You Cant Learn Blood Gases from a Lecture

You Can’t Learn Blood Gases from a Lecture! Lawrence Martin, M. D. Clinical Professor of Medicine Case Western Reserve University School of Medicine, Cleveland larry. martin@roadrunner. com April 1, 2010 3/1/2021 1
Blood Gas Interpretation means analyzing the data to determine patient’s state of: Ventilation Oxygenation Acid-Base You can’t learn this from a lecture! 3/1/2021 2

To interpret ABGs you need to look at all the relevant data

To interpret ABGs you need to look at all the relevant data From the blood gas machine Pa. CO 2 Pa. O 2 p. H HCO 3 From the Co-oximeter Sa. O 2 Co. Hb Met. Hb

To interpret ABGs you need to look at all the relevant data From the blood gas machine Pa. CO 2 Pa. O 2 p. H HCO 3 From the Co-oximeter Sa. O 2 Co. Hb Met. Hb From venous blood Na+ K+ Cl. HCO 3 BUN Creatinine Hgb

To interpret ABGs you need to look at all the relevant data From the blood gas machine Pa. CO 2 Pa. O 2 p. H HCO 3 From the Co-oximeter Sa. O 2 Co. Hb Met. Hb From venous blood Na+ K+ Cl. HCO 3 BUN Creatinine Hgb From the environment FIO 2 Barometric pressure
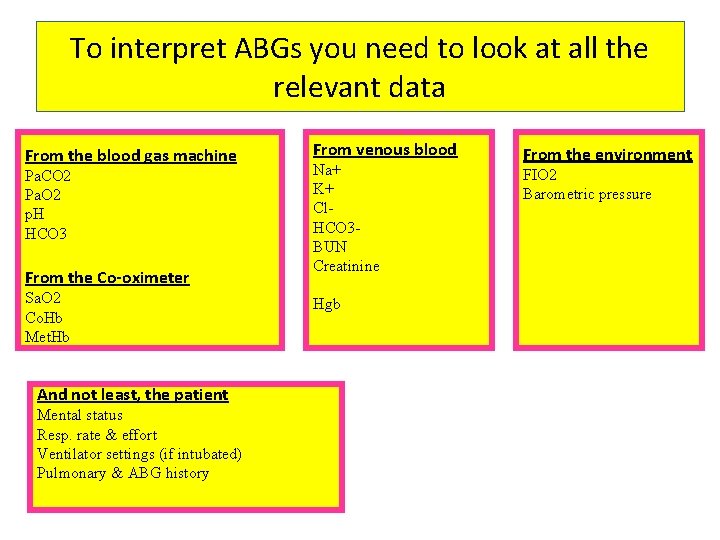
To interpret ABGs you need to look at all the relevant data From the blood gas machine Pa. CO 2 Pa. O 2 p. H HCO 3 From the Co-oximeter Sa. O 2 Co. Hb Met. Hb And not least, the patient Mental status Resp. rate & effort Ventilator settings (if intubated) Pulmonary & ABG history From venous blood Na+ K+ Cl. HCO 3 BUN Creatinine Hgb From the environment FIO 2 Barometric pressure
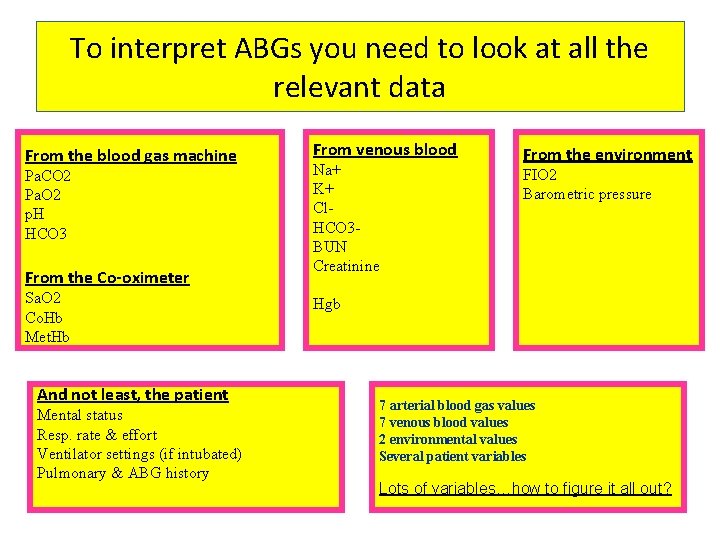
To interpret ABGs you need to look at all the relevant data From the blood gas machine Pa. CO 2 Pa. O 2 p. H HCO 3 From the Co-oximeter Sa. O 2 Co. Hb Met. Hb And not least, the patient Mental status Resp. rate & effort Ventilator settings (if intubated) Pulmonary & ABG history From venous blood Na+ K+ Cl. HCO 3 BUN Creatinine From the environment FIO 2 Barometric pressure Hgb 7 arterial blood gas values 7 venous blood values 2 environmental values Several patient variables Lots of variables…how to figure it all out?

Give a man a fish and you feed him for a day. Teach a man to fish and you feed him for a lifetime. Chinese Proverb What does this have to do with blood gas interpretation? 3/1/2021 9

Just this. You need a framework, a foundation to properly learn ABG interpretation. If I tell you how to interpret a given blood gas, you will understand that blood gas only, and not the next one you may encounter. Much better if I show you an approach to interpreting all blood gases, in all situations, so that you understand them. But… …like any other skill based on interpreting a large number of variables (EKG, chest x-ray, physical exam), You Can’t Learn Blood Gases from a Lecture. 3/1/2021 10

The best way to learn ABGs is to work on blood gas problems with some knowledge of basic physiology, then check your work for instant feedback. This ‘iterative process’ will teach you blood gas interpretation. Books Web sites You Can’t Learn Arterial Blood Gases from a Lecture You CAN learn ABGs from selected web sites. See list at: www. lakesidepress. com/pulmonary/AB G/PREFACEnew. htm 3/1/2021 11
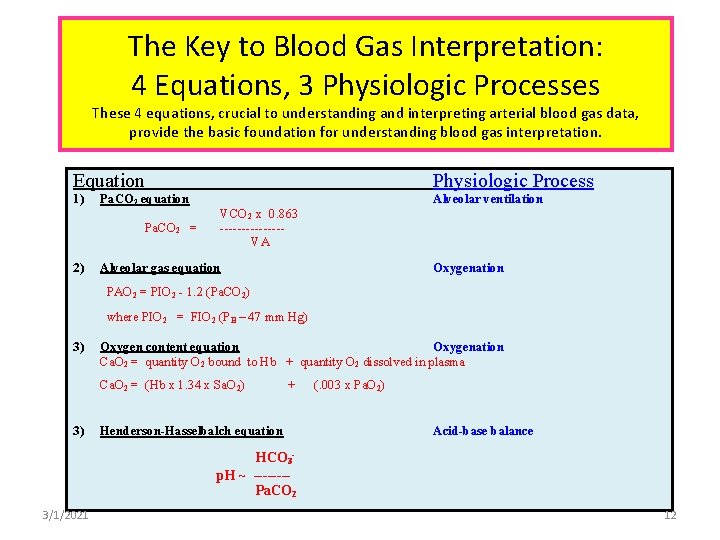
The Key to Blood Gas Interpretation: 4 Equations, 3 Physiologic Processes These 4 equations, crucial to understanding and interpreting arterial blood gas data, provide the basic foundation for understanding blood gas interpretation. Equation 1) Pa. CO 2 equation Pa. CO 2 = 2) Physiologic Process Alveolar ventilation VCO 2 x 0. 863 -------VA Alveolar gas equation Oxygenation PAO 2 = PIO 2 - 1. 2 (Pa. CO 2) where PIO 2 = FIO 2 (PB – 47 mm Hg) 3) Oxygen content equation Oxygenation Ca. O 2 = quantity O 2 bound to Hb + quantity O 2 dissolved in plasma Ca. O 2 = (Hb x 1. 34 x Sa. O 2) 3) + Henderson-Hasselbalch equation (. 003 x Pa. O 2) Acid-base balance HCO 3 p. H ~ -------Pa. CO 2 3/1/2021 12

