Year 10 Paper 1 Combined Chemistry Higher Revision

Year 10 (Paper 1) Combined Chemistry Higher Revision Name………………….
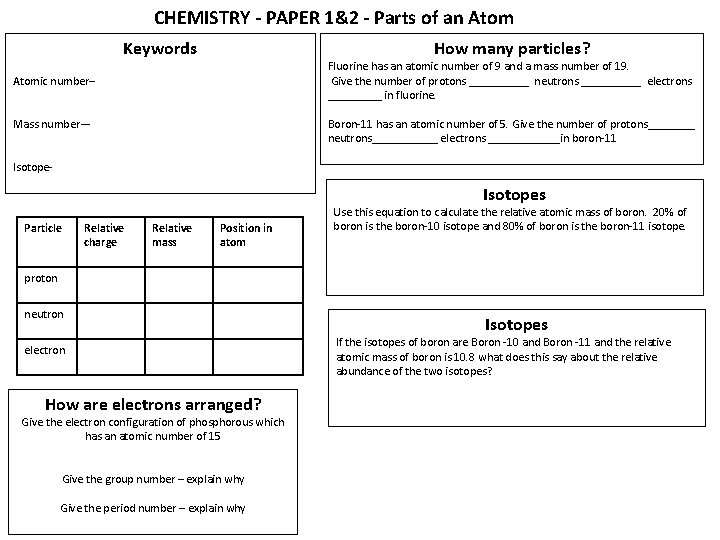
CHEMISTRY - PAPER 1&2 - Parts of an Atom Keywords How many particles? Fluorine has an atomic number of 9 and a mass number of 19. Give the number of protons _____ neutrons _____ electrons _____ in fluorine. Atomic number– Mass number— Boron-11 has an atomic number of 5. Give the number of protons____ neutrons______ electrons ______in boron-11 Isotope- Isotopes Particle Relative charge Relative mass Position in atom Use this equation to calculate the relative atomic mass of boron. 20% of boron is the boron-10 isotope and 80% of boron is the boron-11 isotope. proton neutron electron How are electrons arranged? Give the electron configuration of phosphorous which has an atomic number of 15 Give the group number – explain why Give the period number – explain why Isotopes If the isotopes of boron are Boron -10 and Boron -11 and the relative atomic mass of boron is 10. 8 what does this say about the relative abundance of the two isotopes?

HIGHER - PAPER 1&2 - Metallic & Ionic Bonding Keywords Formation of ions Describe how a chloride ion forms from a chlorine atom. Ionic Bond– Lattice- Describe how a sodium ion forms from a sodium atom. Metallic Bonding Draw and label a diagram showing metallic bonding. Properties of Ionic Compounds Explain why metals conduct electricity Explain why metals are malleable - Explain why ionic compounds, such as Ca. O, have high melting points - Explain why solid ionic compounds, such as sodium chloride, do not conduct as solids but do conduct as molten liquids or when dissolved in water. -

PAPER 1&2 - Covalent Bonding What is a Covalent Bond? Properties of Covalent Compounds Explain why simple covalent compounds, such as chlorine, have low boiling points - Diamond Describe the structure of diamond Explain why diamond has a high melting point. Explain why diamond is hard. - Explain why simple covalent compounds, such as oxygen, do not conduct electricity. - Explain why diamond does not conduct electricity. Graphite Describe the structure of graphite. Dot and Cross Diagrams Draw dot and cross diagrams for the following using the following electron configurations: H: 1 C=2. 4 Cl: 2. 8. 7 O: 2. 6 Explain why graphite has a high melting point. Explain why graphite is slippery and soft. H 2 HCl H 2 O Explain graphite conducts electricity. Graphene Describe the structure of graphene CH 4 Buckminsterfullerene O 2 Describe the structure of buckminsterfullerene. Explain why buckminsterfullerene has a low melting point CO 2

