Xpert MTBRIF Ultra The NextGeneration Assay for Tuberculosis

Xpert® MTB/RIF Ultra: The Next-Generation Assay for Tuberculosis Diagnosis and Rifampin Resistance Detection Diego P. Peralta, M. D. , FACP Assistant Professor Division of Infectious Diseases March 22, 2018 Texas Association for Clinical Laboratory Science
Disclosures • No financial disclosures to report.

Objectives At the end of the session, the participant will be able to: 1. Identify the differences between the Xpert® MTB/RIF Ultra assay and its predecessor. 2. Describe the Xpert® MTB/RIF Ultra assay clinical applications. 3. Appraise the evidence to support the use of Xpert® MTB/RIF Ultra assay in tuberculosis diagnosis and Rifampin resistance detection.
Tuberculosis http: //www. healthtipsever. com/what-is-tuberculosis/
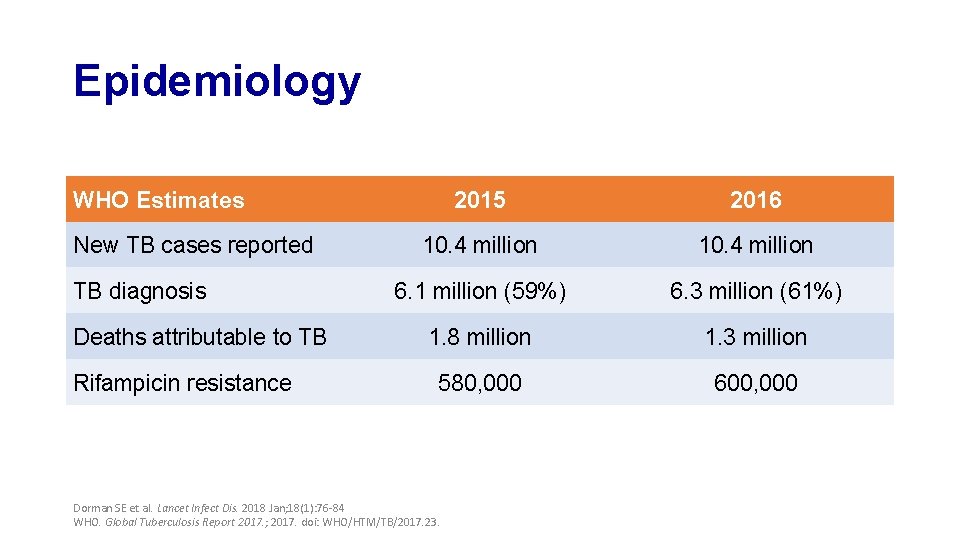
Epidemiology WHO Estimates New TB cases reported TB diagnosis Deaths attributable to TB Rifampicin resistance 2015 2016 10. 4 million 6. 1 million (59%) 6. 3 million (61%) 1. 8 million 1. 3 million 580, 000 600, 000 Dorman SE et al. Lancet Infect Dis. 2018 Jan; 18(1): 76 -84 WHO. Global Tuberculosis Report 2017. ; 2017. doi: WHO/HTM/TB/2017. 23.
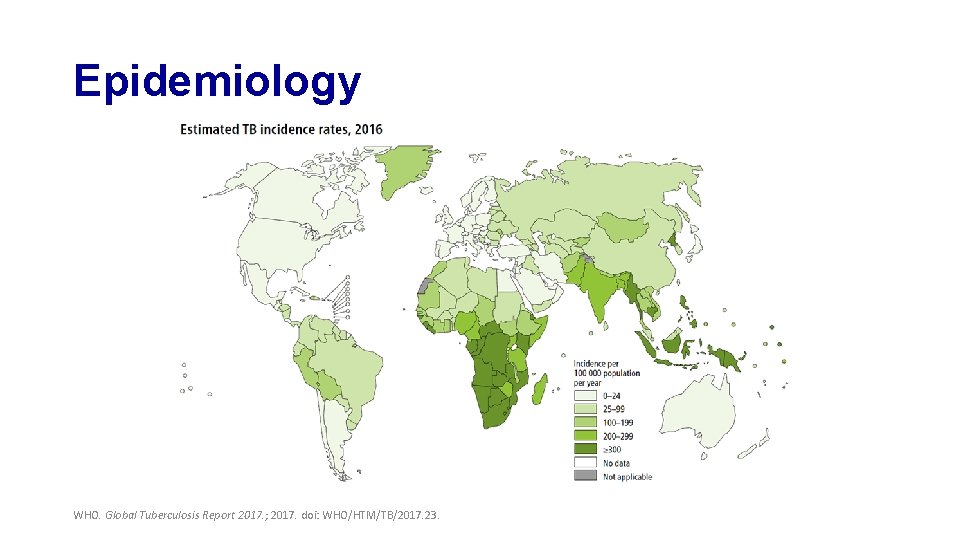
Epidemiology WHO. Global Tuberculosis Report 2017. ; 2017. doi: WHO/HTM/TB/2017. 23.
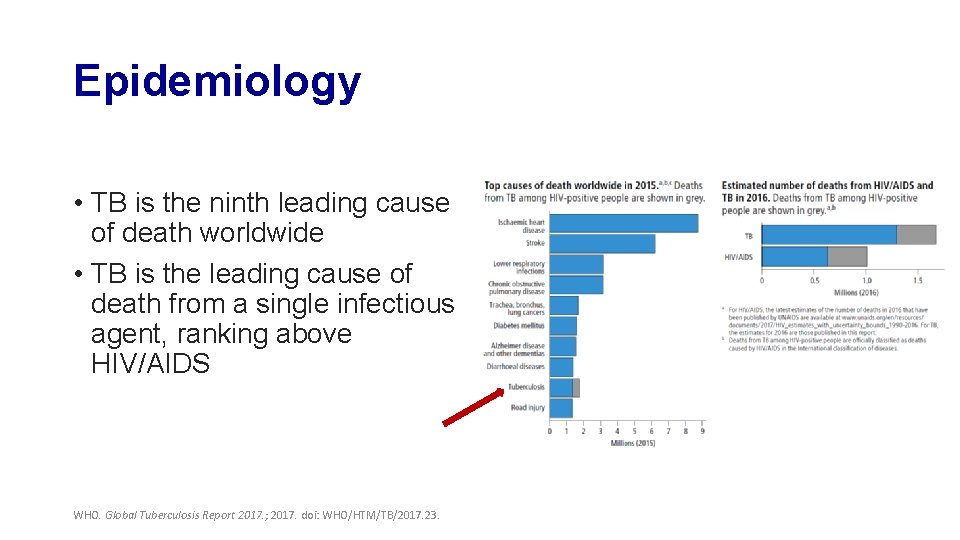
Epidemiology • TB is the ninth leading cause of death worldwide • TB is the leading cause of death from a single infectious agent, ranking above HIV/AIDS WHO. Global Tuberculosis Report 2017. ; 2017. doi: WHO/HTM/TB/2017. 23.