Start with Pa. CO 2 is the center of the blood gas universe. p. H Pa. O 2 Pa. CO 2 HCO 3 Sa. O 2
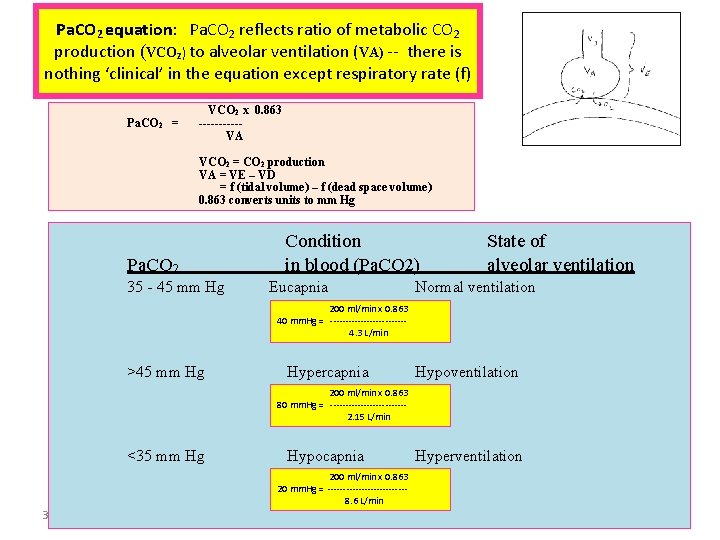
Pa. CO 2 equation: Pa. CO 2 reflects ratio of metabolic CO 2 production (VCO 2) to alveolar ventilation (VA) -- there is nothing ‘clinical’ in the equation except respiratory rate (f) Pa. CO 2 = VCO 2 x 0. 863 -----VA VCO 2 = CO 2 production VA = VE – VD = f (tidal volume) – f (dead space volume) 0. 863 converts units to mm Hg Pa. CO 2 35 - 45 mm Hg Condition in blood (Pa. CO 2) Eucapnia State of alveolar ventilation Normal ventilation 200 ml/min x 0. 863 40 mm. Hg = ------------4. 3 L/min >45 mm Hg Hypercapnia Hypoventilation 200 ml/min x 0. 863 80 mm. Hg = ------------2. 15 L/min <35 mm Hg Hypocapnia Hyperventilation 200 ml/min x 0. 863 20 mm. Hg = -------------8. 6 L/min 3/1/2021 14

Hypercapnia A serious respiratory problem VCO 2 x 0. 863 Pa. CO 2 = ------- VA where VA = VE – VD = f (tidal vol. ) – f (dead space vol. ) • The Pa. CO 2 equation shows that the only physiologic reason for elevated Pa. CO 2 is inadequate alveolar ventilation (VA) for the amount of CO 2 production (VCO 2). Since VA = VE – VD, hypercapnia can arise from insufficient VE (eg, drug overdose), increased VD (eg, COPD), or a combination. • The Pa. CO 2 equation also shows why Pa. CO 2 cannot reliably be assessed clinically. Since you never know the patient's VCO 2 or VA, you cannot determine the VCO 2/VA, which is what Pa. CO 2 provides. (Even if tidal volume is measured, you can’t determine the amount of air going to dead space. ) • There is no predictable correlation between Pa. CO 2 and the clinical picture. In a patient with possible respiratory disease, respiratory rate, depth, and effort cannot be reliably used to predict even a directional change in Pa. CO 2. A patient in respiratory distress can have a high, normal, or low Pa. CO 2. A patient without respiratory distress can have a high, normal, or low Pa. CO 2. 3/1/2021 15

Pa. CO 2 and alveolar ventilation A 60 yo man with severe chronic obstructive pulmonary disease is seen in the ED, anxious and tachypneic with RR=30/minute. The intern (who didn’t attend this lecture) says “he’s hyperventilating” and wants to give him Xanax. You, being wiser (since you attended), know better and demand a blood gas. Blood gas shows: Pa. CO 2 = 85 mm Hg (severe hypo ventilation) p. H = 7. 23 Explain tachypnea and hypoventilation in this patient. 3/1/2021 16

Pa. CO 2 and alveolar ventilation 60 yo man Pa. CO 2 = 85 mm Hg p. H = 7. 23 Pa. CO 2 = VCO 2 x 0. 863 ------VA where VA = VE – VD = f (tidal volume) – f (dead space volume) Clinically all you know is this patient’s resp. rate, the “f” in the Pa. CO 2 equation. You don’t know his tidal volume, dead space volume or CO 2 production, i. e. , you don’t know the numerator or denominator of the equation. Thus by exam alone, you cannot know even if he’s hypo- or hyper- ventilating. The blood gas shows he’s hypoventilating. Though tachypneic, MOST OF EACH BREATH IS GOING TO DEAD SPACE, NOT TO FUNCTIONING ALVEOLI! There is no predictable correlation between Pa. CO 2 and the clinical picture. In a patient with possible respiratory disease, respiratory rate, depth, and effort cannot be reliably used to predict even a directional change in Pa. CO 2. A patient in respiratory distress can have a high, normal, or low Pa. CO 2. A patient without respiratory distress can have a high, normal, or low Pa. CO 2. 3/1/2021 17
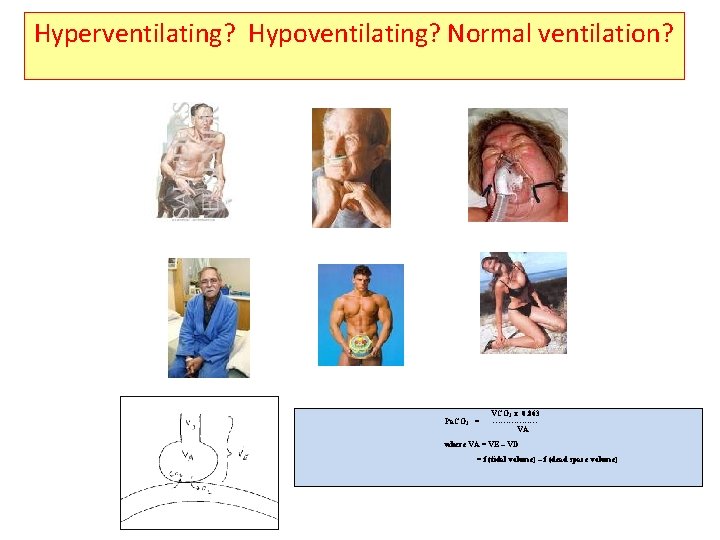
Hyperventilating? Hypoventilating? Normal ventilation? Pa. CO 2 = VCO 2 x 0. 863 --------VA where VA = VE – VD = f (tidal volume) – f (dead space volume)

