Yorkshire Humber Academic Health Science Network Hypo project

Yorkshire & Humber Academic Health Science Network – Hypo project 22 CCGs Mike Stansfield, NHS Outcomes Manager Sanofi Prescribing Information can be found at the end of this presentation GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015

Analysis of Diabetes Activity and Spend- overview GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015

Data Disclaimer ● ● The information in this presentation has been reproduced accurately without amendment or alteration, except to produce visual representations in order to improve clarity and understanding. Every effort has been taken to ensure the information provided is up to date and accurate and complete, but no warranties or representations are given in this regard. GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
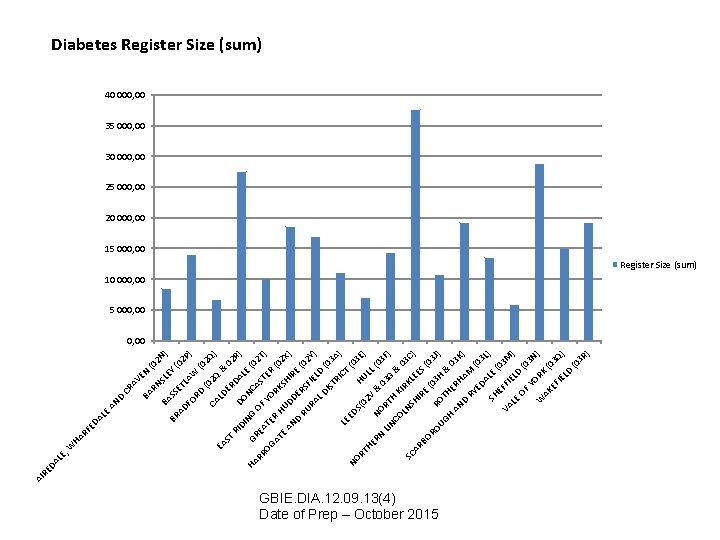
LE , LE HA RF ED A W BA N EY SL RN AV E CR BA D AN 2 N (0 BR SS ) ( E 0 AD T 2 FO LAW P) RD (0 2 ( CA 02 Q Q) EA LD & ST ER 02 D RI DO AL R) DI NG NC E ( HA G AS 02 T RR REA OF OG YO TER ) T RK AT ER H (0 2 S E AN UDD HIR X) E D E (0 RU RS 2 Y F RA IE ) L D LD ( 03 IS TR A) I C LE T ED (0 NO 3 S( H RT 02 UL E) HE V L (0 & NO RN LIN RT 03 G 3 F) & CO H K 03 IR LN K SC C) L S E H AR ES I R BO E (0 (0 RO 3 J 3 R ) H UG OT & HE H RH 03 K AN AM ) D RY (0 ED AL 3 L) SH E (0 E 3 M VA FFI E ) LE LD OF (0 3 N Y ) W ORK AK ( 0 EF 3 Q IE LD ) (0 3 R ) DA RE AI Diabetes Register Size (sum) 40 000, 00 35 000, 00 30 000, 00 25 000, 00 20 000, 00 15 000, 00 Register Size (sum) 10 000, 00 5 000, 00 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
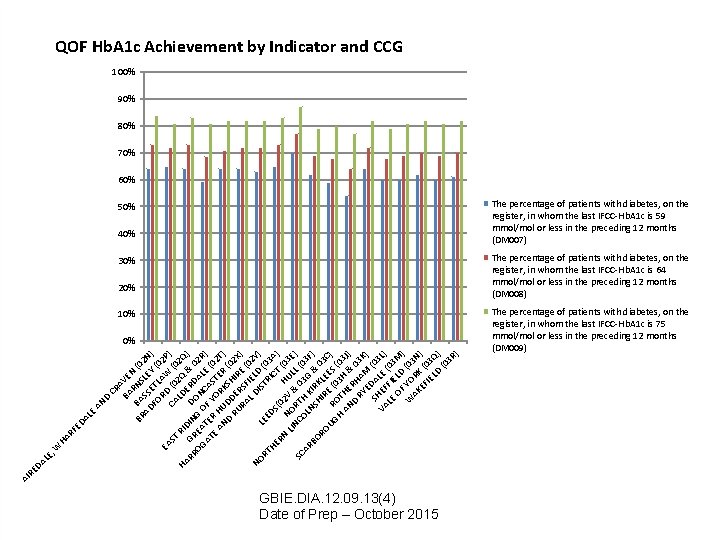
QOF Hb. A 1 c Achievement by Indicator and CCG 100% 90% 80% 70% 60% The percentage of patients with diabetes, on the register, in whom the last IFCC-Hb. A 1 c is 59 mmol/mol or less in the preceding 12 months (DM 007) 50% 40% The percentage of patients with diabetes, on the register, in whom the last IFCC-Hb. A 1 c is 64 mmol/mol or less in the preceding 12 months (DM 008) 30% 20% The percentage of patients with diabetes, on the register, in whom the last IFCC-Hb. A 1 c is 75 mmol/mol or less in the preceding 12 months (DM 009) 10% AI RE DA LE , W HA RF ED A LE AN D CR A BA VE N R BR BA NS (0 AD SS LE 2 N FO ETL Y (0 ) RD AW 2 P EA CA (0 (0 ) ST LD 2 Q 2 Q RI HA RR GR DIN D ERD & 0 ) OG EA G O ON AL 2 R AT TE F CA E ( ) E R H YO STE 02 AN U RK R T) D DD SH (0 RU E IR 2 X RA RSF E ( ) L D IEL 02 NO IS D ( Y) L EE TR 03 RT D IC A HE S ( RN 02 H T (0 ) N LIN O V & UL 3 E SC CO RTH 03 L (0 ) AR LN K G 3 F) BO SH IRK & 0 RO IR LE 3 UG R E ( ES C) H OT 03 H (03 AN HE & J) D RH 03 RY AM K ) E SH DAL (03 VA EF E L) LE FIE (03 OF LD M ) W YO (03 AK RK N EF (0 ) IE 3 Q LD ) (0 3 R ) 0% GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
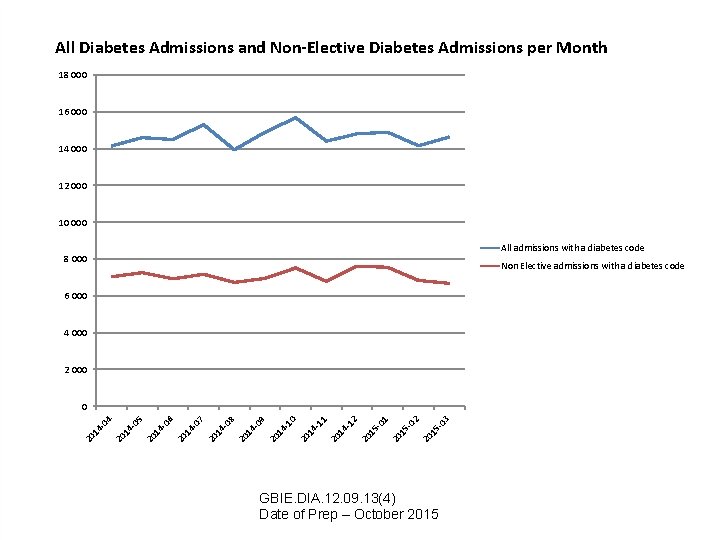
All Diabetes Admissions and Non-Elective Diabetes Admissions per Month 18 000 16 000 14 000 12 000 10 000 All admissions with a diabetes code 8 000 Non Elective admissions with a diabetes code 6 000 4 000 2 000 503 20 1 502 20 1 501 20 1 412 20 1 411 20 1 410 20 1 409 20 1 408 20 1 407 20 1 406 20 1 405 20 1 404 0 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
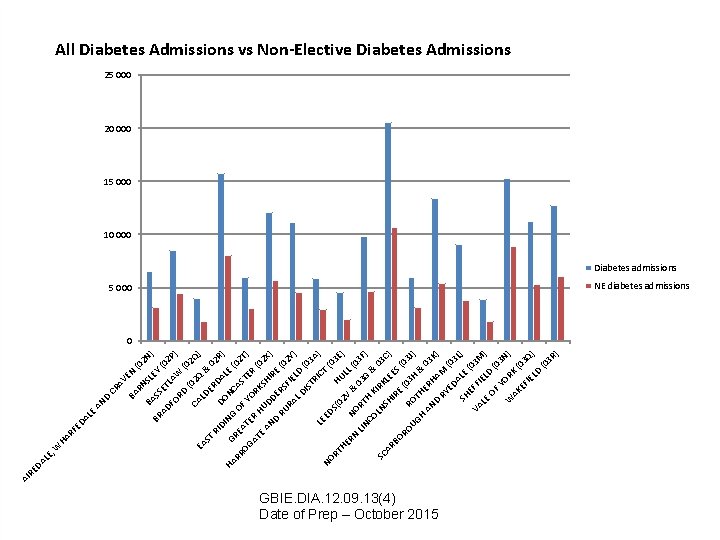
LE , LE HA RF ED A W BA N EY SL RN AV E CR BA D AN 2 N (0 AD BR SS ) (0 ET FO LAW 2 P) RD (0 (02 Q C 2 A EA LD Q & ) ST ER 02 RI R) DO DA DI L N HA E N GR G RR OF CAS (02 E OG AT YO TER T) AT ER RK (0 H E AN UD SHI 2 X) D RU ERS E (0 2 Y F RA IE L D LD ) ( IS TR 03 A I LE CT ) NO ED (0 S( RT 02 HU 3 E) HE LL RN NO V & (0 0 LIN RT 3 G 3 F) CO H K & IR 03 LN SC K S L AR HI EE C) RE BO S( 03 (0 RO R 3 J) UG OT H HE & H AN RH 03 K D AM ) RY (0 ED 3 A SH LE L) (0 3 M VA EFF I E LE ) LD OF (0 3 Y W ORK N) AK EF (03 Q IE LD ) (0 3 R ) DA RE AI All Diabetes Admissions vs Non-Elective Diabetes Admissions 25 000 20 000 15 000 10 000 Diabetes admissions 5 000 NE diabetes admissions 0 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
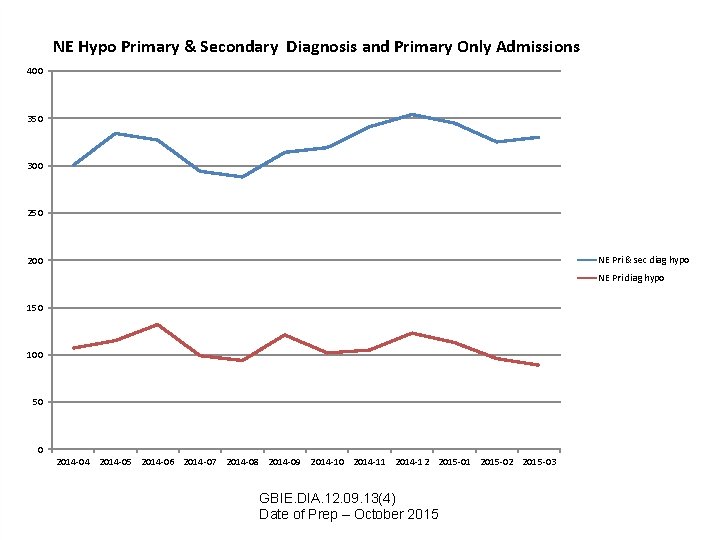
NE Hypo Primary & Secondary Diagnosis and Primary Only Admissions 400 350 300 250 NE Pri & sec diag hypo 200 NE Pri diag hypo 150 100 50 0 2014 -04 2014 -05 2014 -06 2014 -07 2014 -08 2014 -09 2014 -10 2014 -11 2014 -12 2015 -01 2015 -02 2015 -03 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
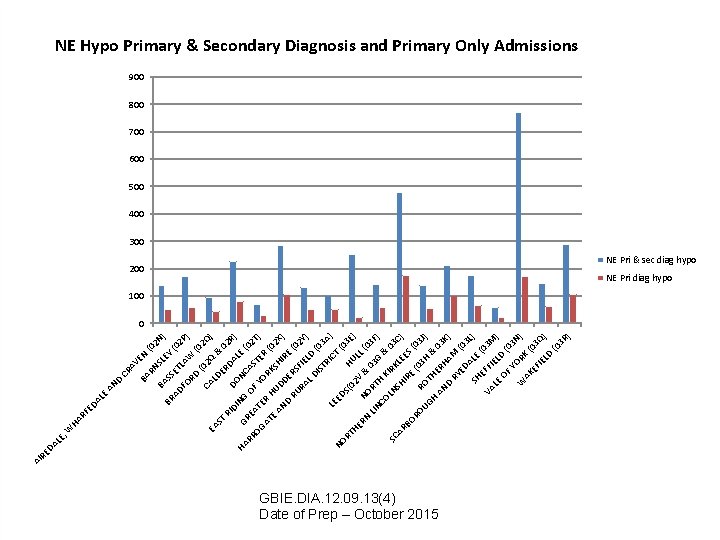
BA SL RN EN CR AV BA D AN (0 2 N AD BR SS EY ) (0 ET FO LAW 2 P) RD (0 (02 Q C 2 A EA LD Q & ) ST ER 02 RI R) DO DA DI L N HA E N GR G RR OF CAS (02 E OG AT YO TER T) AT ER RK (0 H E AN UD SHI 2 X) D RU ERS E (0 2 Y F RA IE L D LD ) ( IS TR 03 A I LE CT ) NO ED (0 S( RT 02 HU 3 E) HE LL RN NO V & (0 0 LIN RT 3 G 3 F) CO H K & IR 03 LN SC K S L AR HI EE C) RE BO S( 03 RO (0 R 3 J) UG OT H & HE H AN RH 03 K D AM ) RY (0 ED 3 A SH LE L) (0 3 VA EFF LE IEL M) OF D ( 03 Y W ORK N) AK EF (03 Q IE LD ) (0 3 R ) AL E HA RF ED , W LE DA RE AI NE Hypo Primary & Secondary Diagnosis and Primary Only Admissions 900 800 700 600 500 400 300 200 NE Pri & sec diag hypo NE Pri diag hypo 100 0 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
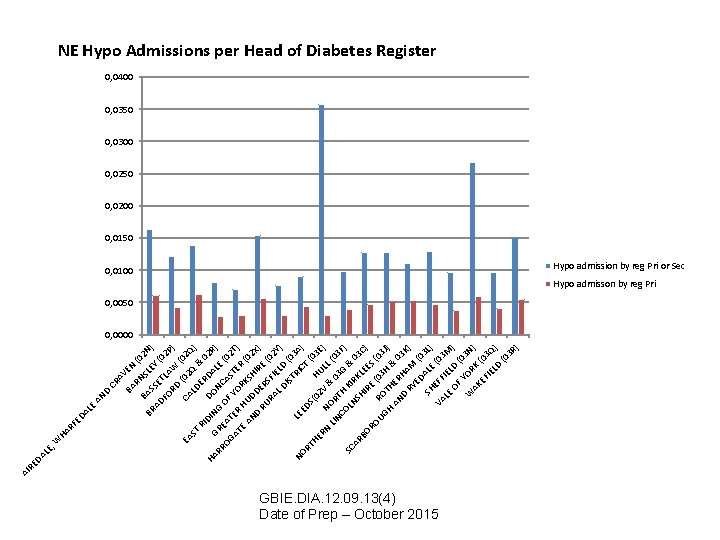
D AN CR A BA VEN RN (0 2 BR BAS SLE N) Y S AD E ( FO TLA 02 P ) RD W ( CA (02 02 Q EA ST LD Q & ) ER RI 0 D HA IN DO DAL 2 R) G NC E RR RE G AS (02 OG AT OF AT ER YO TER T) R E H AN UD KSH (02 X D D I RU ER RE ( ) 02 RA SFI L D ELD Y) IS TR (03 A LE NO IC E T ) DS RT (0 (0 HE 2 V HUL 3 E) RN N L LIN OR & 0 (0 CO TH 3 G 3 F) SC LN KIR & 0 AR SH KL 3 BO IR EE C) S E RO UG RO (03 J) H T H AN HER & 0 3 H D RY AM K) ED (0 SH ALE 3 L) VA EFF (03 LE IEL M ) OF D (0 Y 3 O W N) AK RK EF (03 IE LD Q) (0 3 R ) AL E HA RF ED , W LE DA RE AI NE Hypo Admissions per Head of Diabetes Register 0, 0400 0, 0350 0, 0300 0, 0250 0, 0200 0, 0150 0, 0100 Hypo admission by reg Pri or Sec Hypo admisson by reg Pri 0, 0050 0, 0000 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