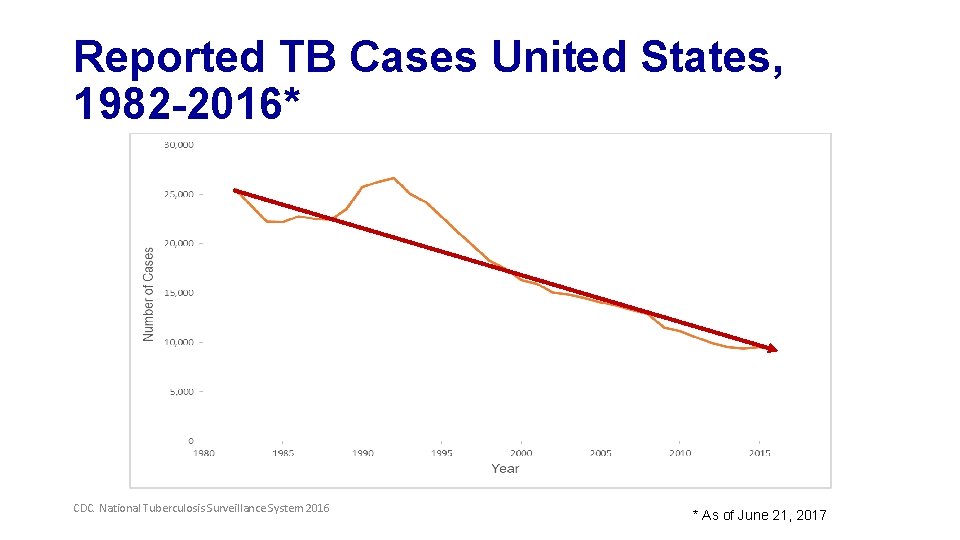
Reported TB Cases United States, 1982 -2016* CDC. National Tuberculosis Surveillance System 2016 * As of June 21, 2017
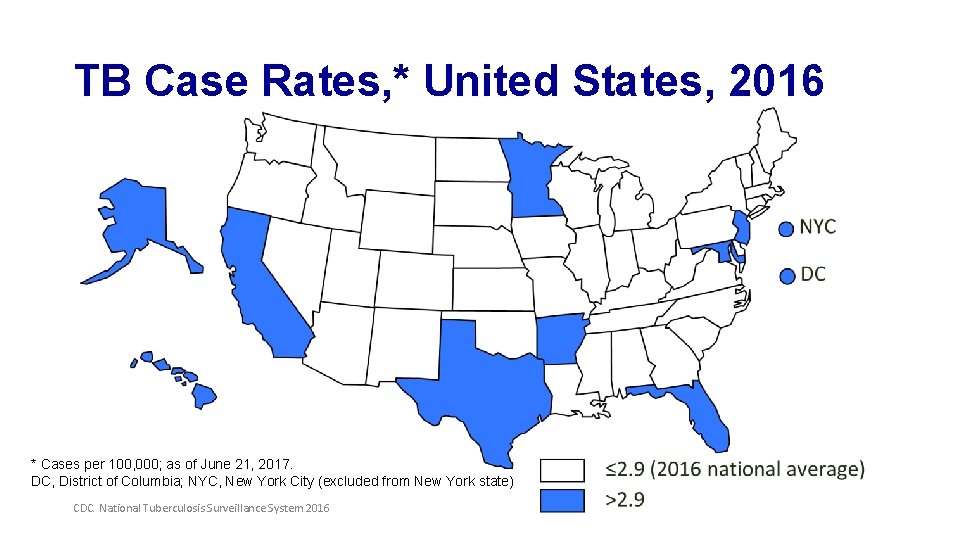
TB Case Rates, * United States, 2016 * Cases per 100, 000; as of June 21, 2017. DC, District of Columbia; NYC, New York City (excluded from New York state) CDC. National Tuberculosis Surveillance System 2016
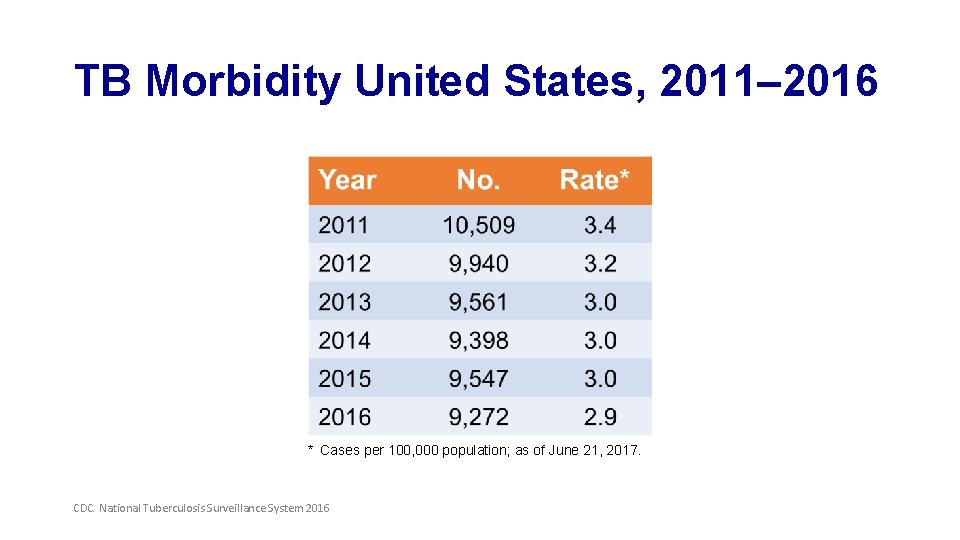
TB Morbidity United States, 2011– 2016 * Cases per 100, 000 population; as of June 21, 2017. CDC. National Tuberculosis Surveillance System 2016
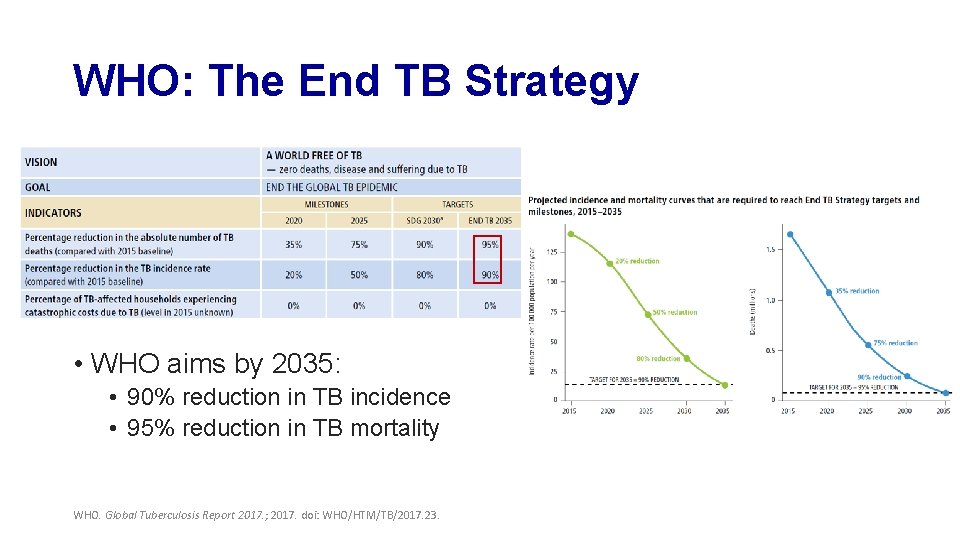
WHO: The End TB Strategy • WHO aims by 2035: • 90% reduction in TB incidence • 95% reduction in TB mortality WHO. Global Tuberculosis Report 2017. ; 2017. doi: WHO/HTM/TB/2017. 23.