PAPER 1&2 - Calculations Calculate Empirical Formula Give the empirical formula of C 5 H 10 If 88. 8 g of copper reacts with 11. 2 g of oxygen calculate the empirical formula. (Relative Atomic Mass Cu: 63. 5, O: 16) Calculate the Rf value If a coloured spot rises by 6 cm and the solvent rises by 10 cm calculate the Rf value Calculating theoretical mass Calculate relative atomic mass A sample contains 70% of copper-63 atoms and 30% of copper 65 atoms. Use this information to calculate the relative atomic mass of copper Calculate concentration A solution is made by dissolving 2. 5 g of sodium chloride in 250 cm 3 of water. Calculate the concentration in gdm-3 of the solution Calculate the number of moles in 26 g of potassium (Relative atomic mass: K=39 2 Na. F 2 Na + F 2 (Relative atomic mass: Na = 23 F= 19) Calculate the maximum mass of sodium that can be formed from 150 g of sodium fluoride In an experiment, 11. 2 g of iron Fe reacts exactly with 76. 2 g of iodine, I 2. Determine the balanced equation for the reaction between iron Fe and iodine. I 2(Relative atomic masses: Fe=56, I=127) Calculate the number of particles in 5 g of potassium (Relative atomic mass: K=39) (Avogadro’s constant = 6. 02 x 1023)

Key words HIGHER PAPER 1 – Equilibrium What does this mean? ⇌ Temperature What is dynamic equilibrium ? Conditions Effect on Equilibrium Effect on Yield Why? Haber Process: N 2 + 3 H 2 ⇌ 2 NH 3 The forward reaction is Exothermic. Where does the nitrogen come from? Where does the hydrogen come from? Pressure Catalyst

Acid – HIGHER - PAPER 1 - Acids Keywords Alkali — Hydrogen Acid + Alkali added together = Carbon dioxide Weak acid Oxygen Gas Tests Strong acid Word equations and balanced equations Sodium + hydrochloric acid Sodium hydroxide + sulfuric acid Calcium carbonate + nitric acid Potassium oxide + hydrochlori c acid Sulfuric acid Acid formulae Give the ionic equation for the neutralisation reaction of an acid + and allkali Hydrochloric acid Indicators Colour in acid Nitric acid Acid dilution If an acid of p. H 2 is diluted from 100 cm 3 to 1000 cm 3 what is the new p. H? If acid of p. H 4 is diluted from 10 cm 3 to 1000 cm 3 what is the new p. H? Universal Indicator Methyl orange Phenol Phthalein Colour in alkali
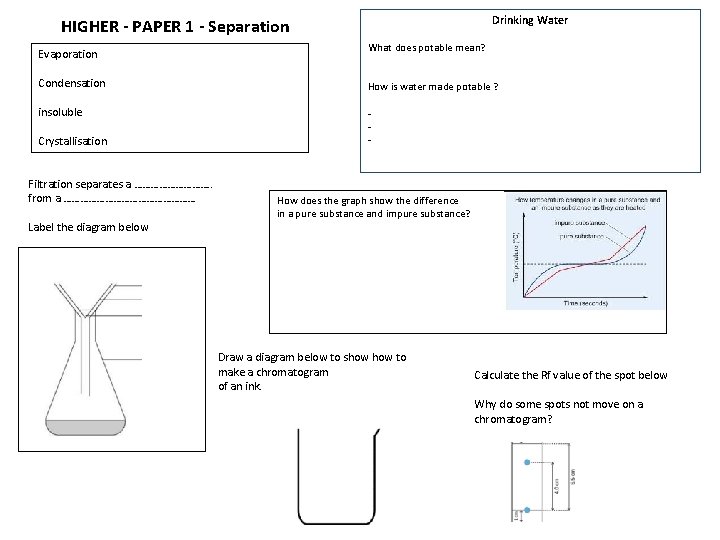
Drinking Water HIGHER - PAPER 1 - Separation Evaporation What does potable mean? Condensation How is water made potable ? insoluble - Crystallisation Filtration separates a ……………. . from a …………………… Label the diagram below How does the graph show the difference in a pure substance and impure substance? Draw a diagram below to show to make a chromatogram of an ink. Calculate the Rf value of the spot below Why do some spots not move on a chromatogram?

PAPER 1 - Extraction of Metals In the following reaction what is reduced and what is oxidised? Explain Extracting metals Give the method to extract each metal and explain why that method is used Gold – Iron oxide + carbon iron + carbon dioxide. Iron- Aluminium – Give 3 benefits of recycling metals - Balance the equation: Fe 2 O 3 + C Fe + CO 2 What factors should life cycle assessment take into account? -

Anion Key words Cation Anode Cathode Electrolyte PAPER 1 - Electrolysis When molten lead bromide undergoes electrolysis what are the products at the Anode: Cathode? Oxidation takes place at which electrode? Oxidation Reduction Complete the following half equations. The first has been done for you Na+ + e- Na Reduction takes place at which electrode? Which ions are present in water and in solutions? What are electrodes made out of? H+ Cu 2+ Cl. O 2 OHCu What does oil rig stand for ?
- Slides: 10