Pa. CO 2 is the center of the blood gas universe. Understanding Pa. CO 2 facilitates understanding oxygenation & acid-base balance. Pa. CO 2 Oxygenation Acid-Base PAO 2 = FIO 2 (BP-47) – 1. 2 (PCO 2) p. H ~ HCO 3 - ------Pa. CO 2 = VCO 2 x. 863 ----------VA where VA = VE – VD = f (tidal volume) – f (dead space volume) Note: Pa. CO 2 is from ABGs. HCO 3 - is calculated from ABG measurement of p. H and Pa. CO 2 or is measured in venous blood (serum) as part of electrolytes. When measured in venous blood it is variously labeled ‘bicarbonate’, ‘HCO 3 - ’ or ‘CO 2’ (the latter NOT to be confused with Pa. CO 2).
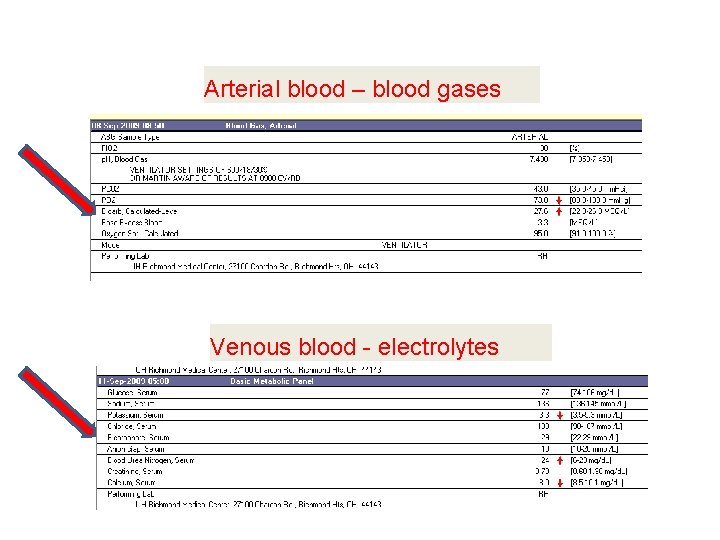
Arterial blood – blood gases Venous blood - electrolytes
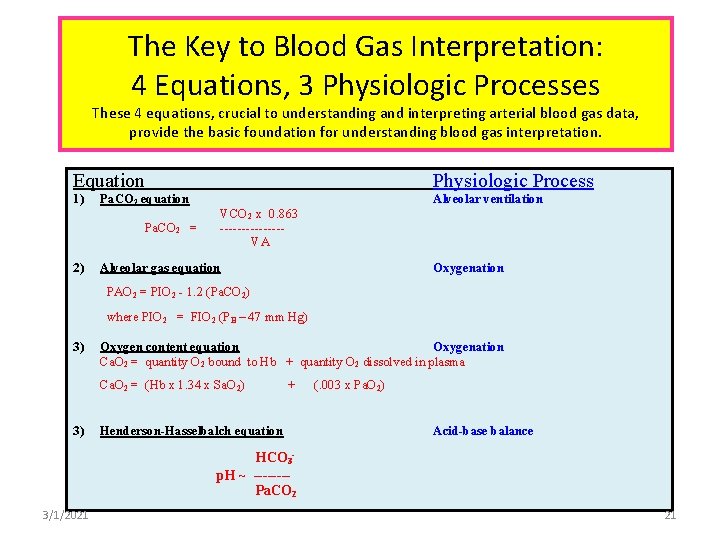
The Key to Blood Gas Interpretation: 4 Equations, 3 Physiologic Processes These 4 equations, crucial to understanding and interpreting arterial blood gas data, provide the basic foundation for understanding blood gas interpretation. Equation 1) Pa. CO 2 equation Pa. CO 2 = 2) Physiologic Process Alveolar ventilation VCO 2 x 0. 863 -------VA Alveolar gas equation Oxygenation PAO 2 = PIO 2 - 1. 2 (Pa. CO 2) where PIO 2 = FIO 2 (PB – 47 mm Hg) 3) Oxygen content equation Oxygenation Ca. O 2 = quantity O 2 bound to Hb + quantity O 2 dissolved in plasma Ca. O 2 = (Hb x 1. 34 x Sa. O 2) 3) + Henderson-Hasselbalch equation (. 003 x Pa. O 2) Acid-base balance HCO 3 p. H ~ -------Pa. CO 2 3/1/2021 21

Alveolar Gas Equation PAO 2 = PIO 2 - 1. 2 (Pa. CO 2) PAO 2 is the average alveolar PO 2 PIO 2 is the partial pressure of inspired oxygen in the trachea. PIO 2 = FIO 2 (PB – 47 mm Hg) = 0. 21 (760 -47) = 150 mm Hg Breathing room air at sea level, PAO 2 = 150 – 1. 2 (40) = 150 - 48 = 102 mm Hg Note: FIO 2 is fraction of inspired oxygen and PB is the barometric pressure. 47 mm Hg is the water vapor pressure at normal body temperature. This is the ‘abbreviated version’ of the AG equation, suitable for clinical purposes. 3/1/2021 22

Alveolar Gas Equation PAO 2 = FIO 2 (PB – 47 mm Hg) – 1. 2(Pa. CO 2) In order to bring O 2 into the blood, alveolar PO 2 (PAO 2) has to always exceed arterial PO 2 (Pa. O 2). Whenever PAO 2 decreases, Pa. O 2 decreases as well. Thus, from the AG equation: • If FIO 2 and PB are constant (i. e. , constant PIO 2), then as Pa. CO 2 increases both PAO 2 and Pa. O 2 will decrease: hypercapnia causes hypoxemia. • If FIO 2 decreases and PB and Pa. CO 2 are constant, both PAO 2 and Pa. O 2 will decrease: suffocation causes hypoxemia. • If PB decreases (e. g. , with altitude), and Pa. CO 2 and FIO 2 are constant, both PAO 2 and Pa. O 2 will decrease: mountain climbing causes hypoxemia. 3/1/2021 See web site: “Blood Gases on Mt. Everest” www. lakesidepress. com/pulmonary/Mt. Everest/bloodgases. htm. 23

Alveolar Gas Equation PAO 2 = FIO 2 (PB – 47 mm Hg) – 1. 2(Pa. CO 2) What is the alveolar PO 2 (PAO 2) at sea level* in the following circumstances? a) FIO 2 =. 21, Pa. CO 2 = 20 mm Hg ANSWER: PAO 2 =. 21(713) - 1. 2(20) = 126 mm Hg b) FIO 2 =. 21, Pa. CO 2 = 60 mm Hg ANSWER: PAO 2 =. 21(713) - 1. 2(60) = 72 mm Hg c) FIO 2 =. 40, Pa. CO 2 = 30 mm Hg ANSWER: PAO 2 =. 40(713) – (1. 2)30 = 249 mm Hg *BP = 760 mm Hg 3/1/2021 24
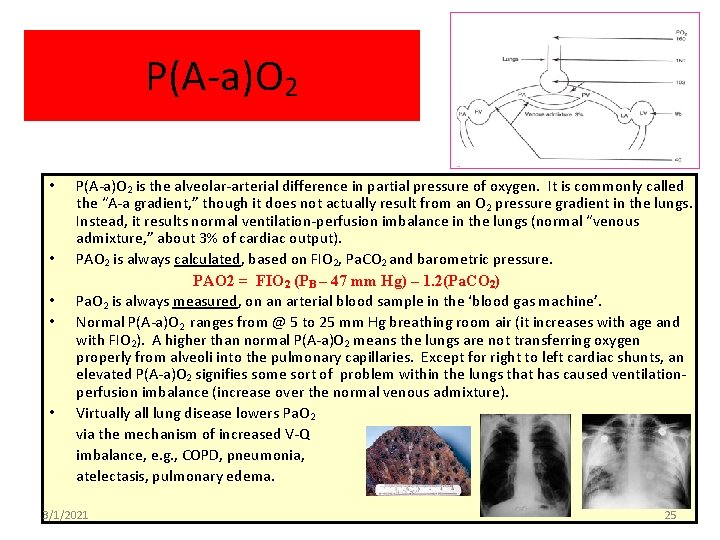
P(A-a)O 2 • • • P(A-a)O 2 is the alveolar-arterial difference in partial pressure of oxygen. It is commonly called the “A-a gradient, ” though it does not actually result from an O 2 pressure gradient in the lungs. Instead, it results normal ventilation-perfusion imbalance in the lungs (normal “venous admixture, ” about 3% of cardiac output). PAO 2 is always calculated, based on FIO 2, Pa. CO 2 and barometric pressure. PAO 2 = FIO 2 (PB – 47 mm Hg) – 1. 2(Pa. CO 2) Pa. O 2 is always measured, on an arterial blood sample in the ‘blood gas machine’. Normal P(A-a)O 2 ranges from @ 5 to 25 mm Hg breathing room air (it increases with age and with FIO 2). A higher than normal P(A-a)O 2 means the lungs are not transferring oxygen properly from alveoli into the pulmonary capillaries. Except for right to left cardiac shunts, an elevated P(A-a)O 2 signifies some sort of problem within the lungs that has caused ventilationperfusion imbalance (increase over the normal venous admixture). Virtually all lung disease lowers Pa. O 2 via the mechanism of increased V-Q imbalance, e. g. , COPD, pneumonia, atelectasis, pulmonary edema. 3/1/2021 25
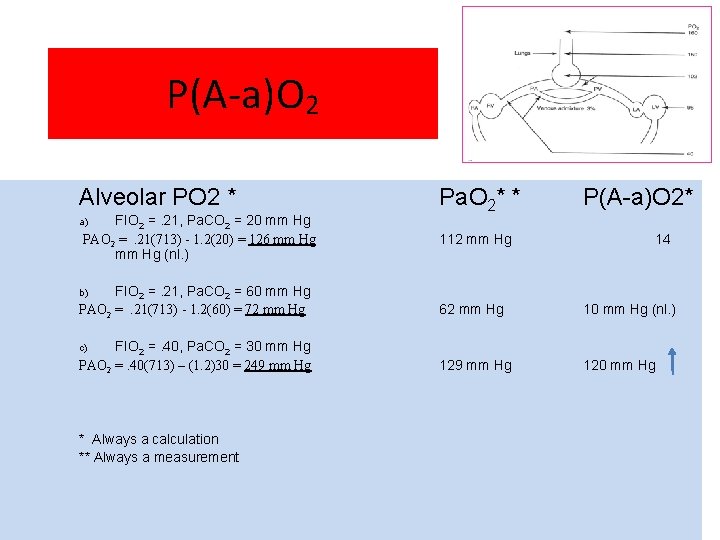
P(A-a)O 2 Alveolar PO 2 * FIO 2 =. 21, Pa. CO 2 = 20 mm Hg PAO 2 =. 21(713) - 1. 2(20) = 126 mm Hg (nl. ) Pa. O 2* * P(A-a)O 2* a) 112 mm Hg 14 FIO 2 =. 21, Pa. CO 2 = 60 mm Hg PAO 2 =. 21(713) - 1. 2(60) = 72 mm Hg 62 mm Hg 10 mm Hg (nl. ) FIO 2 =. 40, Pa. CO 2 = 30 mm Hg PAO 2 =. 40(713) – (1. 2)30 = 249 mm Hg 120 mm Hg b) c) * Always a calculation ** Always a measurement 3/1/2021 26