Diagnosis • Gold standard • Culture and drug susceptibility • Smear microscopy is insensitive • Culture takes too long to be clinically useful • For TB diagnosis, highly sensitive, rapid, and accessible diagnostics are needed WHO. Global Tuberculosis Report 2017. ; 2017. doi: WHO/HTM/TB/2017. 23.

Diagnosis • Several assays are endorsed by the WHO: • Line Probe Assays • Gen-Probe Mycobacterium Tuberculosis Direct • Urine lateral flow lipoarabinomannan (LF-LAM) • Loop-mediated isothermal amplification (TB-LAMP) • Xpert® MTB/RIF assay WHO. Global Tuberculosis Report 2017. ; 2017. doi: WHO/HTM/TB/2017. 23.

Xpert® MTB/RIF Assay • Automated in vitro test • Uses real-time PCR • Qualitative detection of MTB and RIF resistance • Primers amplify a portion of rpo. B gene containing the 81 base pair core region • Probes differentiate the conserved wild-type sequence and mutations in the core region associated with RIF resistance Gene. Xpert Technology. Cepheid Website http: //www. cepheid. com/us/cepheid-solutions/clinical-ivd-tests/critical-infectious-diseases/xpert-mtb-rif/309? view=product
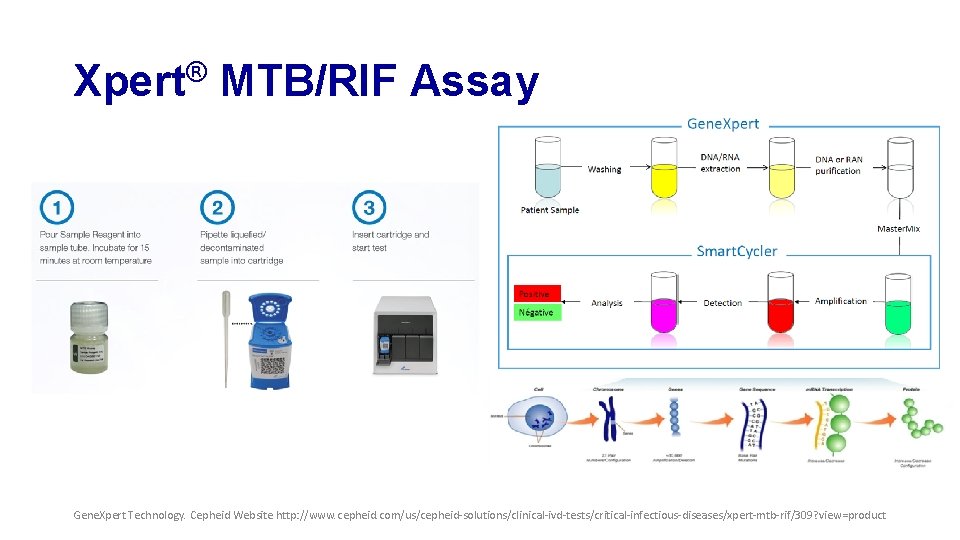
Xpert® MTB/RIF Assay Gene. Xpert Technology. Cepheid Website http: //www. cepheid. com/us/cepheid-solutions/clinical-ivd-tests/critical-infectious-diseases/xpert-mtb-rif/309? view=product

History • 2010 • WHO endorsed the Xpert® MTB/RIF assay for initial diagnostic testing of individuals suspected of MDR-TB or HIV-associated TB • 2013 • FDA permitted marketing of the Xpert® MTB/RIF assay • 2014 • WHO expanded recommendation for use in all patients (including also extra-pulmonary TB) MMWR Morb Mortal Wkly Rep. 2013 Oct 18; 62(41): 821 -7. WHO. Global Tuberculosis Report 2017. ; 2017. doi: WHO/HTM/TB/2017. 23.

• Xpert® MTB/RIF has limitations • March 2017 WHO recommended: • Replacement of Xpert® MTB/RIF by Xpert® MTB/RIF Ultra Going Ultra: Xpert® MTB/RIF Ultra WHO meeting report of a technical expert consultation: non-inferiority analysis of Xpert MTF/RIF Ultra compared to Xpert MTB/RIF. Geneva: World Health Organization; 2017 (WHO/HTM/TB/2017. 04). Licence: CC BY-NCSA 3. 0 IGO.

Differences between Xpert and Ultra Xpert® MTB/RIF Ultra Two multi-copy amplification targets (IS 6110 and IS 1081) Fully nested nucleic acid amplification More rapid thermal cycling Improved fluidics and enzymes DNA PCR reaction chamber 25 µl 50 µl Limit of detection 114 bacterial CFU/m. L 16 bacterial CFU/m. L Semi-quantitative categories High, medium, low and very low High, medium, low, very low, and trace (trace call) 1–log improvement in the lower limit of detection RIF resistance detection Melting temperature-based analysis Improved differentiation of certain silent RIF mutations and detection of RIF resistance in mixed infections and paucibacillary specimens Real-time PCR WHO meeting report of a technical expert consultation: non-inferiority analysis of Xpert MTF/RIF Ultra compared to Xpert MTB/RIF. Geneva: World Health Organization; 2017 (WHO/HTM/TB/2017. 04). Licence: CC BY-NCSA 3. 0 IGO.