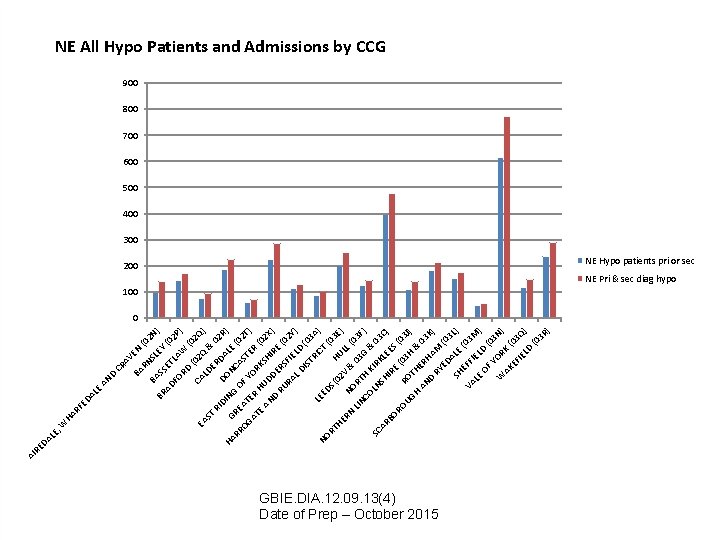
LE , LE HA RF ED A W BA N EY SL RN AV E CR BA D AN AD BR 2 N (0 SS ) (0 ET FO LA 2 P) RD W (0 CA (02 Q 2 Q EA ) LD ST & E 0 RD RI 2 R D D A ) HA GR ING ONC LE ( RR 0 O AS F OG EAT T 2 T AT ER YOR ER ) ( H K E AN UD SH 02 X IR ) D D RU ERS E (0 2 Y RA FIE L D LD ) ( IS TR 03 A L I CT ) EE NO DS ( RT (0 H 03 HE 2 V UL E) RN L( N & LIN ORT 03 03 F CO H K G & ) LN IR SC 0 SH KLE 3 C AR IR BO ES ) E (0 RO ( UG RO 03 H 3 J) TH & H 0 AN ER HA 3 K D M ) RY (0 ED 3 SH ALE L) VA EFF (03 LE IEL M) OF D ( 03 Y W OR N) AK K ( 0 EF IE 3 Q) LD (0 3 R ) DA RE AI NE All Hypo Patients and Admissions by CCG 900 800 700 600 500 400 300 200 NE Hypo patients pri or sec 100 NE Pri & sec diag hypo 0 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
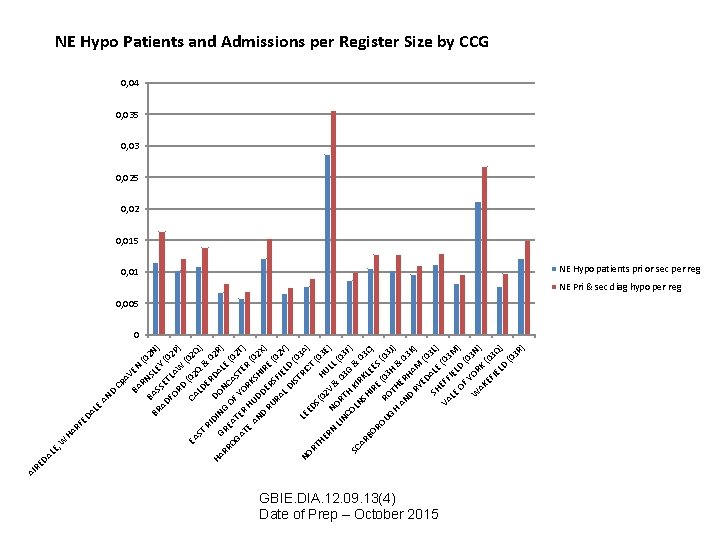
LE , LE HA RF ED A W D AN CR A BA VEN RN (0 2 BR BAS SLE N) AD SE Y ( FO TLA 02 P ) RD W (0 ( 0 CA 2 EA 2 ST LD Q & Q) E RI RD 02 DI HA NG DON AL R) G RR RE E OG AT OF CAS (02 T T AT ER YO R ER ) E H AN UD KSH (02 X D D I RU ER RE ( ) S 02 RA FI L D ELD Y) IS TR (03 A LE NO IC ED T ) RT (0 S (0 HE H 2 V UL 3 E) RN N L LIN OR & 0 (0 T 3 CO H G 3 F) SC KI & L N AR SH RKL 03 C BO IR EE ) S E RO UG RO (03 J) H T H AN HER & 0 3 H D RY AM K) ED (0 SH ALE 3 L) VA EFF (03 LE IEL M ) OF D (0 Y 3 W OR N) AK K EF (03 IE LD Q) (0 3 R ) DA RE AI NE Hypo Patients and Admissions per Register Size by CCG 0, 04 0, 035 0, 03 0, 025 0, 02 0, 015 0, 01 NE Hypo patients pri or sec per reg NE Pri & sec diag hypo per reg 0, 005 0 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
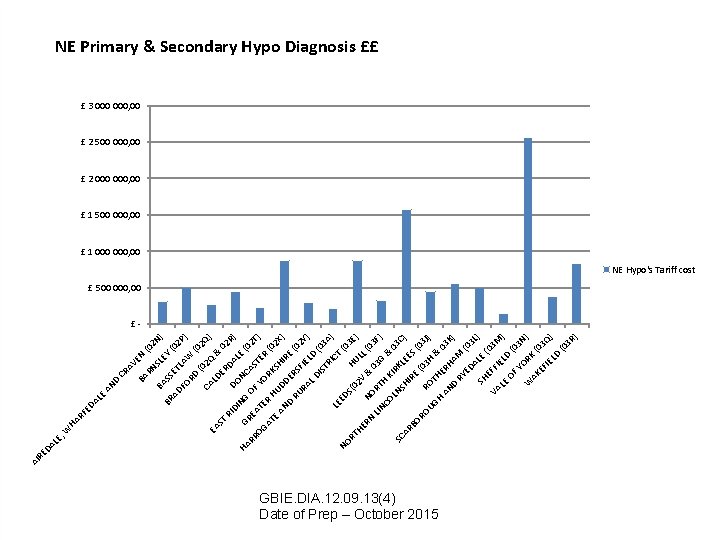
LE , LE HA RF ED A W BA N EY SL RN AV E CR BA D AN 2 N (0 AD BR SS ) (0 ET 2 FO LAW P) RD (0 ( 2 CA 02 Q Q) EA LD & ST ER 02 RI D DO AL R) DI NG E N HA G ( OF CAS 02 T RR RE T OG ATE YO ER ) R AT R (0 K H 2 E AN UD SHIR X) D D E (0 RU ERS 2 Y F RA IE L D LD ) (0 IS 3 A TR ) IC LE T ED NO (0 3 E S( RT 02 HU HE LL ) V RN NO & (0 LIN RT 03 G 3 F) CO H K & IR 03 LN SC K C SH LE AR ES ) IR BO E (0 (0 RO 3 J ) UG ROT 3 H HE & 0 H AN RH 3 D AM K) RY (0 ED AL 3 L) SH E ( E VA FFI 03 M EL LE ) OF D (0 3 N Y ) W ORK AK ( 0 EF 3 Q IE LD ) (0 3 R ) DA RE AI NE Primary & Secondary Hypo Diagnosis ££ £ 3 000, 00 £ 2 500 000, 00 £ 2 000, 00 £ 1 500 000, 00 £ 1 000, 00 NE Hypo's Tariff cost £ 500 000, 00 £- GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
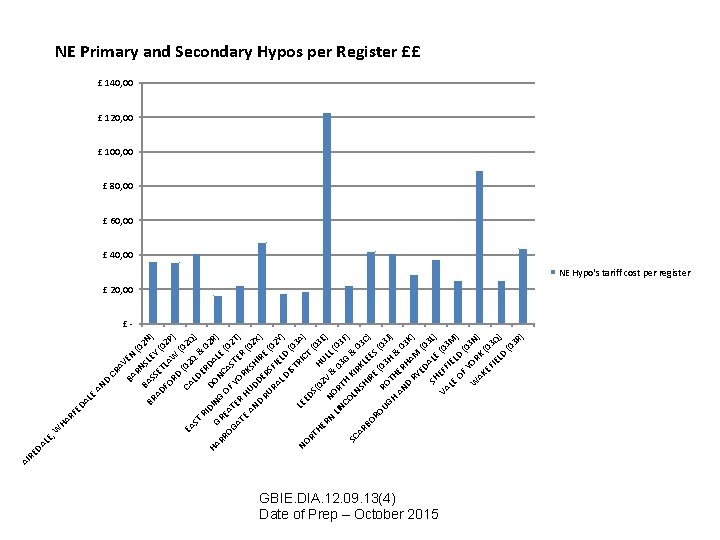
D AN CR AV BA EN R (0 2 B N BR AS SLE N) Y AD SET ( FO LA 02 P ) RD W (0 ( 0 2 C EA AL 2 Q Q) ST DE & RI RD 02 D D HA R IN A O RR GRE G O NC LE ( ) AS 02 OG AT F AT ER YO TER T) R H E AN UD KSH (02 DE IR X) D RU RS E ( RA FIE 02 L D LD Y) IS TR (03 A LE NO IC ED T ) RT (0 S (0 H HE 2 V UL 3 E) RN N L LIN OR & 0 (03 3 T F) G CO H SC LN KIR & 0 AR SH KL 3 BO IR EE C) S E RO ( UG RO (03 H 03 J ) T H AN HER & 0 3 K H D RY AM ) ED (0 SH ALE 3 L) VA EFF (03 LE IEL M ) OF D YO (03 N W ) AK RK EF (03 IE LD Q) (0 3 R ) AL E HA RF ED , W LE DA RE AI NE Primary and Secondary Hypos per Register ££ £ 140, 00 £ 120, 00 £ 100, 00 £ 80, 00 £ 60, 00 £ 40, 00 NE Hypo's tariff cost per register £ 20, 00 £- GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
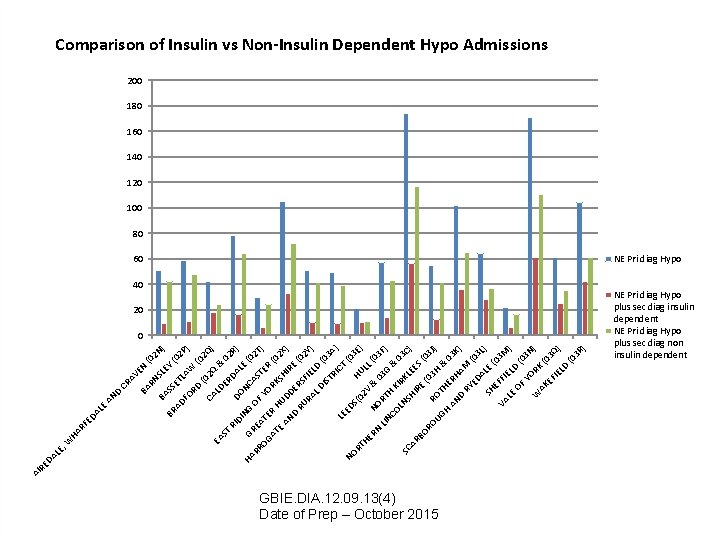
LE , SS BA N EY SL RN AV E CR BA D AN AD BR LE HA RF ED A W (0 ) 2 N (0 FO ET LA 2 P) RD W ( 02 ( CA 02 Q Q) EA LD & ST ER 02 RI DO DAL R) DI NG NC E ( HA G AS 02 T RR REA OF TE ) OG Y T AT ER H ORK R (0 2 S E AN UDD HIR X) E D E (0 RU RS 2 Y F RA IE ) L D LD ( 03 IS TR A) I C LE T E (0 NO DS HU 3 E) (0 RT 2 V LL HE (0 & NO RN 0 3 G 3 F) LIN RT CO H K & 03 IR LN K SC C) S L E HI AR ES R BO E (0 (0 RO 3 J R 3 ) H UG OT & HE H AN RH 03 K AM ) D RY (0 ED AL 3 L) SH E (0 E 3 VA FFI EL M) LE D OF (0 YO 3 N ) W R AK K ( 0 EF 3 Q IE LD ) (0 3 R ) DA RE AI Comparison of Insulin vs Non-Insulin Dependent Hypo Admissions 200 180 160 140 120 100 80 60 NE Pri diag Hypo 40 20 0 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015 NE Pri diag Hypo plus sec diag insulin dependent NE Pri diag Hypo plus sec diag non insulin dependent
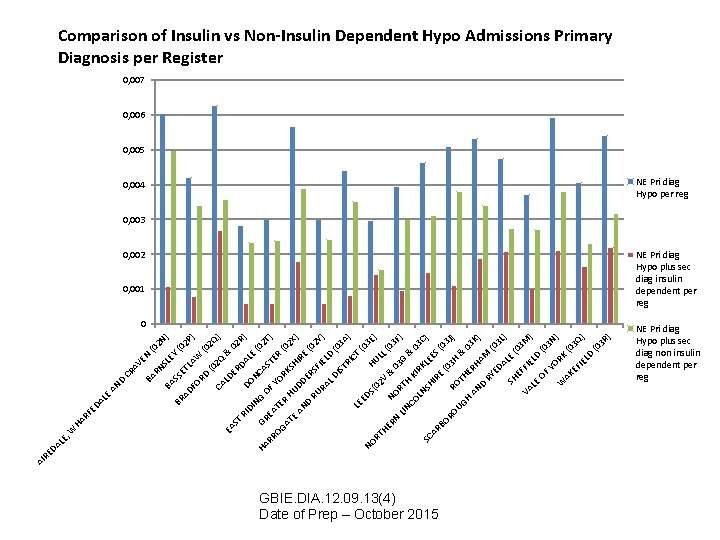
LE , CA 2 Q (0 EY N W ET LA RD FO SS BA SL RN AV E CR BA D AN AD BR LE HA RF ED A W 2 Q ) (0 ) 2 P ) (0 2 N (0 ST EA LD & 0 ER DA 2 R) RI D DI L NG ONC E (0 HA GR 2 T AS EA OF RR ) T E YO TE OG R ( RK R 02 AT HU S X) E DD HIR AN E E D RU RSF (02 Y) IE RA L D LD (0 IS 3 A TR ) IC T LE ( E 0 DS NO HU 3 E) (0 RT 2 V LL HE (0 & NO RN 3 F 03 ) LIN RT G H & CO KI 0 R 3 C LN ) SC SH KLE ES AR IR E (0 BO ( 3 J RO ) RO 03 H UG & TH 0 H 3 K AN ERH AM ) D RY (0 ED 3 L AL ) E SH (0 EF 3 M FI VA EL ) LE D OF (0 YO 3 N ) R W AK K (0 3 Q EF IE LD ) (0 3 R ) DA RE AI Comparison of Insulin vs Non-Insulin Dependent Hypo Admissions Primary Diagnosis per Register 0, 007 0, 006 0, 005 0, 004 NE Pri diag Hypo per reg 0, 003 0, 002 0, 001 NE Pri diag Hypo plus sec diag insulin dependent per reg 0 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015 NE Pri diag Hypo plus sec diag non insulin dependent per reg
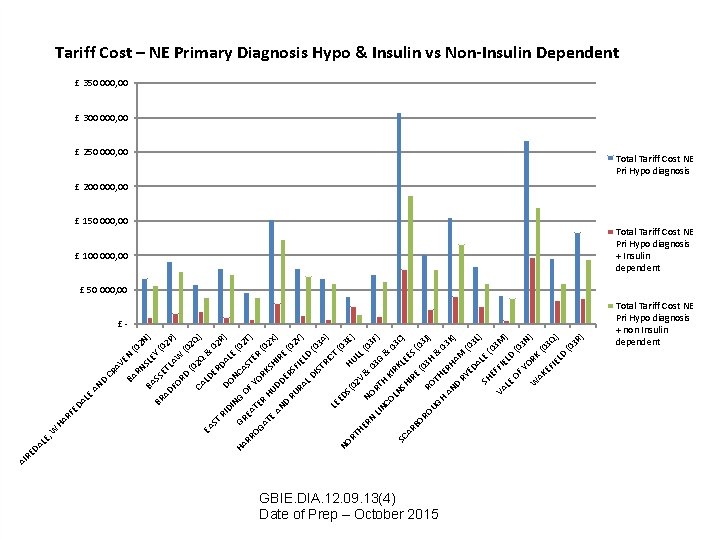
LE , LE HA RF ED A W N EY SL RN AV E CR BA D AN 2 N (0 BR SS BA ) ( 0 E AD 2 T FO LAW P) RD (0 2 ( CA 02 Q Q) LD EA & 0 ER ST DA 2 R) RI D DI NG ONC LE ( HA GR AS 02 T OF RR E YO TER ) OG ATE R (0 RK AT HU 2 X SH E ) DD AN IR E ER D ( 0 RU S 2 Y RA FIEL ) D LD ( 03 IS TR A) I C LE T (0 ED NO 3 S( RT 02 HUL E) HE V L( & NO 03 RN 0 F) 3 G LIN RT & CO H K 03 IR LN C) SC SH KLE AR ES IR BO E (0 ( RO RO 03 H 3 J) UG TH & H 03 AN ERH K AM ) D RY (0 ED AL 3 L) E SH (0 E 3 M VA FFI EL ) LE D OF (0 YO 3 N ) R W AK K (0 EF 3 Q IE LD ) (0 3 R ) DA RE AI Tariff Cost – NE Primary Diagnosis Hypo & Insulin vs Non-Insulin Dependent £ 350 000, 00 £ 300 000, 00 £ 250 000, 00 Total Tariff Cost NE Pri Hypo diagnosis £ 200 000, 00 £ 150 000, 00 £ 100 000, 00 Total Tariff Cost NE Pri Hypo diagnosis + Insulin dependent £ 50 000, 00 £- GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015 Total Tariff Cost NE Pri Hypo diagnosis + non Insulin dependent