P(A-a)O 2: Test your understanding Calculate P(A-a)O 2 using the alveolar gas equation (assume PB = 760 mm Hg). a) A 28 yo woman with Pa. CO 2 30 mm Hg, Pa. O 2 88 mm Hg, FIO 2 0. 21. b) A 22 yo anxious man with Pa. CO 2 15 mm Hg, Pa. O 2 120 mm Hg, FIO 2 0. 21. PAO 2 =. 21 (760 - 47) - 1. 2(30) = 150 - 36 = 114 mm Hg; P(A-a)O 2 = 114 - 88 = 26 mm Hg The P(A-a)O is elevated. This means the Pa. O 2 is lower than expected for this degree of hyperventilation, indicating a gas-exchange problem. The patient was diagnosed with pulmonary embolism. PAO 2 =. 21(760 – 47) - 1. 2(15) = 150 - 18 = 132 mm Hg; P(A-a)O 2 = 132 - 120 = 12 mm Hg The patient had anxiety-hyperventilation syndrome. Hyperventilation can easily raise Pa. O 2 > 100 mm Hg when the lungs are normal, as in this case. c) A 54 yo woman with Pa. CO 2 75 mm Hg, Pa. O 2 95 mm Hg, FIO 2 0. 28. PAO 2 =. 28(760 - 47) - 1. 2(75) = 200 - 90 = 110 mm Hg; P(A-a)O 2 = 110 - 95 = 15 mm Hg Despite severe hypoventilation, there is no evidence here for lung disease. Hypercapnia is most likely a result of disease elsewhere in the respiratory system, either the central nervous system or chest bellows. 3/1/2021 27
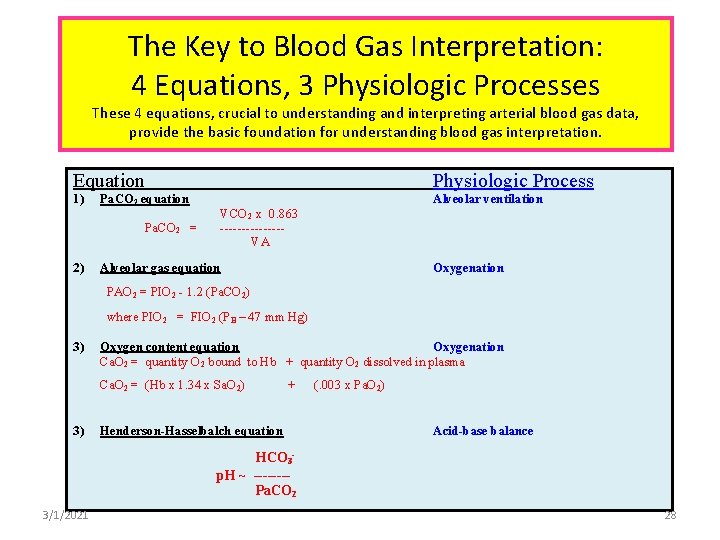
The Key to Blood Gas Interpretation: 4 Equations, 3 Physiologic Processes These 4 equations, crucial to understanding and interpreting arterial blood gas data, provide the basic foundation for understanding blood gas interpretation. Equation 1) Pa. CO 2 equation Pa. CO 2 = 2) Physiologic Process Alveolar ventilation VCO 2 x 0. 863 -------VA Alveolar gas equation Oxygenation PAO 2 = PIO 2 - 1. 2 (Pa. CO 2) where PIO 2 = FIO 2 (PB – 47 mm Hg) 3) Oxygen content equation Oxygenation Ca. O 2 = quantity O 2 bound to Hb + quantity O 2 dissolved in plasma Ca. O 2 = (Hb x 1. 34 x Sa. O 2) 3) + Henderson-Hasselbalch equation (. 003 x Pa. O 2) Acid-base balance HCO 3 p. H ~ -------Pa. CO 2 3/1/2021 28

Sa. O 2 and oxygen content • Tissues need a requisite amount of oxygen molecules for metabolism. Neither the Pa. O 2 nor the Sa. O 2 tells how much oxygen is in the blood. How much is provided by the oxygen content, Ca. O 2 (units = ml O 2/dl). Ca. O 2 is calculated as: Ca. O 2 = quantity O 2 bound to hemoglobin + quantity O 2 dissolved in plasma Ca. O 2 = (Hb x 1. 34 x Sa. O 2) + (. 003 x Pa. O 2) Ca. O 2 = 15 x 1. 34 x. 98 Ca. O 2 = 19. 7 (. 003 x 100) 0. 3 + + = 20 ml O 2/dl blood Hb = hemoglobin in gm%; 1. 34 = ml O 2 that can be bound to each gm of Hb; Sa. O 2 is percent saturation of hemoglobin with oxygen; . 003 is solubility coefficient of oxygen in plasma: . 003 ml dissolved O 2/mm Hg PO 2. 3/1/2021 29
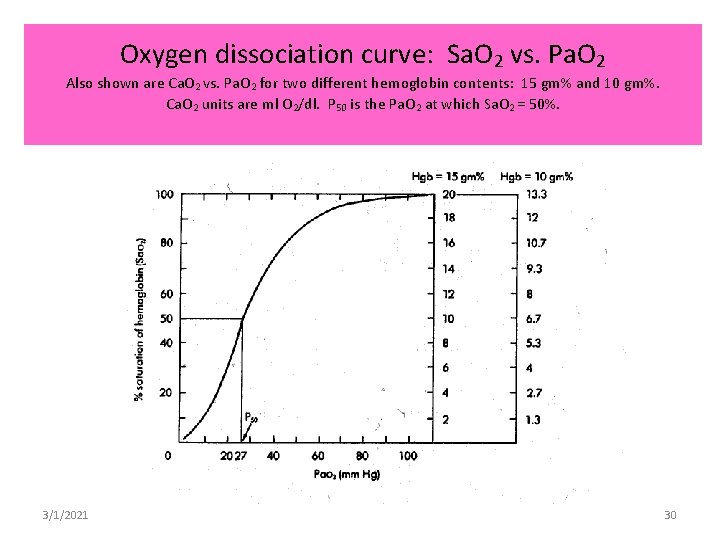
Oxygen dissociation curve: Sa. O 2 vs. Pa. O 2 Also shown are Ca. O 2 vs. Pa. O 2 for two different hemoglobin contents: 15 gm% and 10 gm%. Ca. O 2 units are ml O 2/dl. P 50 is the Pa. O 2 at which Sa. O 2 = 50%. 3/1/2021 30