Xpert® MTB/RIF Ultra • Pros: Higher sensitivity • Increase diagnosis of paucibacillary forms of TB • Childhood TB • HIV-associated TB • Extrapulmonary TB • Cons: Lower specificity • Previously treated/history of TB • High-incidence countries WHO meeting report of a technical expert consultation: non-inferiority analysis of Xpert MTF/RIF Ultra compared to Xpert MTB/RIF. Geneva: World Health Organization; 2017 (WHO/HTM/TB/2017. 04). Licence: CC BY-NCSA 3. 0 IGO. Arch Bronconeumol. 2017 Dec; 53(12): 665 -666. doi: 10. 1016/j. arbres. 2017. 05. 023. Epub 2017 Jul 10.

Xpert® MTB/RIF Ultra • Lower specificity represents • Higher proportion of false positive results • Empiric treatment and possibly overtreatment that might undermine clinical effect • Possible explanation • Detection of DNA from non-viable M. tuberculosis • Prolonged excretion of M. tuberculosis DNA form TB lesions after treatment • M. tuberculosis DNA from dead bacilli has high stability (DNA can be detected in millennia-old human remains) WHO meeting report of a technical expert consultation: non-inferiority analysis of Xpert MTF/RIF Ultra compared to Xpert MTB/RIF. Geneva: World Health Organization; 2017 (WHO/HTM/TB/2017. 