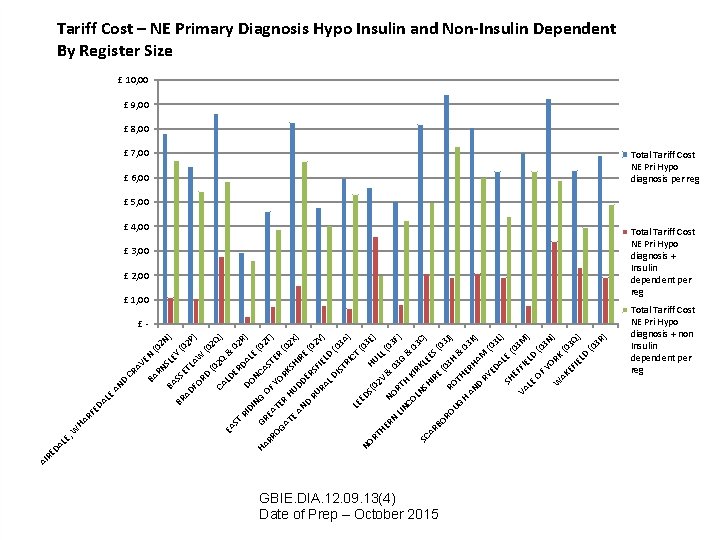
Tariff Cost – NE Primary Diagnosis Hypo Insulin and Non-Insulin Dependent By Register Size £ 10, 00 £ 9, 00 £ 8, 00 £ 7, 00 Total Tariff Cost NE Pri Hypo diagnosis per reg £ 6, 00 £ 5, 00 £ 4, 00 Total Tariff Cost NE Pri Hypo diagnosis + Insulin dependent per reg £ 3, 00 £ 2, 00 £ 1, 00 CA RD FO AD BR (0 2 Q (0 W ET LA SS BA 2 Q ) LD EA & 0 E ST 2 R R RI ) DO DAL DI E N N ( G CA HA 0 G ST 2 T) RR REA OF E YO OG TE R (0 R RK AT H 2 X S E ) AN UDD HIR E ER D ( 02 RU S Y) RA FIEL D LD (0 IS 3 A TR ) IC LE T ( E 0 DS NO HU 3 E) (0 RT 2 LL HE V (0 & NO RN 3 F 03 R ) LIN G TH & CO KI 03 R LN C) SC SH KLE AR ES IR E BO (0 ( 3 J RO RO 03 H ) UG TH & H 03 AN ERH K AM ) D RY (0 ED AL 3 L) E SH (0 EF 3 M FI VA E ) LE LD OF (0 YO 3 N ) R W AK K (0 EF 3 Q IE LD ) (0 3 R ) ) 2 P (0 EY SL RN BA AI RE DA LE , W HA RF ED A LE AN D CR AV E N (0 2 N ) £- GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015 Total Tariff Cost NE Pri Hypo diagnosis + non Insulin dependent per reg
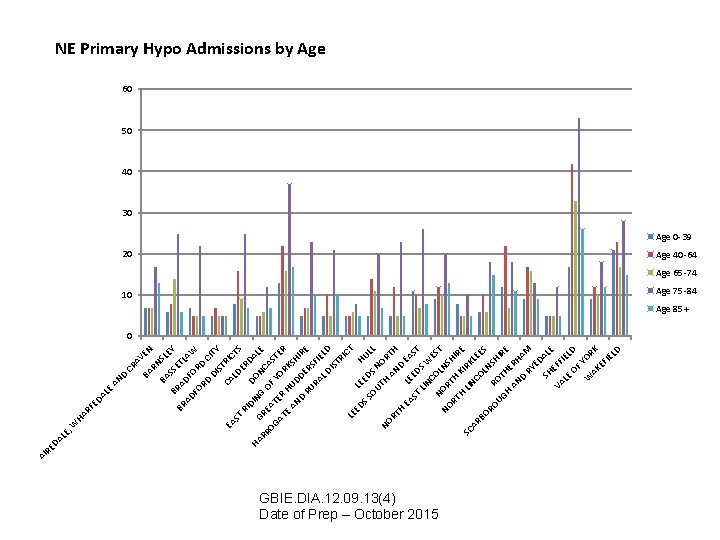
LE , LE HA RF ED A W D AN CR A BA VEN RN BA SL SS EY B BR RA ETL A AD DF FO OR W RD D C DI ITY S CA TRIC EA LD T ST ER S RI DI DO DA HA N L RR GRE G O NC E A OG AT F S AT ER YOR TER H E AN UD KSH IR D D E RU ER S RA FI L D ELD IS TR IC T LE ED LE H S S ED U OU S N LL TH OR NO AN TH RT D H EA LE EA ST ED ST LIN S W CO ES N T NO OR LNS H T RT H IR K E H SC LIN IRK L AR CO EE BO LN S RO S R UG OT HIR H E H AN ERH A D RY M ED SH ALE VA EFF LE IEL OF D W YOR AK K EF IE LD DA RE AI NE Primary Hypo Admissions by Age 60 50 40 30 Age 0 -39 20 Age 40 -64 Age 65 -74 10 Age 75 -84 Age 85+ 0 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
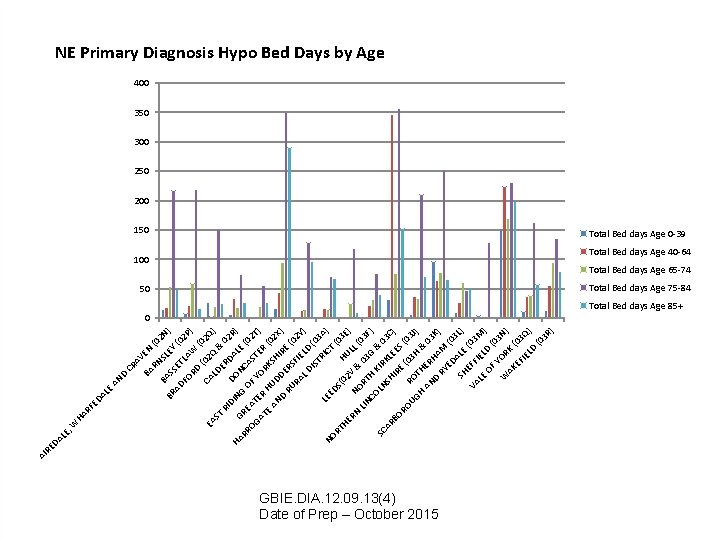
LE , LE HA RF ED A W BA N EY SL RN AV E CR BA D AN 2 N (0 AD BR SS ) ET (0 FO LA 2 P ) RD W (0 ( 2 Q CA 02 EA LD Q & ) ST ER RI DA 02 R D D I O HA NG G NC LE ) RR RE (0 O A A OG TE F Y STE 2 T) OR AT R R H ( E AN UD KSH 02 X DE IR ) D RU RS E (0 RA FIE 2 L D LD Y) ( IS TR 03 A L I E C NO ED T ) (0 RT S( 02 HU 3 E HE ) V L RN L( N & 03 LIN OR 0 3 G F) CO TH K SC LN IR & 0 3 C AR SH KL BO IR EES ) E RO (0 ( UG RO 03 H 3 J) T H & H AN ER 03 H K D RY AM ) ED (0 3 SH ALE L) VA EFF (03 LE IEL M) OF D ( 03 Y W OR N) AK K EF (03 Q IE LD ) (0 3 R ) DA RE AI NE Primary Diagnosis Hypo Bed Days by Age 400 350 300 250 200 150 Total Bed days Age 0 -39 100 Total Bed days Age 40 -64 Total Bed days Age 65 -74 50 Total Bed days Age 75 -84 0 Total Bed days Age 85+ GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
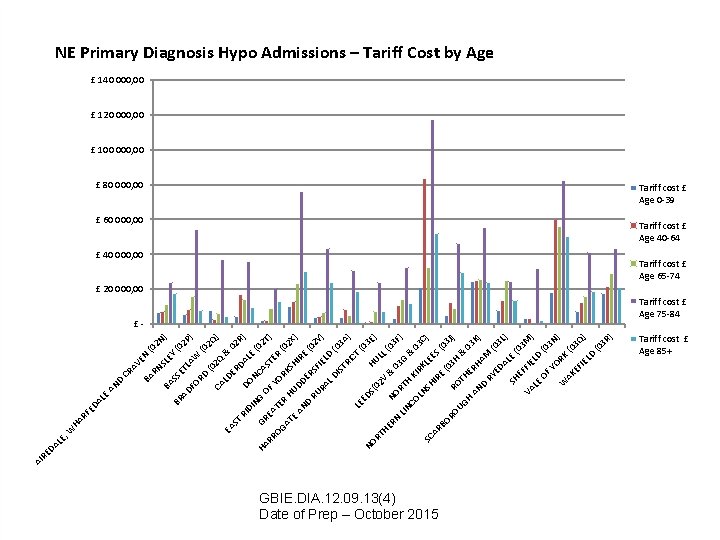
LE , LE HA RF ED A W N EY SL RN AV E CR BA D AN 2 N (0 BA ) SS BR ( 02 AD ETL AW P) FO RD (0 2 Q Q) CA LD & EA 0 ER ST DA 2 R) RI D LE DI ON NG (0 CA GR HA ST 2 T) EA OF RR ER YO TE OG (0 RK R AT 2 X HU SH E ) DD IR AN E ER D ( 0 RU S 2 Y ) RA FIEL D LD ( 03 IS TR A) IC T LE (0 ED NO S( HU 3 E) 02 RT LL V HE (0 & N RN 3 F O 0 ) LIN RTH 3 G & CO KI 03 RK LN C) LE SC SH E AR IR S( E BO 03 (0 J) RO 3 H RO UG & TH 03 H AN ERH K AM ) D RY (0 ED 3 L AL ) E SH ( 0 E VA FFIE 3 M) LD LE OF (0 YO 3 N) R W AK K (0 3 Q EF IE LD ) (0 3 R ) DA RE AI NE Primary Diagnosis Hypo Admissions – Tariff Cost by Age £ 140 000, 00 £ 120 000, 00 £ 100 000, 00 £ 80 000, 00 Tariff cost £ Age 0 -39 £ 60 000, 00 Tariff cost £ Age 40 -64 £ 40 000, 00 Tariff cost £ Age 65 -74 £ 20 000, 00 £Tariff cost £ Age 75 -84 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015 Tariff cost £ Age 85+
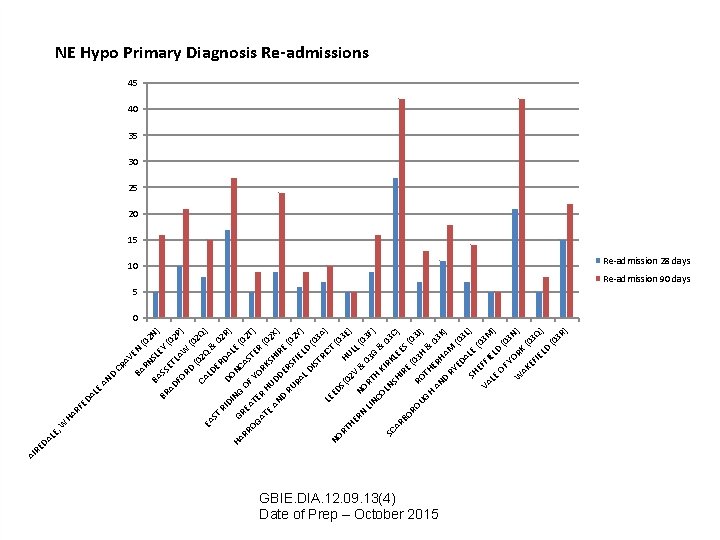
LE , LE HA RF ED A W BA N EY SL RN AV E CR BA D AN 2 N (0 AD BR SS ) (0 ET FO LAW 2 P) RD (0 ( CA 02 Q 2 Q) EA LD ST ER & 0 RI DA 2 R D DI ) O N HA NC LE ( GR G 0 AS RR OF 2 E OG AT YO TER T) E R AT RK (0 H E AN UD SHI 2 X) RE D D (0 RU ERS 2 Y F RA IE L LD D ) ( IS TR 03 A I LE CT ) ED NO (0 S( RT 02 HU 3 E) HE LL RN NO V & (0 0 LIN RT 3 G 3 F) CO H K & IR 03 LN SC KL S AR HI EE C) RE BO S( 03 (0 RO R 3 J) UG OT H HE & 0 H AN RH 3 D AM K) RY (0 ED AL 3 L) SH E ( E VA FFI 03 M E LE ) L OF D (0 3 Y W ORK N) AK EF (03 Q IE LD ) (0 3 R ) DA RE AI NE Hypo Primary Diagnosis Re-admissions 45 40 35 30 25 20 15 10 Re-admission 28 days 5 Re-admission 90 days 0 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
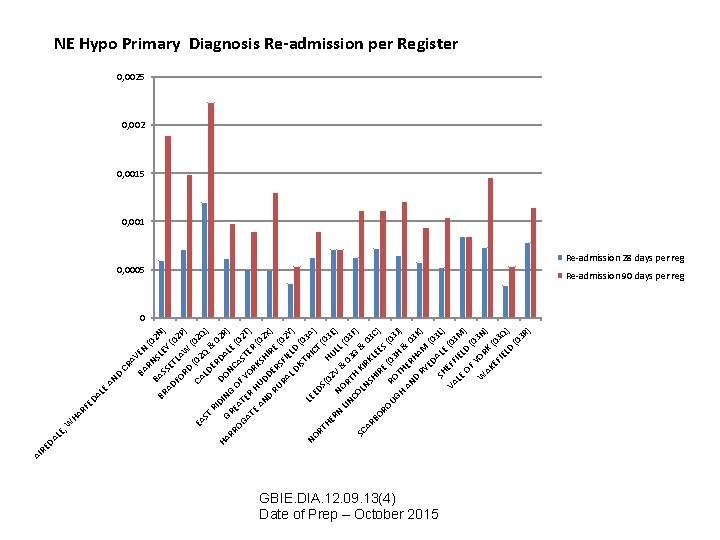
D AN CR A BA VEN RN (0 2 BR BAS SLE N) Y S AD E ( FO TLA 02 P ) RD W ( CA (02 02 Q EA ST LD Q & ) ER RI 0 D HA IN DO DAL 2 R) G NC E RR RE G AS (02 OG AT OF AT ER YO TER T) R H E AN UD KSH (02 X D D I RU ER RE ( ) 02 RA SFI L D ELD Y) IS TR (03 A LE NO IC E T ) DS RT (0 (0 HE 2 V HUL 3 E) RN N L LIN OR & 0 (0 CO TH 3 G 3 F) SC LN KIR & 0 AR SH KL 3 BO IR EE C) S E RO UG RO (03 J) H T H AN HER & 0 3 H D RY AM K) ED (0 SH ALE 3 L) VA EFF (03 LE IEL M ) OF D (0 Y 3 O W N) AK RK EF (03 IE LD Q) (0 3 R ) AL E HA RF ED , W LE DA RE AI NE Hypo Primary Diagnosis Re-admission per Register 0, 0025 0, 002 0, 0015 0, 001 0, 0005 Re-admission 28 days per reg Re-admission 90 days per reg 0 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
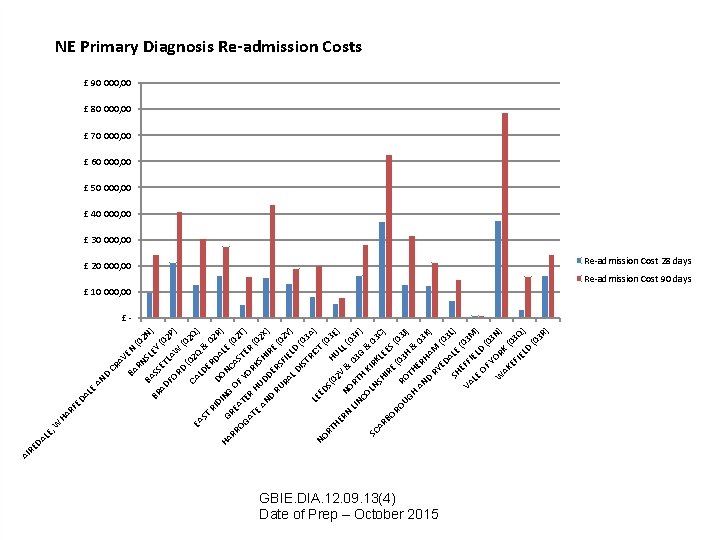