How much oxygen is in the blood? Pa. O 2 vs. Sa. O 2 vs. Ca. O 2 OXYGEN PRESSURE: Pa. O 2 • Since Pa. O 2 reflects only free oxygen molecules dissolved in plasma and not those bound to hemoglobin, Pa. O 2 cannot tell us “how much” oxygen is in the blood; for that you need to know how much oxygen is also bound to hemoglobin, information given by the Sa. O 2 and hemoglobin content. OXYGEN SATURATION: Sa. O 2 • The percentage of all the available heme binding sites saturated with oxygen is the hemoglobin oxygen saturation (in arterial blood, the Sa. O 2). Note that Sa. O 2 alone doesn’t reveal how much oxygen is in the blood; for that we also need to know the hemoglobin content. OXYGEN CONTENT: Ca. O 2 • Tissues need a requisite amount of O 2 molecules for metabolism. Neither the Pa. O 2 nor the Sa. O 2 provide information on the number of oxygen molecules, i. e. , how much oxygen is in the blood. (Neither Pa. O 2 nor Sa. O 2 have units that denote any quantity. ) Only Ca. O 2 (units ml O 2/dl) tells how much oxygen is in the blood; this is because Ca. O 2 is the only value that incorporates the hemoglobin content. Oxygen content can be measured directly or calculated by the oxygen content equation: Ca. O 2 = (Hb x 1. 34 x Sa. O 2) + (. 003 x Pa. O 2) 3/1/2021 See: “The Differences Between Pa. O 2, Sa. O 2 and Oxygen Content” www. lakesidepress. com/pulmonary/ABG/PO 2. htm 31

Sa. O 2 and Ca. O 2 Which patient, (a) or (b), is more hypoxemic? (a) Hb 15 gm%, Pa. O 2 65 mm Hg, Sa. O 2=88% (b) Hb 10 gm %, Pa. O 2 100 mm Hg, Sa. O 2=98% 3/1/2021 32

Sa. O 2 and Ca. O 2 Which patient, (a) or (b), is more hypoxemic? (a) Hb 15 gm%, Pa. O 2 65 mm Hg, Sa. O 2=88% (b) Hb 10 gm %, Pa. O 2 100 mm Hg, Sa. O 2=98% ANSWER: (b) (a) Ca. O 2 =. 88 x 15 x 1. 34 = 17. 6 ml O 2/dl + dissolved O 2 (b) Ca. O 2 =. 98 x 10 x 1. 34 = 13. 1 ml O 2/dl + dissolved O 2 Oxygen content determines hypoxemia. Patient (b) has a much lower oxygen content and so is more hypoxemic than (a). Note that Pa. O 2 is not a significant factor in determining oxygen content and (for this question) can be ignored. Note that the low Pa. O 2 in patient (a) means there is an oxygen transfer problem from air to blood, but in terms of what the body really needs – OXYGEN CONTENT – patient (b) is definitely more hypoxemic. 3/1/2021 33
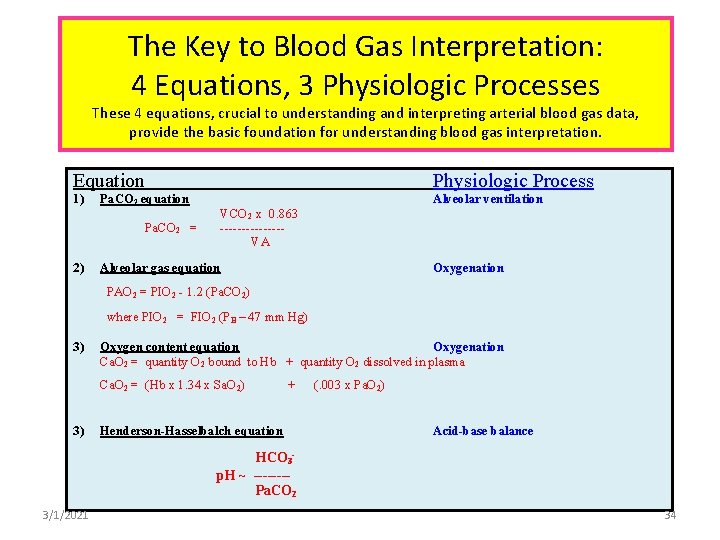
The Key to Blood Gas Interpretation: 4 Equations, 3 Physiologic Processes These 4 equations, crucial to understanding and interpreting arterial blood gas data, provide the basic foundation for understanding blood gas interpretation. Equation 1) Pa. CO 2 equation Pa. CO 2 = 2) Physiologic Process Alveolar ventilation VCO 2 x 0. 863 -------VA Alveolar gas equation Oxygenation PAO 2 = PIO 2 - 1. 2 (Pa. CO 2) where PIO 2 = FIO 2 (PB – 47 mm Hg) 3) Oxygen content equation Oxygenation Ca. O 2 = quantity O 2 bound to Hb + quantity O 2 dissolved in plasma Ca. O 2 = (Hb x 1. 34 x Sa. O 2) 3) + Henderson-Hasselbalch equation (. 003 x Pa. O 2) Acid-base balance HCO 3 p. H ~ -------Pa. CO 2 3/1/2021 34

<> ACID-BASE: traditionally the most difficult of the 3 physiologic processes • • • 3/1/2021 Acidemia: blood p. H < 7. 35 Acidosis: a primary physiologic process that, occurring alone, tends to cause acidemia, Alkalemia: blood p. H > 7. 45 Alkalosis: a primary physiologic process that, occurring alone, tends to cause alkalemia. Primary acid-base disorder: One of the four acidbase disturbances manifested by an initial change in HCO 3 - or Pa. CO 2. They are: metabolic acidosis (MAc) metabolic alkalosis (MAlk) respiratory acidosis (RAc) respiratory alkalosis (Ralk) Compensation: The change in HCO 3 - or Pa. CO 2 that results from the primary event. Compensatory changes are not classified by the terms used for the four primary acid-base disturbances. For example, a patient who hyperventilates (lowers Pa. CO 2) solely as compensation for MAc does not have a RAlk, the latter being a primary disorder that, alone, would lead to alkalemia. In simple, uncomplicated MAc the patient will never develop alkalemia. 35

Primary acid-base disorders: Respiratory alkalosis • Respiratory alkalosis - A primary disorder where the first change is a lowering of Pa. CO 2, resulting in an elevated p. H. Compensation (bringing the p. H back down toward normal) is a secondary lowering of bicarbonate (HCO 3) by the kidneys; this reduction in HCO 3 - is not metabolic acidosis, since it is not a primary process. Primary Event HCO 38 p. H ~ ))))) 9 Pa. CO 2 Compensatory Event 8 9 HCO 3 - p. H ~ ))))) 9 Pa. CO 2 RESPIRATORY ALKALOSIS 9 Pa. CO 2 & 8 p. H Hypoxemia (includes altitude) Anxiety Sepsis Any acute pulmonary insult, e. g. , pneumonia, mild asthma attack, early pulmonary edema, pulmonary embolism 3/1/2021 36

Primary acid-base disorders: Respiratory acidosis • Respiratory acidosis - A primary disorder where the first change is an elevation of Pa. CO 2, resulting in decreased p. H. Compensation (bringing p. H back up toward normal) is a secondary retention of bicarbonate by the kidneys; this elevation of HCO 3 - is not metabolic alkalosis, since it is not a primary process. Primary Event HCO 39 p. H ~ ))))) 8 Pa. CO 2 Compensatory Event HCO 39 p. H ~ ))))) 8 Pa. CO 2 8 RESPIRATORY ACIDOSIS 8 Pa. CO 2 & 9 p. H Central nervous system depression (e. g. , drug overdose) Chest bellows dysfunction (e. g. , Guillain-Barré syndrome, myasthenia gravis) Disease of lungs and/or upper airway (e. g. , chronic obstructive lung disease, severe asthma attack, severe pulmonary edema) 3/1/2021 37