04). Licence: CC BY-NCSA 3. 0 IGO. Arch Bronconeumol. 2017 Dec; 53(12): 665 -666. doi: 10. 1016/j. arbres. 2017. 05. 023. Epub 2017 Jul 10.


Study Justification • Limitations of Xpert MTB/RIF assay • Imperfect sensitivity for paucibacillary disease (HIV, early disease, children, etc) • Imperfect sensitivity for RIF-resistance detection in case of heteroresistance • Imperfect specificity for RIF-resistance detection due to silent mutation detection • Cross-reactivity with some nontuberculous mycobacteria (NTM) Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf. CROI 2017. Rodwell T. A Multicenter Diagnostic Accuracy Study of the Xpert Ultra for Tuberculosis Diagnosis. http: //www. croiwebcasts. org/console/player/33479? media. Type=audio&

Study • Design: • Blinded, multicenter, prospective non-inferiority study. • Index test: • Xpert® MTB/RIF Ultra • Comparator: • Xpert ® MTB/RIF • Reference standard: • Culture, drug susceptibility, sequencing Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Primary Outcomes: • MTB detection • ∆ Sensitivity: among smear-negative/culture-positive patients • ∆ Specificity • RIF detection • ∆ Sensitivity: among smear-negative/culture-positive patients • ∆ Specificity Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Secondary Outcomes • Estimates of accuracy for MTB detection • • • Overall/pooled By smear-status By HIV status By TB history and time since treatment completion By site On secondary study samples Root-cause analysis of discordant results between Ultra and culture Analyses reclassifying the ‘trace’ call Analysis of NTMs Analysis of semi-quantitative results Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
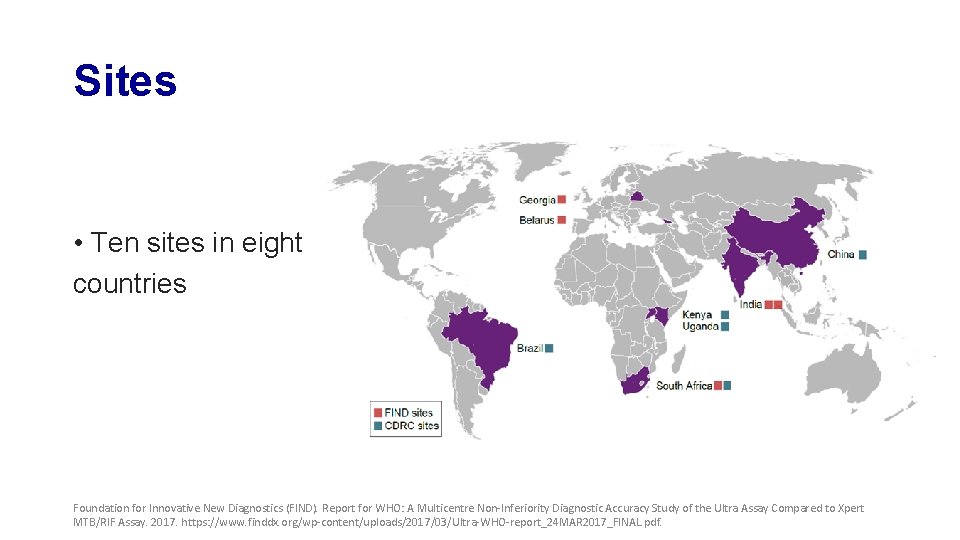
Sites • Ten sites in eight countries Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
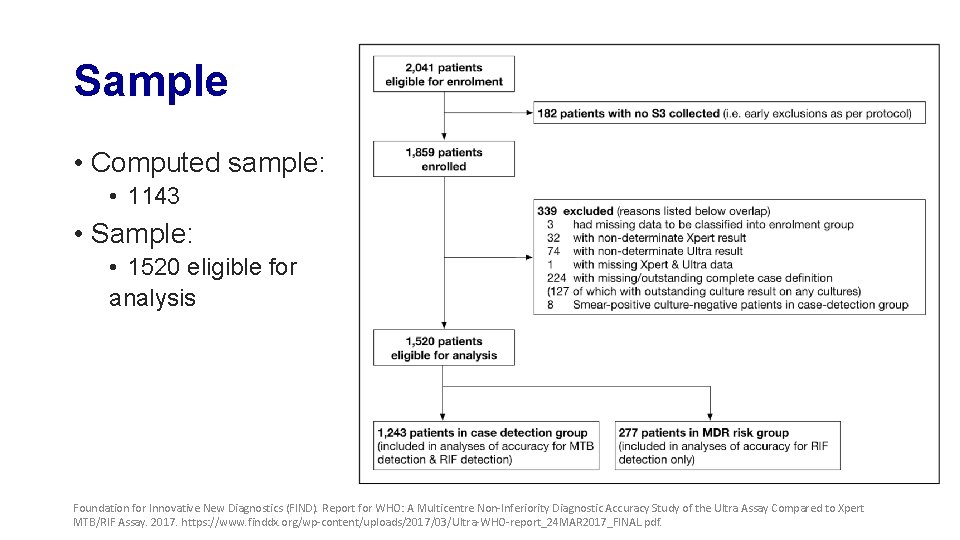
Sample • Computed sample: • 1143 • Sample: • 1520 eligible for analysis Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
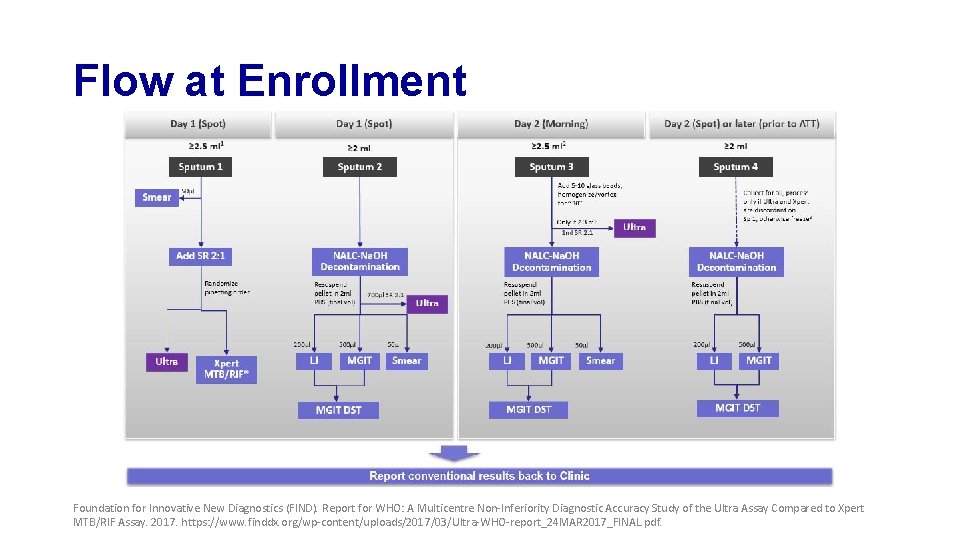
Flow at Enrollment Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
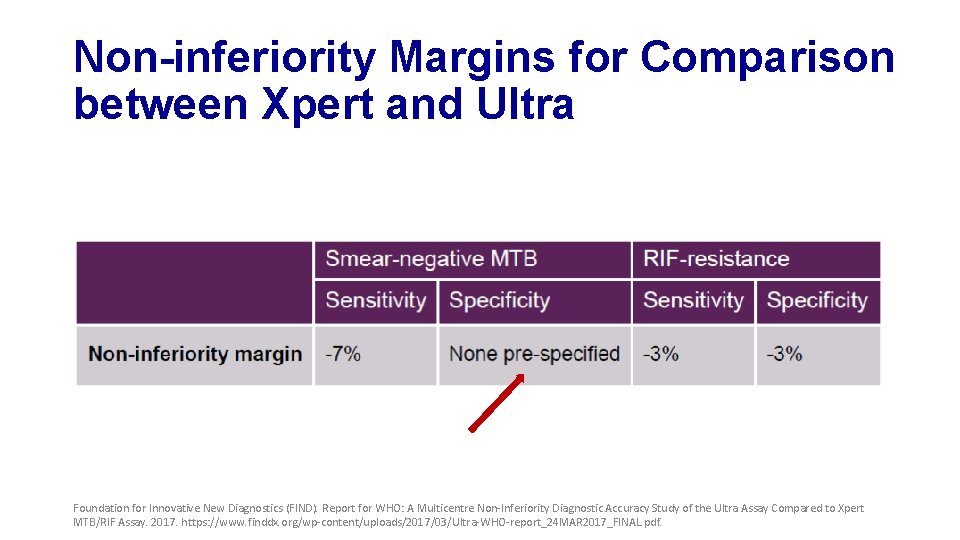
Non-inferiority Margins for Comparison between Xpert and Ultra Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Main Results