LE , LE HA RF ED A W BA N EY SL RN AV E CR BA D AN 2 N (0 AD BR SS ) (0 ET 2 FO LA P) RD W (0 CA (02 Q 2 Q EA ) LD & ST E 0 R RI DO DA 2 R) D LE HA N GR ING RR OF CAS (02 E A OG T YO TER T) AT ER RK (0 H E 2 X AN UD SH I DE RE ) D RU RS ( RA FIE 02 Y L D LD ) ( IS TR 03 A IC LE ) NO T ED ( 0 S( RT 02 HU 3 E) HE RN N V & LL ( LIN ORT 03 03 F CO H K G & ) IR LN 0 SC SH KLE 3 C) AR I E BO RE S( RO (0 03 R 3 J) UG O H T HE & H 0 AN RH 3 K D RY AM ) (0 ED 3 SH ALE L) VA EFF (03 M LE IEL ) OF D ( 0 3 Y W OR N) AK K ( 0 EF IE 3 Q) LD (0 3 R ) DA RE AI NE Primary Diagnosis Re-admission Costs £ 90 000, 00 £ 80 000, 00 £ 70 000, 00 £ 60 000, 00 £ 50 000, 00 £ 40 000, 00 £ 30 000, 00 £ 20 000, 00 Re-admission Cost 28 days £ 10 000, 00 Re-admission Cost 90 days £- GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
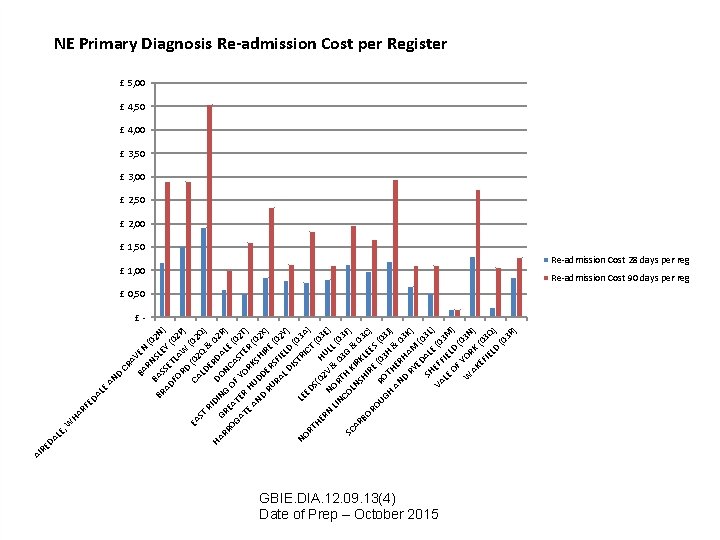
LE , LE HA RF ED A W D AN CR A BA VEN RN (0 BR BAS SLE 2 N AD SE Y ( ) FO TLA 02 RD W P) EA CA (02 ST LD Q Q) RI ER & D HA IN DO DA 02 R G RR R G L ) N OG EA OF CA E (0 T S 2 AT ER YO TE T R ) E AN HUD RKS (0 2 X D D H RU ER IRE ) RA SFI (02 L D ELD Y) IS TR (03 LE NO IC A) E RT DS T (0 HE (0 H 3 2 RN NO V & ULL E) LIN R (0 CO TH 03 G 3 F SC ) K LN IR & 0 AR S K 3 H BO IR LEE C) RO E S UG RO (03 H J) T H AN HER & 0 H 3 D RY AM K) ED (0 SH ALE 3 L) E VA FF (0 LE IE 3 M ) OF LD (0 W YOR 3 N AK K ) EF (03 IE LD Q) (0 3 R ) DA RE AI NE Primary Diagnosis Re-admission Cost per Register £ 5, 00 £ 4, 50 £ 4, 00 £ 3, 50 £ 3, 00 £ 2, 50 £ 2, 00 £ 1, 50 £ 1, 00 Re-admission Cost 28 days per reg Re-admission Cost 90 days per reg £ 0, 50 £- GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
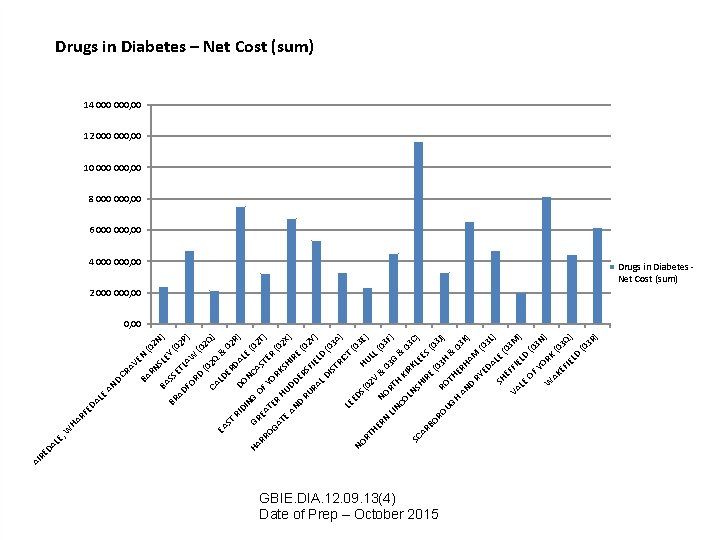
LE , CA N (0 2 Q W EY SL ET LA RD FO SS BA RN AV E CR BA D AN AD BR LE HA RF ED A W ) ) 2 Q ) (0 2 P (0 2 N (0 ST EA LD & 02 ER D RI AL R) DO DI NG NC E (0 HA GR A 2 T O RR E F Y STE ) OG ATE R OR R ( AT 0 KS H 2 X E ) AN UDD HIR E ER D ( 02 RU S Y) RA FIEL D LD (0 IS 3 A TR ) I C LE T ( E 0 NO DS HU 3 E) (0 RT 2 LL HE V (0 & NO RN 3 F 0 3 R ) LIN G TH & CO KI 0 R LN 3 C SC ) SH KLE AR E IR S( BO E 0 (0 RO 3 H 3 J) R UG OT & H H 03 AN ERH K AM ) D RY (0 ED AL 3 L) E SH (0 EF 3 M VA FI E ) LE LD OF (0 YO 3 N ) R W AK K (0 EF 3 Q IE LD ) (0 3 R ) DA RE AI Drugs in Diabetes – Net Cost (sum) 14 000, 00 12 000, 00 10 000, 00 8 000, 00 6 000, 00 4 000, 00 Drugs in Diabetes Net Cost (sum) 2 000, 00 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
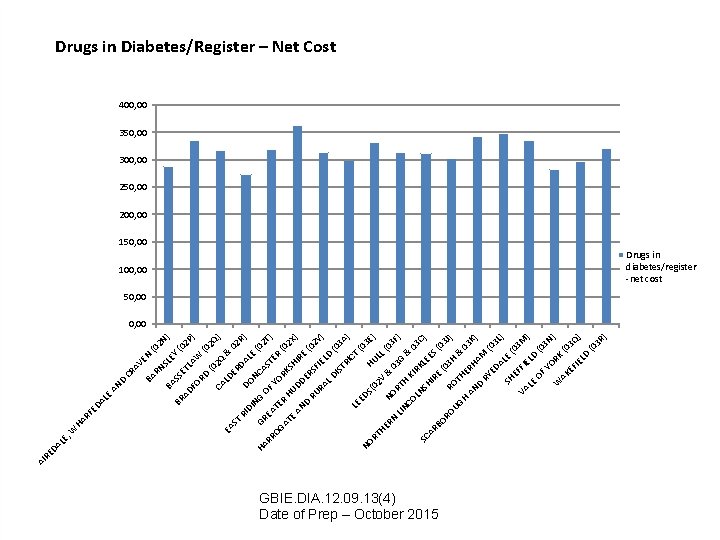
EY ) (0 2 N FO (0 ET LA 2 P) RD W ( 02 (0 2 Q Q) CA LD & EA 0 ER ST DA 2 R) RI D DI L NG ONC E (0 HA GR A ST 2 T) RR EA OF Y OG TE OR ER ( R 02 AT KS H X) E AN UDD HIR E ER D (0 RU S 2 Y RA FIEL ) D LD ( 03 IS TR A I CT ) LE (0 ED NO 3 S( RT 02 HUL E) L( HE V & 03 NO RN 0 F) 3 G LIN RT H & CO KI 03 R LN C) SC SH KLE ES AR IR E BO (0 ( 3 J RO RO 03 H ) UG TH & H 03 AN ERH K AM ) D RY (0 ED AL 3 L) E SH (0 EF 3 M FI VA E ) LE LD OF (0 YO 3 N ) R W AK K (0 3 Q EF IE LD ) (0 3 R ) SS BA SL RN EN CR AV BA D AN AD BR AL E HA RF ED , W LE DA RE AI Drugs in Diabetes/Register – Net Cost 400, 00 350, 00 300, 00 250, 00 200, 00 150, 00 100, 00 Drugs in diabetes/register -net cost 50, 00 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
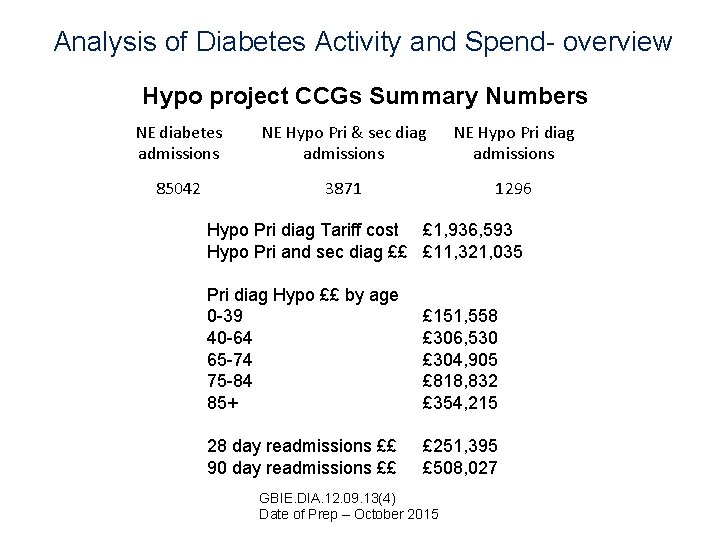
Analysis of Diabetes Activity and Spend- overview Hypo project CCGs Summary Numbers NE diabetes admissions NE Hypo Pri & sec diag admissions NE Hypo Pri diag admissions 85042 3871 1296 Hypo Pri diag Tariff cost £ 1, 936, 593 Hypo Pri and sec diag ££ £ 11, 321, 035 Pri diag Hypo ££ by age 0 -39 40 -64 65 -74 75 -84 85+ £ 151, 558 £ 306, 530 £ 304, 905 £ 818, 832 £ 354, 215 28 day readmissions ££ 90 day readmissions ££ £ 251, 395 £ 508, 027 GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015

Analysis of Diabetes Activity and Spend- overview ● Thank You GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015

Lantus Prescribing Information Lantus® (insulin glargine). Please refer to Summary of Product Characteristics prior to use of Lantus cartridges and Solostar prefilled pens each contain 300 Units of insulin glargine in 3 ml, equivalent to 10. 92 mg. Lantus vials contain 1000 Units insulin glargine in 10 ml, equivalent to 36. 4 mg. Indications: Treatment of diabetes mellitus in adults, adolescents and children of 2 years or above. Dosage and administration: Lantus is administered subcutaneously once daily, at any time but at the same time each day. Do not administer intravenously. Insulin glargine dosage should be individually adjusted. In type 2 diabetes mellitus, Lantus can also be used in combination with orally active antidiabetic medicinal products. Close metabolic monitoring is recommended during, and for a period after, transition from other insulins to Lantus. Dose and timing of other antidiabetic medicines may need to be adjusted. Dose adjustments may also be required if the patient’s weight or lifestyle changes, the timing of insulin dose is changed or other circumstances arise that increase susceptibility to hypo- or hyperglycaemia. Lantus must not be mixed with other insulins or diluted. Insulin requirements may be diminished in the elderly or patients with renal or hepatic impairment. The safety and efficacy of Lantus has not been established in children below 2 years of age. No data are available. Contraindications: Hypersensitivity to insulin glargine or any excipients. Precautions and warnings: Switch from twice daily NPH: To reduce the risk of nocturnal and early morning hypoglycaemia, patients who are changing to a once daily regimen with Lantus should reduce their daily dose of basal insulin by 20 -30% during the first weeks of treatment. Switch from Toujeo (insulin glargine 300 units/ml): Lantus and Toujeo are not bioequivalent and are not directly interchangeable. To reduce the risk of hypoglycaemia, patients changing from once daily insulin glargine 300 units/ml to once daily Lantus should reduce their dose by approximately 20%. Close metabolic monitoring is recommended during the switch and in the initial weeks