Primary acid-base disorders: Metabolic acidosis • Metabolic Acidosis - A primary acid-base disorder where the first change is a lowering of HCO 3 -, resulting in decreased p. H. Compensation (bringing p. H back up toward normal) is a secondary hyperventilation; this lowering of Pa. CO 2 is not respiratory alkalosis, since it is not a primary process. Primary Event 9 p. H – Compensatory Event 9 HCO 3))))) Pa. CO 2 9 HCO 39 p. H – ))))) 9 Pa. CO 2 AG = Na+ - (Cl- + HCO 3); nl = 12 +/- 4 3/1/2021 METABOLIC ACIDOSIS 9 HCO 3 - & 9 p. H Increased anion gap (>16 m. Eq/L) lactic acidosis; ketoacidosis; drug poisonings (e. g. , aspirin, ethyelene glycol, methanol) Normal anion gap (<= 16 m. Eq/L) diarrhea; some kidney problems, e. g. , renal tubular acidosis, intersititial nephritis 38

Primary acid-base disorders: Metabolic alkalosis • Metabolic alkalosis - A primary acid-base disorder where the first change is an elevation of HCO 3 -, resulting in increased p. H. Compensation is a secondary hypoventilation (increased Pa. CO 2) which is not respiratory acidosis, since it is not a primary process. Compensation for metabolic alkalosis (attempting to bring p. H back down toward normal) is less predictable than for the other three acid-base disorders. Primary Event Compensatory Event 8 HCO 3 - 8 p. H – ))))) Pa. CO 2 8 HCO 38 p. H – ))))) 8 Pa. CO 2 METABOLIC ALKALOSIS 8 HCO 3 - & 8 p. H • Chloride responsive (responds to Na. Cl or KCl therapy): contraction alkalosis, diuretics; corticosteroids; gastric suctioning; vomiting • Chloride resistant: any hyperaldosterone state, e. g. , Cushings’s syndrome; Bartter’s syndrome; severe K + depletion 3/1/2021 39

Mixed Acid-base disorders are common • In chronically ill respiratory patients, mixed disorders are probably more common than single disorders, e. g. , RAc + MAlk, RAc + Mac, RAlk + MAlk. • In renal failure (and other conditions) combined MAlk + MAc is also encountered. p. H 7. 40, Pa. CO 2=40 mm Hg, HCO 3 - = 24 m. Eq/L, AG=24 m. Eq/L • In sepsis patients, MAc + Ralk is common. p. H 7. 40, PCO 2=20 mm Hg, HCO 3 - = 12 m. Eq/L • Always be on lookout for mixed acid-base disorders. They can be missed! 3/1/2021 40

Tips to diagnosing mixed acid-base disorders TIP 1. Don’t interpret any blood gas data for acid-base diagnosis without closely examining the venous electrolytes: Na+, K+, Cl- and HCO 3 -. • • A venous HCO 3 - out of the normal range always represents some type of acid-base disorder (barring lab or transcription error). High venous HCO 3 - indicates metabolic alkalosis &/or bicarbonate retention as compensation for respiratory acidosis Low venous HCO 3 - indicates metabolic acidosis &/or bicarbonate excretion as compensation for respiratory alkalosis Note that venous HCO 3 - may be normal in the presence of two or more acid-base disorders. 3/1/2021 41

Tips to diagnosing mixed acid-base disorders (cont. ) TIP 2. Single acid-base disorders do not lead to normal blood p. H. Although p. H can end up in the normal range (7. 35 - 7. 45) with a mild single disorder, a truly normal p. H with distinctly abnormal HCO 3 - and Pa. CO 2 invariably suggests two or more primary disorders. • Example: p. H 7. 40, Pa. CO 2 20 mm Hg, HCO 3 - 12 m. Eq/L, in a patient with sepsis. Normal p. H results from two co-existing and unstable acid-base disorders: acute respiratory alkalosis and metabolic acidosis. 3/1/2021 42

Tips to diagnosing mixed acid-base disorders (cont. ) TIP 3. Simplified rules predict the p. H and HCO 3 - for a given change in Pa. CO 2. If the p. H or HCO 3 - is higher or lower than expected for the change in Pa. CO 2, the patient probably has a metabolic acid-base disorder as well. Below are expected changes in p. H and HCO 3 - (in m. Eq/L) for a 10 mm Hg change in Pa. CO 2 resulting from either primary hypoventilation (respiratory acidosis) or primary hyperventilation (respiratory alkalosis). • • ACUTE CHRONIC Resp Acidosis – p. H 9 by 0. 07 p. H 9 by 0. 03 – HCO 3 8 by 1*HCO 3 8 by 3 -4 Resp Alkalosis – p. H 8 by 0. 08 p. H 8 by 0. 03 – HCO 3 - 9 by 2 HCO 3 - 9 by 5 *Units for HCO 3 - are m. Eq/L 3/1/2021 43

Predicted changes in HCO 3 - for a directional change in Pa. CO 2 can help uncover mixed acid-base disorders. 3/1/2021 a) A normal or slightly low HCO 3 - in the presence of hypercapnia suggests a concomitant metabolic acidosis, e. g. , p. H 7. 27, Pa. CO 2 50 mm Hg, HCO 3 - 22 m. Eq/L. Based on the rule for increase in HCO 3 - with hypercapnia, it should be at least 25 m. Eq/L in this example; that it is only 22 m. Eq/L suggests a concomitant metabolic acidosis. b) A normal or slightly elevated HCO 3 - in the presence of hypocapnia suggests a concomitant metabolic alkalosis, e. g. , p. H 7. 56, Pa. CO 2 30 mm Hg, HCO 3 - 26 m. Eq/L. Based on the rule for decrease in HCO 3 with hypocapnia, it should be at least 23 m. Eq/L in this example; that it is 26 m. Eq/L suggests a concomitant metabolic alkalosis. 44
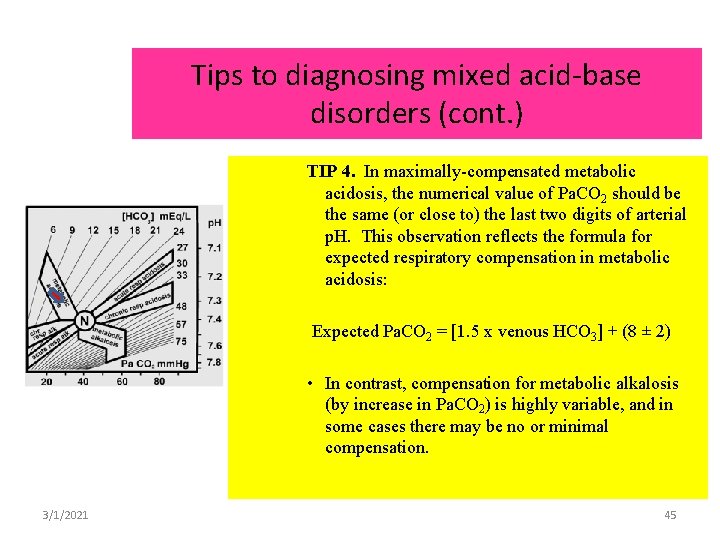
Tips to diagnosing mixed acid-base disorders (cont. ) TIP 4. In maximally-compensated metabolic acidosis, the numerical value of Pa. CO 2 should be the same (or close to) the last two digits of arterial p. H. This observation reflects the formula for expected respiratory compensation in metabolic acidosis: Expected Pa. CO 2 = [1. 5 x venous HCO 3] + (8 ± 2) • In contrast, compensation for metabolic alkalosis (by increase in Pa. CO 2) is highly variable, and in some cases there may be no or minimal compensation. 3/1/2021 45

Acid-base disorders: test your understanding A patient’s arterial blood gas shows p. H of 7. 14, Pa. CO 2 of 70 mm Hg, and HCO 3 - of 23 m. Eq/L. How would you describe the likely acid-base disorder(s)? Acute elevation of Pa. CO 2 leads to reduced p. H, i. e. , an acute respiratory acidosis. However, is the problem only acute respiratory acidosis or is there some additional process? For every 10 mm Hg rise in Pa. CO 2 (before any renal compensation), p. H falls about 0. 07 units. Because this patient's p. H is down 0. 26, or 0. 05 more than expected for a 30 mm Hg increase in Pa. CO 2, there must be an additional, metabolic problem. Also, note that with acute CO 2 retention of this degree, the HCO 3 - should be elevated 3 m. Eq/L. Thus a low-normal HCO 3 - with increased Pa. CO 2 is another way to uncover an additional, metabolic disorder. Decreased perfusion leading to mild lactic acidosis would explain the metabolic component. 3/1/2021 46

