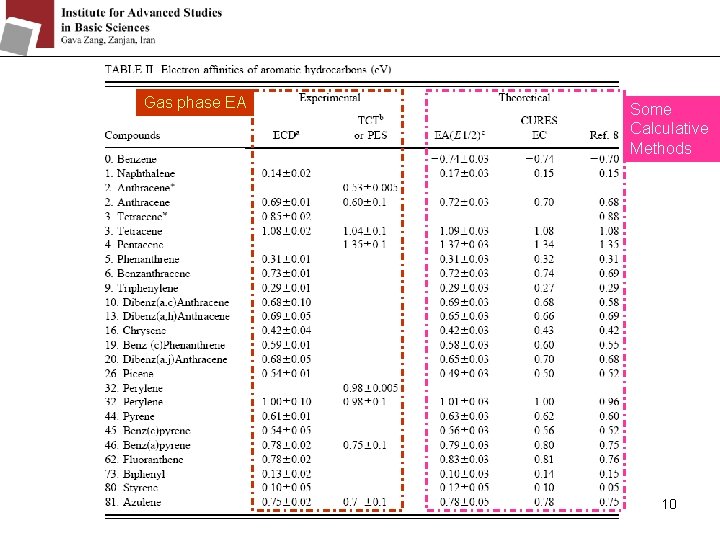
Gas phase EA Some Calculative Methods 10