thereafter. Lantus is not the insulin of choice for treatment of diabetic ketoacidosis. In case of insufficient glucose control or a tendency to hypo/hyperglycaemic episodes all relevant factors must be reviewed before dose adjustment is considered. Particular caution should be exercised, and intensified blood monitoring is advisable for patients in whom hypoglycaemic episodes might be of clinical relevance and in those where dose adjustments may be required. Warning signs of hypoglycaemia may be changed, less pronounced or absent in certain risk groups, potentially resulting in severe hypoglycaemia and loss of consciousness. Risk groups include patients in whom glycaemic control is markedly improved, hypoglycaemia develops gradually, an autonomic neuropathy is present, or in elderly patients. The prolonged effect of subcutaneous insulin glargine may delay recovery from hypoglycaemia. Due to more sustained basal insulin supply with Lantus, less nocturnal but more early morning hypoglycaemia can be expected. Insulin administration may cause insulin antibodies to form. Rarely, this may necessitate dose adjustment. Cases of cardiac failure have been reported when pioglitazone was used in combination with insulin, especially in patients with risk factors for development of cardiac heart failure. Patients on this combination should be observed for signs and symptoms of heart failure, weight gain and oedema. Pioglitazone should be discontinued if any deterioration in cardiac symptoms occurs. Pregnancy and lactation: No clinical data on exposed pregnancies from controlled clinical trials are available. A large amount of post-marketing data indicates no specific adverse effects of insulin glargine on pregnancy and no specific malformative nor feto/neonatal toxicity. Use of Lantus in pregnancy can be considered if clinically needed. Insulin requirements may decrease during the first trimester and generally increase during the second and third trimesters. Immediately after delivery, insulin requirements decline rapidly (increased risk of hypoglycaemia). Careful monitoring of glucose control is essential. It is unknown if insulin glargine is excreted in breast milk. Adverse reactions: Very common: hypoglycaemia. Prolonged or severe hypoglycaemia may be life-threatening. Common: lipohypertrophy, injection site reactions, including redness, itching, pain, hives, swelling or inflammation. Rarely: immediate-type allergic reactions; which may be associated with generalised skin reactions, angio-oedema, bronchospasm, hypotension and shock and may be life threatening; visual impairment, retinopathy and oedema. Very rare: dysgeusia, myalgia Overdose may lead to severe and sometimes long-term and life-threatening hypoglycaemia. Please consult Summary of Product Characteristics for full details of the recognised side effects with Lantus. NHS price: 1 x 10 ml vial £ 30. 68; 5 x 3 ml cartridge £ 41. 50; 5 x 3 ml Solo. Star £ 41. 50 Legal category: POM. MA holder: Sanofi Aventis Deutschland Gmb. H, D-65926 Frankfurt am Main, Germany. MA Numbers: Lantus cartridge: EU/1/00/134/006. Lantus vial EU/1/00/134/012. Lantus Solo. Star: EU/1/00/134/033. Full prescribing information is available from: Sanofi, One Onslow Street, Guildford, Surrey, GU 1 4 YS. Tel: 01483 505515 or the Sanofi Diabetes Care Line 08000 35 25 25. Date of PI Revision: August 2015 Adverse events should be reported. Reporting forms and information can be found at www. mhra. gov. uk/yellowcard. Adverse events should also be reported to the Sanofi drug safety department on 01483 554242. GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015

Apidra Prescribing Information Adverse events should be reported. Reporting forms and information can be found at www. mhra. gov. uk/yellowcard. Adverse events should also be reported to the Sanofi drug safety department on 01483 554242. GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015

Insuman Prescribing Information Insuman® (insulin human). Please refer to Summary of Product Characteristics prior to use of Insuman Basal, Comb 15, Comb 25, Comb 50 and Rapid cartridges, in addition to the Insuman Basal and Comb 25 Solostar pens each contain 300 IU of insulin human in 3 ml, equivalent to 10. 5 mg. Insuman Basal and Comb 25 vials contain 500 IU of insulin human in 5 ml, equivalent to 17. 5 mg. Insuman Rapid contains neutral insulin solution (regular insulin). Insuman Basal contains isophane insulin suspension. Insuman Comb 15, 25 and 50 contain 15% regular and 85% isophane, 25% regular and 75% isophane, and 50% regular and 50% isophane insulin, respectively. Indications: Diabetes mellitus. Insuman Rapid cartridges are also suitable for the treatment of hyperglycaemic coma, ketoacidosis, and for achieving pre-, intra- and post-operative stabilisation. Dosage and administration: Insulin doses and timings should be determined individually. There are no fixed rules for insulin dosage. Average insulin requirement is often 0. 5 to 1. 