Acid-base disorders: test your understanding A 45 -year-old man comes to hospital complaining of dyspnea for three days. Arterial blood gas reveals p. H 7. 35, Pa. CO 2 60 mm Hg, Pa. O 2 57 mm Hg, HCO 3 - 31 m. Eq/L. How would you characterize his acid-base status? Pa. CO 2 and HCO 3 - are elevated, but HCO 3 - is elevated more than would be expected from acute respiratory acidosis. Since the patient has been dyspneic for several days it is fair to assume a chronic acid-base disorder. Most likely this patient has a chronic or compensated respiratory acidosis. Without electrolyte data and more history, you cannot diagnose an accompanying metabolic disorder. 3/1/2021 47

Acid-base disorders: test your understanding State whether each of the following statements is true or false. a) Metabolic acidosis is always present when the measured serum CO 2 changes acutely from 24 to 21 m. Eq/L. b) In acute respiratory acidosis, bicarbonate initially rises because of the reaction of CO 2 with water and the resultant formation of H 2 CO 3. c) If p. H and Pa. CO 2 are both above normal, the calculated bicarbonate must also be above normal. d) An abnormal serum CO 2 value always indicates an acid-base disorder of some type. e) The compensation for chronic elevation of Pa. CO 2 is renal excretion of bicarbonate. f) A normal p. H with abnormal HCO 3 - or Pa. CO 2 suggests the presence of two or more acid-base disorders. g) A normal venous HCO 3 - value indicates there is no acid-base disorder. h) Normal arterial blood gas values rule out the presence of an acid-base disorder. 3/1/2021 48

Acid-base disorders: test your understanding State whether each of the following statements is true or false. a) Metabolic acidosis is always present when the measured serum CO 2 changes acutely from 24 to 21 m. Eq/L. b) In acute respiratory acidosis, bicarbonate initially rises because of the reaction of CO 2 with water and the resultant formation of H 2 CO 3. c) If p. H and Pa. CO 2 are both above normal, the calculated bicarbonate must also be above normal. d) An abnormal serum CO 2 value always indicates an acid-base disorder of some type. a) false b) true c) true d) true e) false e) The compensation for chronic elevation of Pa. CO 2 is renal excretion of bicarbonate. f) true f) A normal p. H with abnormal HCO 3 - or Pa. CO 2 suggests the presence of two or more acid-base disorders. g) false g) A normal venous HCO 3 - value indicates there is no acid-base disorder. h) false h) Normal arterial blood gas values rule out the presence of an acid-base disorder. 3/1/2021 49

Summary ) Clinical and laboratory approach to acid-base diagnosis • Determine existence of acid-base disorder from ABG and/or venous electrolytes. • Examine p. H, Pa. CO 2 and HCO 3 - for obvious primary acid-base disorder, and for deviations that indicate mixed acid-base disorders (TIPS 2 through 4). • Use a full clinical assessment (hx, phys exam, other lab data, previous ABGs) to explain each acid-base disorder. Co-existing clinical conditions may lead to opposing acid-disorders, so that p. H can be high when there is an obvious acidosis, or low when there is an obvious alkalosis. • Treat the underlying clinical condition(s); this will usually suffice to correct most acid-base disorders. Clinical judgment should always apply. 3/1/2021 50
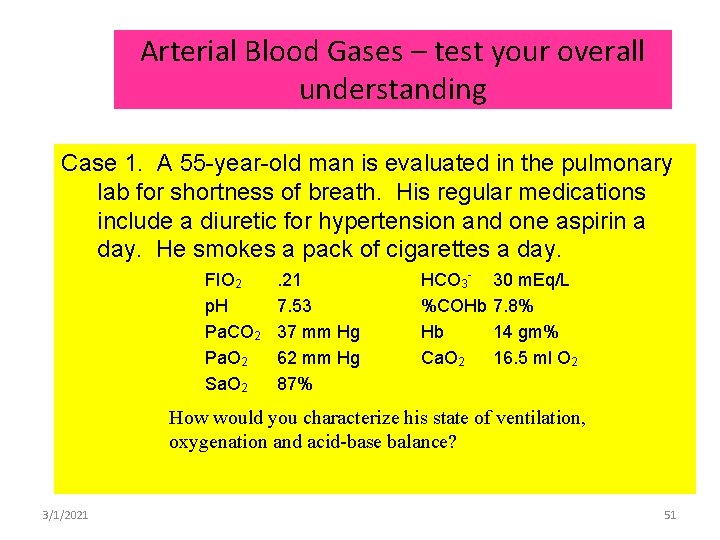
Arterial Blood Gases – test your overall understanding Case 1. A 55 -year-old man is evaluated in the pulmonary lab for shortness of breath. His regular medications include a diuretic for hypertension and one aspirin a day. He smokes a pack of cigarettes a day. FIO 2 p. H Pa. CO 2 Pa. O 2 Sa. O 2 . 21 7. 53 37 mm Hg 62 mm Hg 87% HCO 3%COHb Hb Ca. O 2 30 m. Eq/L 7. 8% 14 gm% 16. 5 ml O 2 How would you characterize his state of ventilation, oxygenation and acid-base balance? 3/1/2021 51
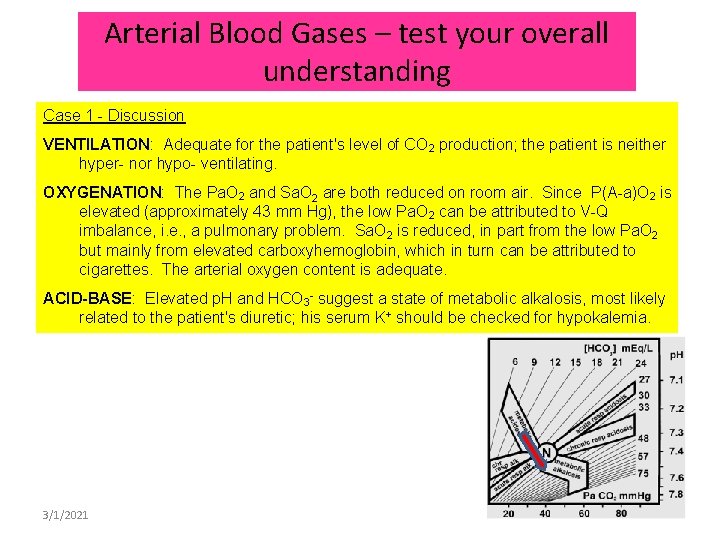
Arterial Blood Gases – test your overall understanding Case 1 - Discussion VENTILATION: Adequate for the patient's level of CO 2 production; the patient is neither hyper- nor hypo- ventilating. OXYGENATION: The Pa. O 2 and Sa. O 2 are both reduced on room air. Since P(A-a)O 2 is elevated (approximately 43 mm Hg), the low Pa. O 2 can be attributed to V-Q imbalance, i. e. , a pulmonary problem. Sa. O 2 is reduced, in part from the low Pa. O 2 but mainly from elevated carboxyhemoglobin, which in turn can be attributed to cigarettes. The arterial oxygen content is adequate. ACID-BASE: Elevated p. H and HCO 3 - suggest a state of metabolic alkalosis, most likely related to the patient's diuretic; his serum K+ should be checked for hypokalemia. 3/1/2021 52
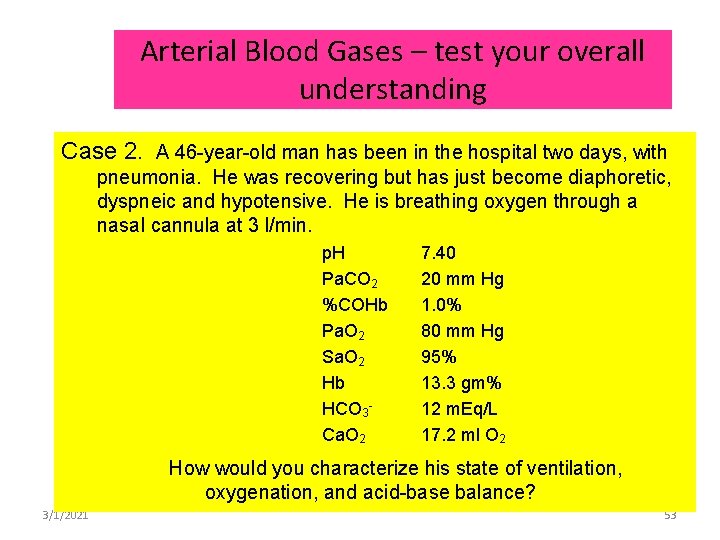
Arterial Blood Gases – test your overall understanding Case 2. A 46 -year-old man has been in the hospital two days, with pneumonia. He was recovering but has just become diaphoretic, dyspneic and hypotensive. He is breathing oxygen through a nasal cannula at 3 l/min. p. H Pa. CO 2 %COHb Pa. O 2 Sa. O 2 Hb HCO 3 Ca. O 2 7. 40 20 mm Hg 1. 0% 80 mm Hg 95% 13. 3 gm% 12 m. Eq/L 17. 2 ml O 2 How would you characterize his state of ventilation, oxygenation, and acid-base balance? 3/1/2021 53
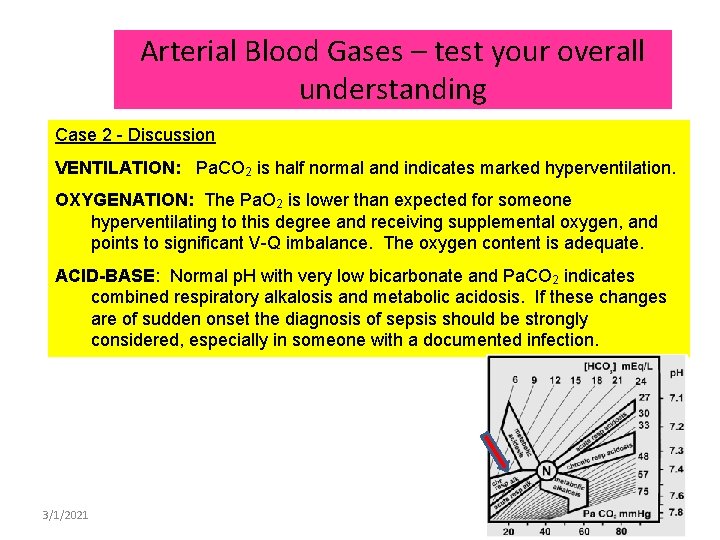
Arterial Blood Gases – test your overall understanding Case 2 - Discussion VENTILATION: Pa. CO 2 is half normal and indicates marked hyperventilation. OXYGENATION: The Pa. O 2 is lower than expected for someone hyperventilating to this degree and receiving supplemental oxygen, and points to significant V-Q imbalance. The oxygen content is adequate. ACID-BASE: Normal p. H with very low bicarbonate and Pa. CO 2 indicates combined respiratory alkalosis and metabolic acidosis. If these changes are of sudden onset the diagnosis of sepsis should be strongly considered, especially in someone with a documented infection. 3/1/2021 54