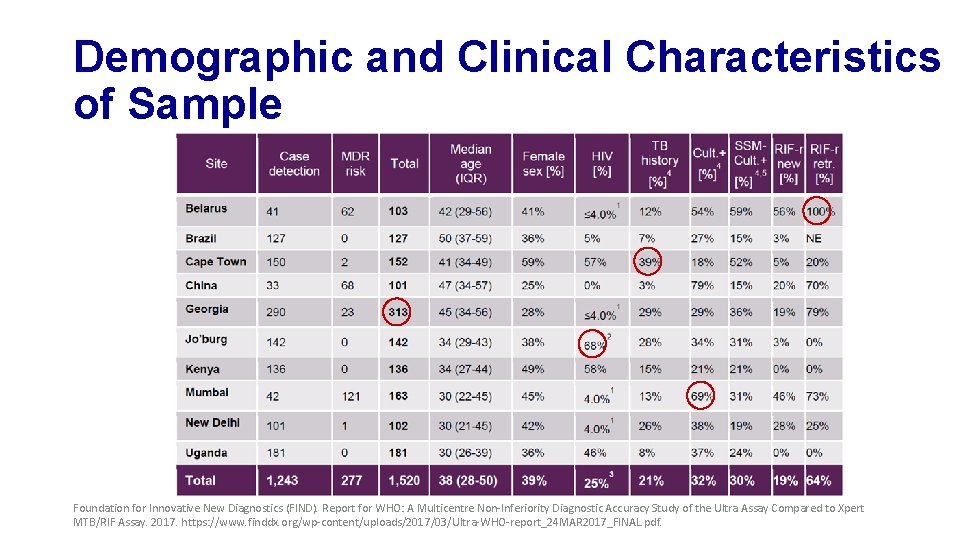
Demographic and Clinical Characteristics of Sample Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Sensitivity and Specificity of Xpert and Ultra for MTB Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
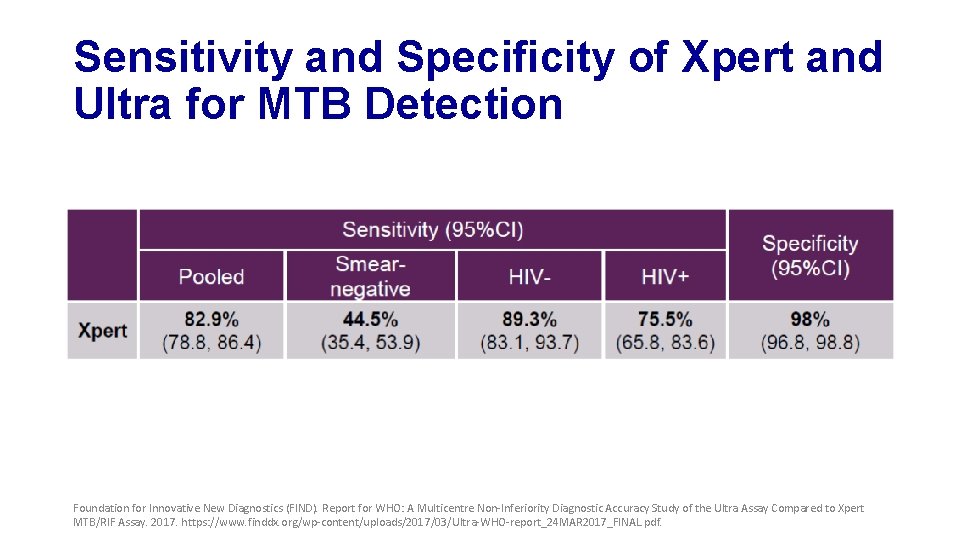
Sensitivity and Specificity of Xpert and Ultra for MTB Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
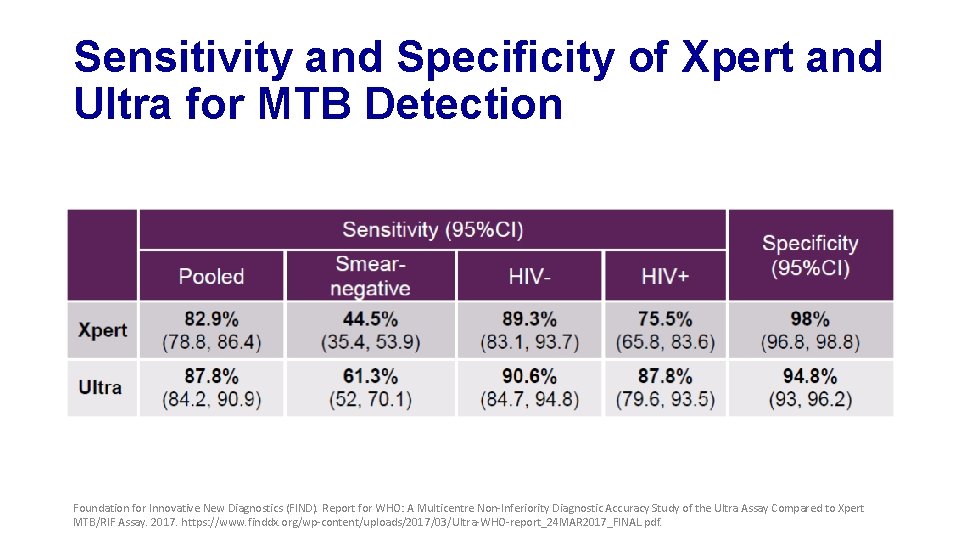
Sensitivity and Specificity of Xpert and Ultra for MTB Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
Sensitivity and Specificity of Xpert and Ultra for RIF Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
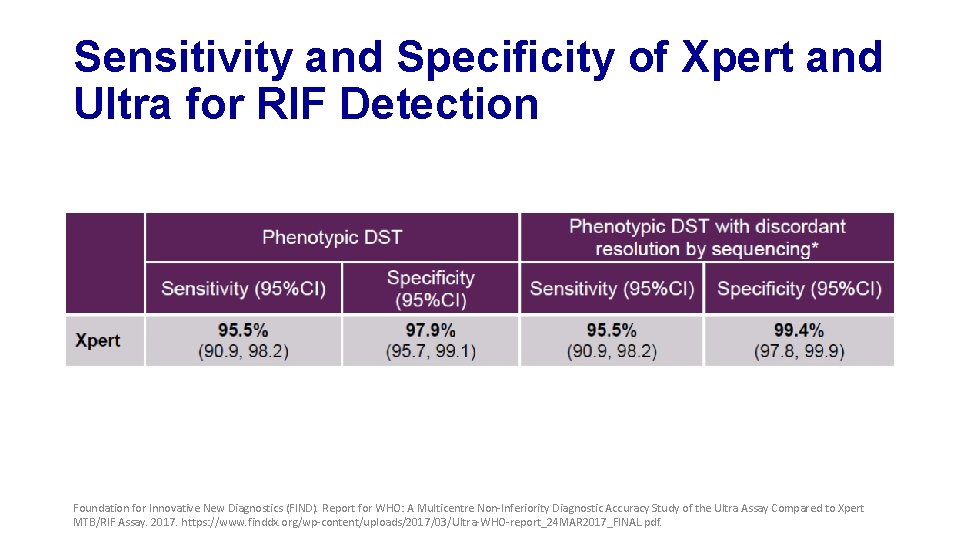
Sensitivity and Specificity of Xpert and Ultra for RIF Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
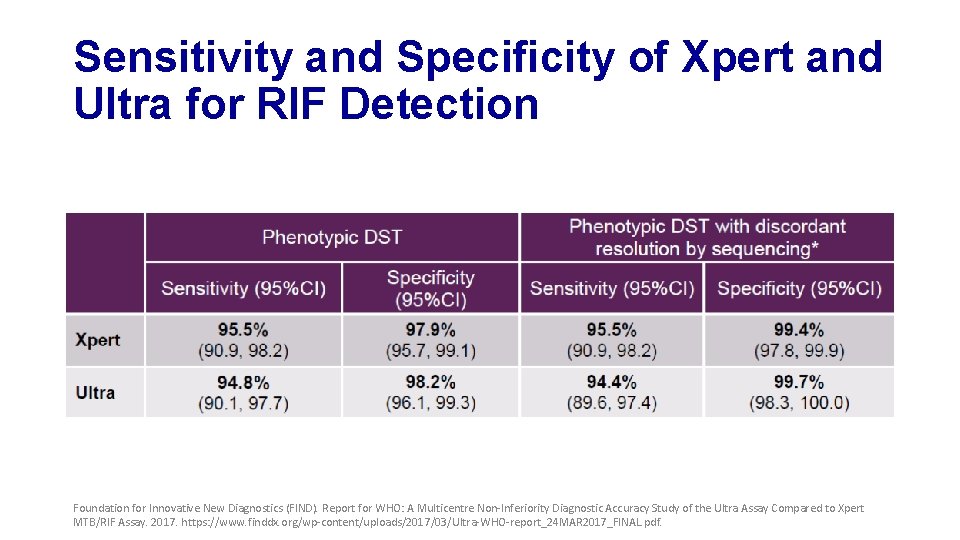
Sensitivity and Specificity of Xpert and Ultra for RIF Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Specificity Overall and by Treatment History Status Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
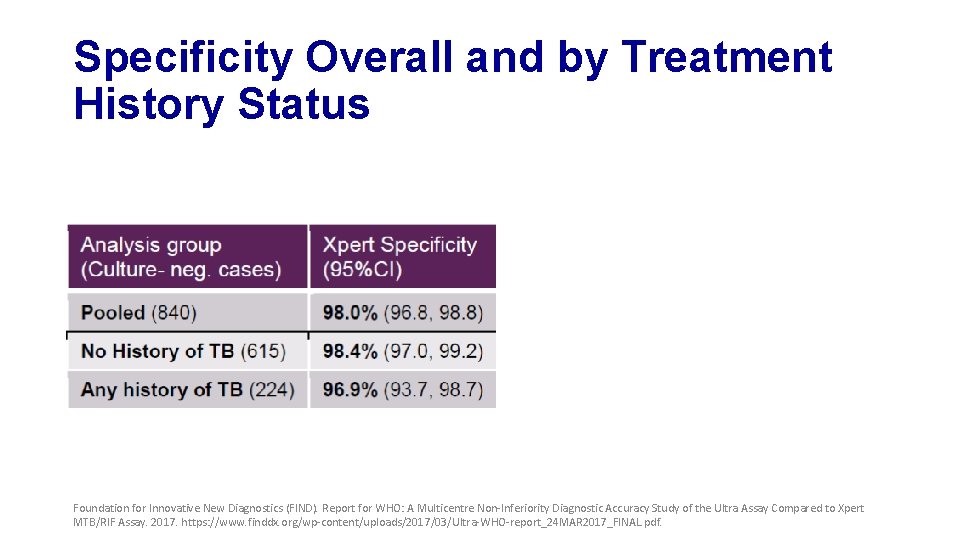