0 IU/kg body weight/day. Basal metabolic requirement is 40% to 60% of total daily requirement. Insuman should be injected subcutaneously: Insuman Rapid should be injected 15 to 20 minutes before a meal; Insuman Basal should be injected 45 to 60 minutes before a meal; Insuman Comb 15 and 25 should be injected 30 to 45 minutes before a meal; Insuman Comb 50 should be injected 20 to 30 minutes before a meal. Insuman Rapid cartridges may also be administered intravenously under close medical supervision. Close metabolic monitoring is recommended during, and for a period after, transition from other insulins to Insuman. Dose and timing of other antidiabetic medicines may need to be adjusted. Dose adjustments may also be required if the patient’s weight or lifestyle changes, the timing of insulin dose is changed or other circumstances arise that increase susceptibility to hypo- or hyperglycaemia. Insulin requirements may be diminished in the elderly or patients with renal or hepatic impairment. Contra-indications: Hypersensitivity to human insulin or to any excipients. IV administration contraindicated in all preparations except Insuman Rapid. Insuman Basal contraindicated in infusion pumps or external or implanted insulin pumps. Insuman Rapid must not be used in external or implanted insulin pumps or in peristaltic pumps with silicone tubing. Precautions and warnings: Patients hypersensitive to Insuman Rapid, Basal or Comb 15, 25 or 50 for whom no better tolerated preparation is available must only continue treatment under close medical supervision, in conjunction with anti-allergic treatment if necessary. In patients allergic to animal insulin, intradermal skin testing is recommended prior to initiation. Hypoglycaemia may occur. Particular caution should be exercised, and intensified blood monitoring is advisable for patients in whom hypoglycaemic episodes might be of clinical relevance and in those where dose adjustments may be required. Warning signs of hypoglycaemia may be changed, less pronounced or absent in certain risk groups, potentially resulting in severe hypoglycaemia and loss of consciousness. Risk groups include patients in whom glycaemic control is markedly improved, hypoglycaemia develops gradually, an autonomic neuropathy is present, or in elderly patients. Intensified metabolic monitoring is necessary during intercurrent illness. Cases of cardiac failure have been reported when pioglitazone was used in combination with insulin, especially in patients with risk factors for development of cardiac heart failure. Patients should be observed for signs and symptoms of heart failure, weight gain and oedema. Pioglitazone should be discontinued if any deterioration in cardiac symptoms occurs. Pregnancy and lactation: There is no clinical data on the use of Insuman in pregnant women. Insuman can be used during breast feeding. Adverse Reactions: Hypoglycaemia is the most common reaction. Prolonged or severe hypoglycaemia may be life-threatening. Severe hypoglycaemic attacks, especially if recurrent, may lead to neurological damage. Common: oedema, injection site reactions. Uncommon: shock, injection site urticaria. Immediate type allergic reactions to insulin or to the excipients may be life-threatening (hypotension, angioneurotic oedema, bronchospasm, generalised skin reactions). Insulin administration may cause insulin antibodies to form and may, in rare cases, necessitate adjustment of the insulin dose. Product Pack NHS Price MA Number Insuman Rapid 5 cartridges of 3 ml £ 17. 50 EU/1/97/030 Insuman Basal 1 vial of 5 ml: £ 5. 61 EU/1/97/030/033 Insuman Basal 5 cartridges of 3 ml £ 17. 50 EU/1/97/030/035 Insuman Basal 5 Solo. STAR pens of 3 ml £ 19. 80 EU/1/97/030/148 Insuman Comb 15 5 cartridges of 3 ml £ 17. 50 EU/1/97/030/040 Insuman Comb 25 1 vial of 5 ml £ 5. 61 EU/1/97/030/043 Insuman Comb 25 5 cartridges of 3 ml £ 17. 50 EU/1/97/030/045 Insuman Comb 25 5 Solo. STAR pens of 3 ml £ 19. 80 EU/1/97/030/160 Insuman Comb 50 5 cartridges of 3 ml £ 17. 50 EU/1/97/030/050 Legal category: POM. MA holder: Sanofi Aventis Deutschland Gmb. H, D-65926 Frankfurt am Main, Germany. Full prescribing information is available from: Sanofi, One Onslow Street, Guildford, Surrey, GU 1 4 YS. Tel: 0845 372 7101. Date of Revision: October 2014 Adverse events should be reported. Reporting forms and information can be found at www. mhra. gov. uk/yellowcard. Adverse events should also be reported to the Sanofi drug safety department on 01483 554242. GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015

Lyxumia Prescribing Information careful clinical monitoring or have a narrow therapeutic ratio. Lyxumia has not been studied in combination with dipeptidyl peptidase 4 (DPP-4) inhibitors. Lyxumia▼® (lixisenatide). There is limited experience in patients with congestive heart failure. Patients treated with Please refer to Summary of Product Characteristics prior to use of Lyxumia should be advised of the potential risk of dehydration in relation to gastrointestinal Lyxumia 10 micrograms solution for injection: adverse reactions and take precautions to avoid fluid depletion. This medicinal product contains Each dose (0. 2 ml) contains 10 micrograms (mcg) of lixisenatide (50 mcg per ml). metacresol, which may cause allergic reactions. Lyxumia 20 micrograms solution for injection: Interactions: Paracetamol: No dose adjustment for paracetamol is required but the delayed t max Each dose (0. 2 ml) contains 20 micrograms (mcg) of lixisenatide (100 mcg per ml). observed when paracetamol is administered 1 -4 hours after Lyxumia should be taken into Indications: Treatment of adults with type 2 diabetes mellitus to achieve glycaemic control in account when a rapid onset of action is required for efficacy. Oral contraceptives: The reduction combination with oral glucose‑lowering medicinal products and/or basal insulin when these, in Cmax is of limited clinical relevance and no dose adjustment for oral contraceptives is required. together with diet and exercise, do not provide adequate glycaemic control. Atorvastatin: Changes to t max and Cmax are not clinically relevant and therefore, no dose Dosage and administration: Lyxumia is to be injected subcutaneously in the thigh, abdomen adjustment for atorvastatin is required when co ‑administered with Lyxumia. Warfarin and other coumarin derivatives: Frequent monitoring of INR in patients on warfarin and/or coumarin or upper arm. Starting dose: dosing is initiated at 10 mcg