Arterial Blood Gases – test your overall understanding Case 3. A 58 -year-old woman is being evaluated in the emergency department for acute dyspnea. FIO 2 p. H Pa. CO 2 %COHb Pa. O 2 Sa. O 2 Hb HCO 3 Ca. O 2 . 21 7. 19 65 mm Hg 1. 1% 45 mm Hg 90% 15. 1 gm% 24 m. Eq/L 18. 3 ml O 2 How would you characterize her state of ventilation, oxygenation and acid-base balance? 3/1/2021 55

Arterial Blood Gases – test your overall understanding Case 3 - Discussion VENTILATION: The patient is hypoventilating. OXYGENATION: The patient's Pa. O 2 is reduced for two reasons: hypercapnia and V-Q imbalance, the latter apparent from an elevated P(A-a)O 2 (approximately 27 mm Hg). ACID-BASE: p. H and Pa. CO 2 are suggestive of acute respiratory acidosis plus metabolic acidosis; the calculated HCO 3 - is lower than expected from acute respiratory acidosis alone. . 3/1/2021 56
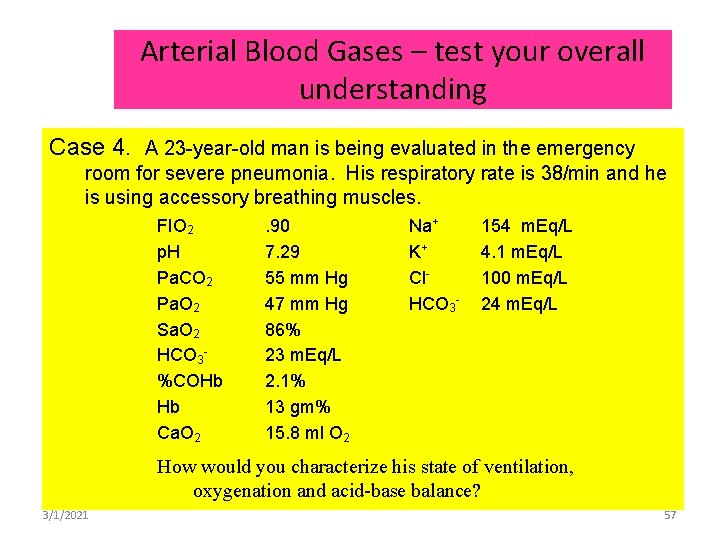
Arterial Blood Gases – test your overall understanding Case 4. A 23 -year-old man is being evaluated in the emergency room for severe pneumonia. His respiratory rate is 38/min and he is using accessory breathing muscles. FIO 2 p. H Pa. CO 2 Pa. O 2 Sa. O 2 HCO 3%COHb Hb Ca. O 2 . 90 7. 29 55 mm Hg 47 mm Hg 86% 23 m. Eq/L 2. 1% 13 gm% 15. 8 ml O 2 Na+ K+ Cl. HCO 3 - 154 m. Eq/L 4. 1 m. Eq/L 100 m. Eq/L 24 m. Eq/L How would you characterize his state of ventilation, oxygenation and acid-base balance? 3/1/2021 57

Arterial Blood Gases – test your overall understanding Case 4 - Discussion VENTILATION: The patient is hypoventilating despite the presence of tachypnea, indicating significant dead space ventilation. This is a dangerous situation that suggests the need for mechanical ventilation. OXYGENATION: The Pa. O 2 and Sa. O 2 are both markedly reduced on 90% inspired oxygen, indicating severe ventilation-perfusion imbalance. ACID-BASE: The low p. H (7. 29), high Pa. CO 2 (55) and normal HCO 3 - all point to combined acute respiratory acidosis and metabolic acidosis. Anion gap is elevated to 30 m. Eq/L indicating a clinically significant anion gap (AG) acidosis, possibly from lactic acidosis. AG = Na - (Cl + HCO 3 -) = 154 - (100 + 24) = 30 m. Eq/L With an of AG of 30 m. Eq/L his venous HCO 3 - should be much lower, to reflect buffering of the increased acid. However, his venous HCO 3 - is normal, indicating a primary process that is increasing it, i. e. , a metabolic alkalosis in addition to a metabolic acidosis. The cause of the alkalosis is as yet undetermined. In summary this patient has respiratory acidosis, metabolic acidosis and metabolic alkalosis. 3/1/2021 58

Blood Gas Interpretation means analyzing the data to determine patient’s state of: Ventilation Oxygenation Acid-Base Oh, did I mention you can’t learn this from a lecture? 3/1/2021 59

The best way to learn ABGs is to work on blood gas problems with some knowledge of basic physiology, then check your work for instant feedback. This ‘iterative process’ will teach you blood gas interpretation. Books Web sites You Can’t Learn Arterial Blood Gases from a Lecture You CAN learn ABGs from selected web sites. See list at: www. lakesidepress. com/pulmonary/AB G/PREFACEnew. htm 3/1/2021 60
- Slides: 60