11

12
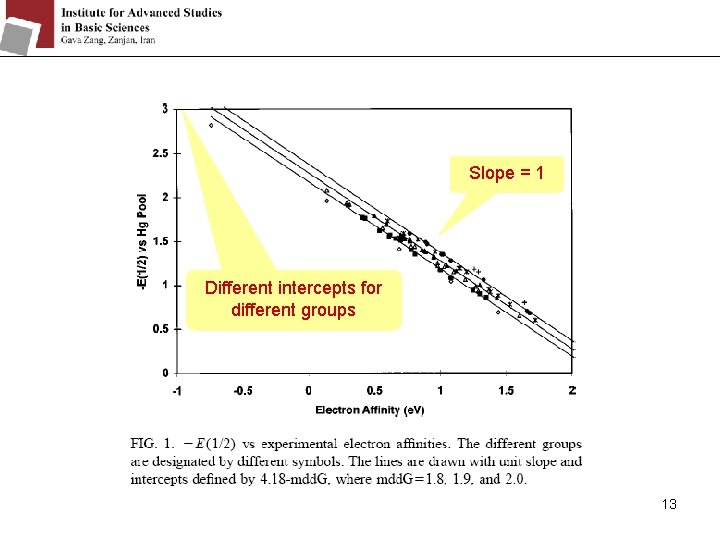
Slope = 1 Different intercepts for different groups 13
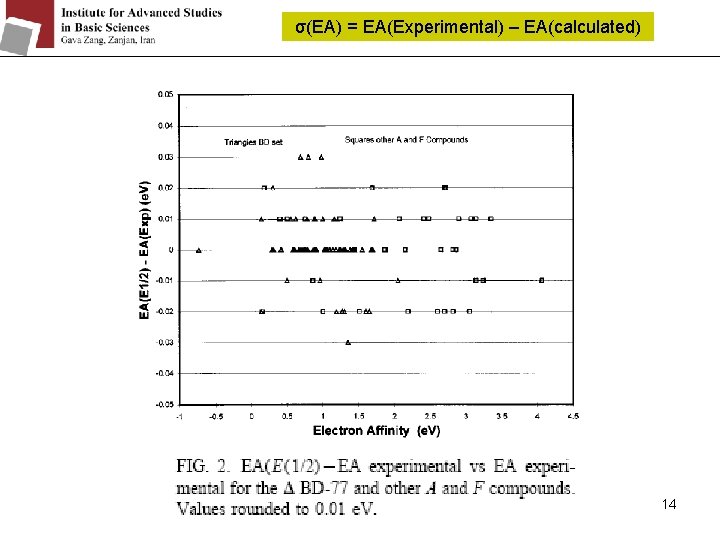
σ(EA) = EA(Experimental) – EA(calculated) 14
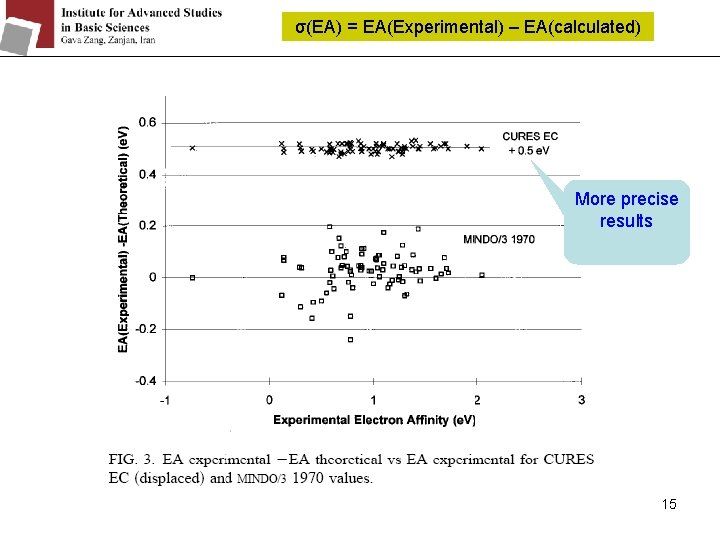
σ(EA) = EA(Experimental) – EA(calculated) More precise results 15
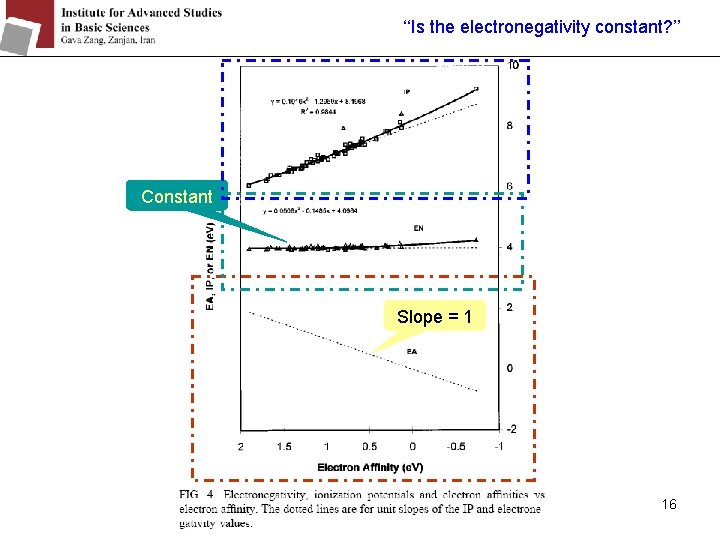
‘‘Is the electronegativity constant? ’’ Constant Slope = 1 16
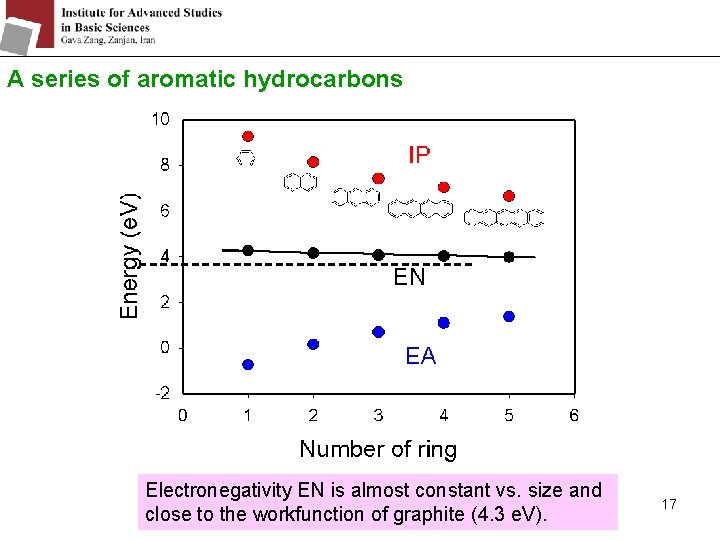
A series of aromatic hydrocarbons Electronegativity EN is almost constant vs. size and close to the workfunction of graphite (4. 3 e. V). 17

18
- Slides: 18