Lyxumia once daily for 14 days. Maintenance dose: a fixed maintenance dose of 20 mcg Lyxumia once daily is started on Day derivatives is recommended at the time of initiation or ending of Lyxumia treatment. Digoxin: No dose adjustment for digoxin is required when co ‑administered with Lyxumia. Ramipril: No dose 15. Lyxumia 20 micrograms solution for injection is available for the maintenance dose. Lyxumia is adjustment for ramipril is required when co‑administered with Lyxumia. administered once daily, within the hour prior to any meal of the day, preferably the same meal Adverse reactions: Very common: Hypoglycaemia (in combination with a sulphonylurea and / each day. If a dose of Lyxumia is missed, it should be injected within the hour prior to the next or a basal insulin). Headache. Nausea, vomiting & diarrhoea; These tend to be mostly mild and meal. The clinical experience in patients ≥ 75 years is limited. Limited therapeutic experience in transient during the first 3 weeks after starting treatment. Thereafter, they progressively decrease during the following weeks. Common: Influenza, upper respiratory tract infection, patients with moderate renal impairment (creatinine clearance: 30 ‑ 50 ml/min) and Lyxumia cystitis & viral infection. Hypoglycaemia (in combination with metformin alone). Dizziness, should be used with caution in this population. There is no therapeutic experience in patients somnolence, dyspepsia, back pain, injection site pruritus. Uncommon: Anaphylactic reaction with severe renal impairment (creatinine clearance less than 30 ml/min) or end-stage renal disease and therefore, it is not recommended to use Lyxumia in these populations. The safety and urticaria. Please consult the Summary of Product Characteristics in relation to other adverse reactions and efficacy of Lyxumia in children and adolescents less than 18 years of age have not yet been established. NHS price: Lyxumia 10 mcg: 1 pre-filled pen: £ 31. 67, Lyxumia 20 mcg: 2 pre-filled pens: Contraindications: Hypersensitivity to Lyxumia or to any of the excipients £ 57. 93, Lyxumia 10 mcg + 20 mcg: 1 pre-filled pen + 1 pre-filled pen: £ 57. 93 Precautions and warnings: No therapeutic experience with Lyxumia in patients with type 1 Legal category: POM. diabetes mellitus and it should not be used in these patients. Lyxumia should not be used for treatment of diabetic ketoacidosis. Use of glucagon ‑like peptide-1 (GLP‑ 1) receptor agonists MA holder: sanofi-aventis groupe, 54, rue La Boétie, F – 75008 Paris, France has been associated with a risk of developing acute pancreatitis. There have been few reported MA Number(s): Lyxumia 10 mcg: 1 pre-filled pen: EU/1/12/811/001, Lyxumia 20 mcg: 2 prefilled pens: EU/1/12/811/003, Lyxumia 10 mcg + 20 mcg: 1 pre-filled pen + 1 pre-filled pen: events of acute pancreatitis with Lyxumia although a causal relationship has not been established. If pancreatitis is suspected, Lyxumia should be discontinued; if acute pancreatitis EU/1/12/811/005 Full prescribing information is available from: Sanofi, One Onslow Street, Guildford, Surrey, is confirmed, Lyxumia should not be restarted. Caution should be exercised in patients with a GU 1 4 YS. Tel: 0845 372 7101. history of pancreatitis. Lyxumia has not been studied in patients with severe gastrointestinal disease, including severe gastroparesis and therefore, the use of Lyxumia is not recommended Date of Revision: June 2015 in these patients. There is limited therapeutic experience in patients with moderate renal impairment (creatinine clearance: 30‑ 50 ml/min) and no therapeutic experience in patients with Adverse events should be reported. Reporting forms and severe renal impairment (creatinine clearance less than 30 ml/min) or end-stage renal disease. information can be found at www. mhra. gov. uk/yellowcard. Lyxumia should be used with caution in patients with moderate renal impairment. Use is not recommended in patients with severe renal impairment or end-stage renal disease. Patients Adverse events should also be reported to the Sanofi drug receiving Lyxumia with a sulphonylurea or with a basal insulin may have an increased risk of safety department on 01483 554242. hypoglycaemia. Lyxumia should not be given in combination with basal insulin and a sulphonylurea due to increased risk of hypoglycaemia. Lyxumia should be used with caution in patients receiving oral medicinal products that require rapid gastrointestinal absorption, require GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015

Toujeo® Prescribing Information Toujeo® (insulin glargine 300 U/ml). Please refer to Summary of Product Characteristics prior to use of Toujeo ®. Toujeo® Solo. Star® pre-filled pens each contain 450 Units of insulin glargine in 1. 5 ml of solution for injection, equivalent to 10. 91 mg/ml. Indications: Treatment of diabetes mellitus in adults. Administration: Toujeo® is administered subcutaneously once daily, at any time of the day, preferably at the same time every day. Do not administer intravenously. Insulin glargine dose regimen (dose and timing) should be individually adjusted. In type 1 diabetes mellitus, Toujeo® must be combined with short-/rapid-acting insulin to cover mealtime insulin requirements. In patients with type 2 diabetes mellitus, Toujeo® can also be given together with other antihyperglycaemic medicinal products. Close metabolic monitoring is recommended during the switch and in the initial weeks thereafter. Dose and timing of other antidiabetic medicines may need to be adjusted. Dose adjustments may also be required if the patient’s weight or lifestyle changes, the timing of insulin dose is changed or other circumstances arise that increase susceptibility to hypo- or hyperglycaemia. Toujeo® must not be mixed or diluted with any other insulin or other medicinal products. Mixing or diluting Toujeo® changes its time/action profile and mixing causes precipitation. Insulin requirements may be diminished in the elderly or patients with renal or hepatic impairment. The safety and efficacy of Toujeo® have not been established in children and adolescents below 18 years of age. No data are available. Contraindications: Hypersensitivity to insulin glargine or any excipients. Precautions and warnings: Insulin glargine 100 units/ml and Toujeo® are not bioequivalent and are not directly interchangeable. When switching from insulin glargine 100 units/ml to Toujeo®, this can be done on a unit‑to‑unit basis, but a higher Toujeo® dose (approximately 10 -18%) may be needed to achieve target ranges for plasma glucose levels. When switching from Toujeo® to insulin glargine 100 units/ml, the dose should be reduced (approximately by 20%) to reduce the risk of hypoglycaemia. Close metabolic monitoring is recommended during the switch and in the initial weeks thereafter. Toujeo® is not the insulin of choice for treatment of diabetic ketoacidosis. In case of insufficient glucose control or a tendency to hyper/hypoglycaemic episodes, the patient's adherence to the prescribed treatment regimen, injection sites and proper injection technique and all other relevant factors must be reviewed before dose adjustment is considered. Insulin administration may cause insulin antibodies to form. Rarely, this may necessitate dose adjustment. Particular caution should be exercised, and intensified blood glucose monitoring is advisable for patients in whom hypoglycaemic episodes might be of clinical relevance and in those where dose adjustments may be required. Warning signs of hypoglycaemia may be changed, less pronounced or absent in certain risk groups, potentially resulting in severe hypoglycaemia and loss of consciousness. Risk groups include patients in whom glycaemic control is markedly improved, hypoglycaemia develops gradually, an autonomic neuropathy is present, or who are elderly. The prolonged effect of subcutaneous insulin glargine may delay recovery from hypoglycaemia. Cases of cardiac failure have been reported when pioglitazone was used in combination with insulin, especially in patients with risk factors for development of cardiac heart failure. If the combination is used, patients should be observed for signs and symptoms of heart failure, weight gain and oedema. Pioglitazone should be discontinued if any deterioration in cardiac symptoms occurs. Pregnancy and lactation: No clinical data on exposed pregnancies from controlled clinical trials are available. A large amount of data on pregnant women (more than 1000 pregnancy outcomes with a medicinal product containing insulin glargine 100 units/ml (Lantus ®)) indicate no specific adverse effects on pregnancy and no specific malformative nor feto/neonatal toxicity of insulin glargine. Animal data do not indicate reproductive toxicity. The use of Toujeo ® may be considered during pregnancy, if clinically needed. Insulin requirements may decrease during the first trimester and generally increase during the second and third trimesters. Immediately after delivery, insulin requirements decline rapidly (increased risk of hypoglycaemia). Careful monitoring of glucose control is essential. It is unknown if insulin glargine is excreted in breast milk. Adverse reactions: Very common: Hypoglycaemia. Prolonged or severe hypoglycaemia may be life-threatening. Common: Lipohypertrophy, injection site reactions, including redness, pain, itching, hives, swelling, or inflammation. Uncommon: Lipoatrophy. Rarely: Immediate-type allergic reactions; which may be associated with generalised skin reactions, angio-oedema, bronchospasm, hypotension and shock and may be life threatening; visual impairment, retinopathy and oedema. Very rare: Dysgeusia, myalgia. Insulin administration may cause insulin antibodies to form and may, in rare cases, necessitate adjustment of the insulin dose. Overdose may lead to severe and sometimes long-term and lifethreatening hypoglycaemia. Please consult Summary of Product Characteristics for full details of the recognised side effects with Toujeo ®. NHS price: £ 33. 13 for pack of x 3 1. 5 ml pens Legal category: POM. MA holder: Sanofi Aventis Deutschland Gmb. H, D-65926 Frankfurt am Main, Germany. MA Numbers: Solo. Star ®: 3 Pen pack: EU/1/00/133/034 Full prescribing information is available from: Sanofi, One Onslow Street, Guildford, Surrey, GU 1 4 YS. Tel: 01483 505515 or the Sanofi Diabetes Care Line 08000 35 25 25. Date of PI Revision: May 2015 Adverse events should be reported. Reporting forms and information can be found at www. mhra. gov. uk/yellowcard. Adverse events should also be reported to the Sanofi drug safety department on 01483 554242. GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015 29

References and links Data in this presentation are from publicly available websites courtesy of Public Health England which host NCVIN (National Cardiovascular Intelligence Network) and NDIS (National Diabetes Intelligence Service); and NHS BSA (NHS Business Services Authority) ● ● DOVE – accessed 05/15 at http: //www. yhpho. org. uk/resource/view. aspx? RID=88739 Diabetes Community Health Profiles – accessed 05/15 at http: //www. yhpho. org. uk/diabetescommunityhealthprofiles/default. aspx National General Practice Profiles – accessed 05/15 at http: //fingertips. phe. org. uk/profile/general-practice NHS Business Services Authority MO KTT indicators accessed 05/15 via https: //apps. nhsbsa. nhs. uk/infosystems/homepage. do GBIE. DIA. 12. 09. 13(4) Date of Prep – October 2015
- Slides: 35