Specificity Overall and by Treatment History Status Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
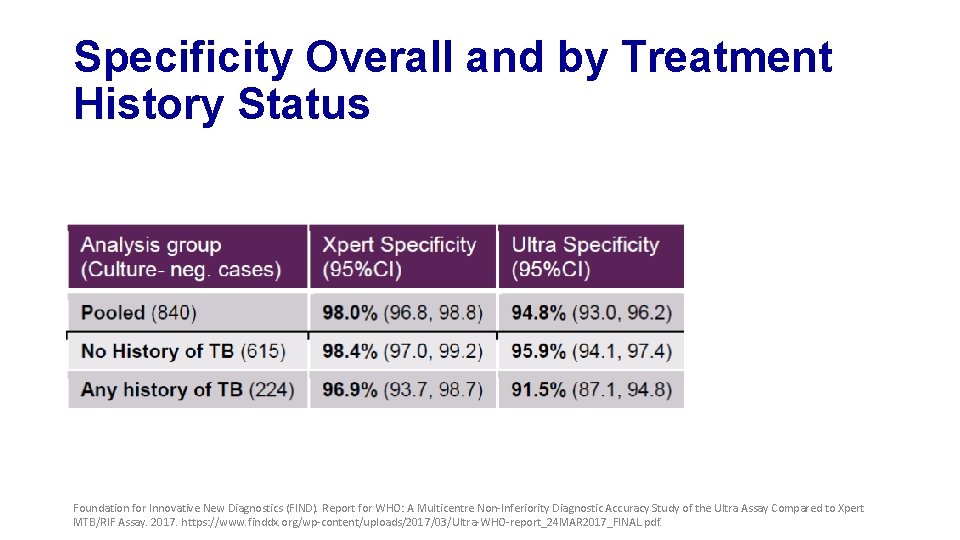
Specificity Overall and by Treatment History Status Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
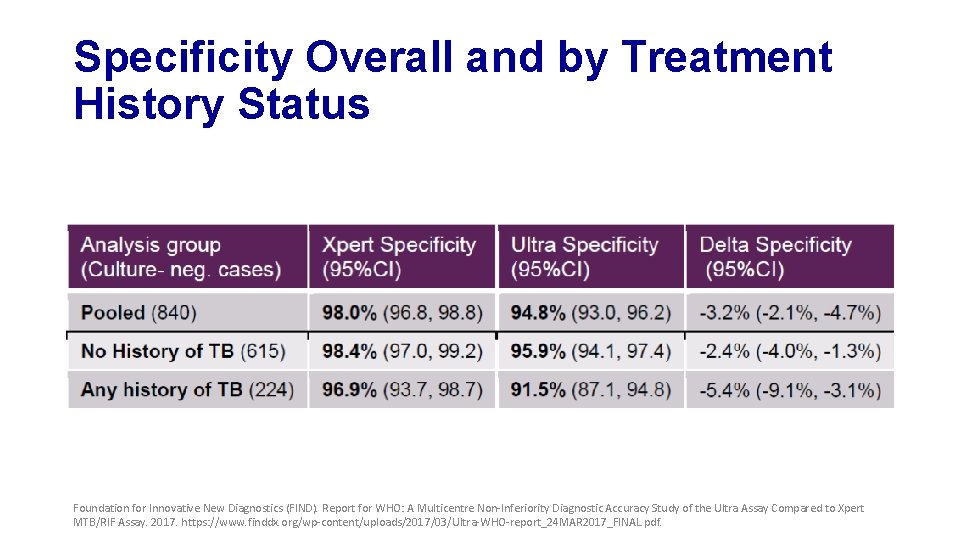
Specificity Overall and by Treatment History Status Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Non-inferiority Analysis for MTB Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Non-inferiority Analysis for MTB Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
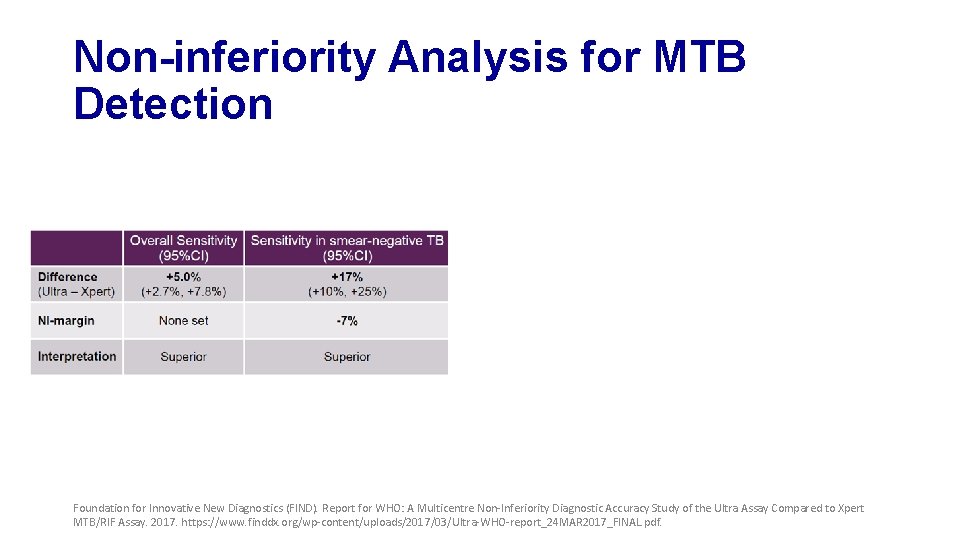
Non-inferiority Analysis for MTB Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
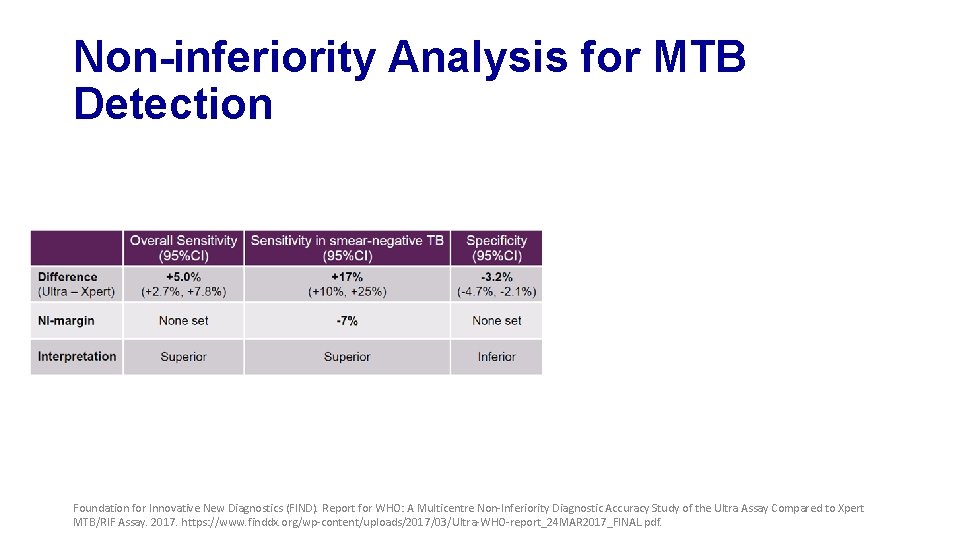
Non-inferiority Analysis for MTB Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
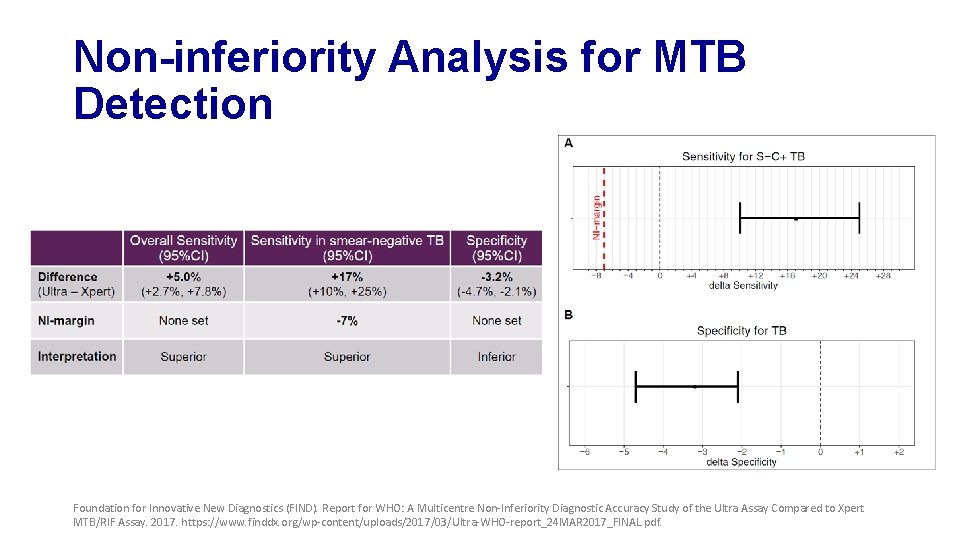
Non-inferiority Analysis for MTB Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
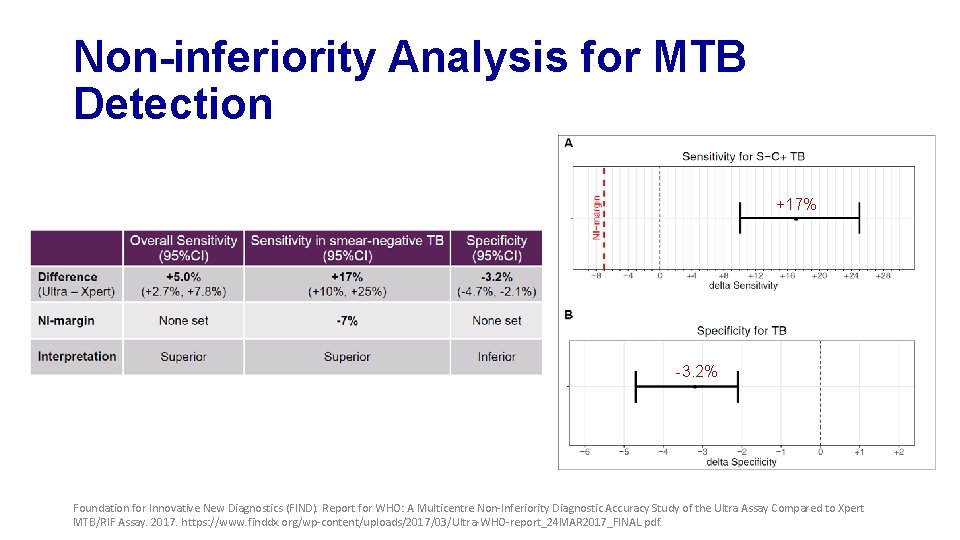
Non-inferiority Analysis for MTB Detection +17% -3. 2% Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Non-inferiority Analysis for RIF Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
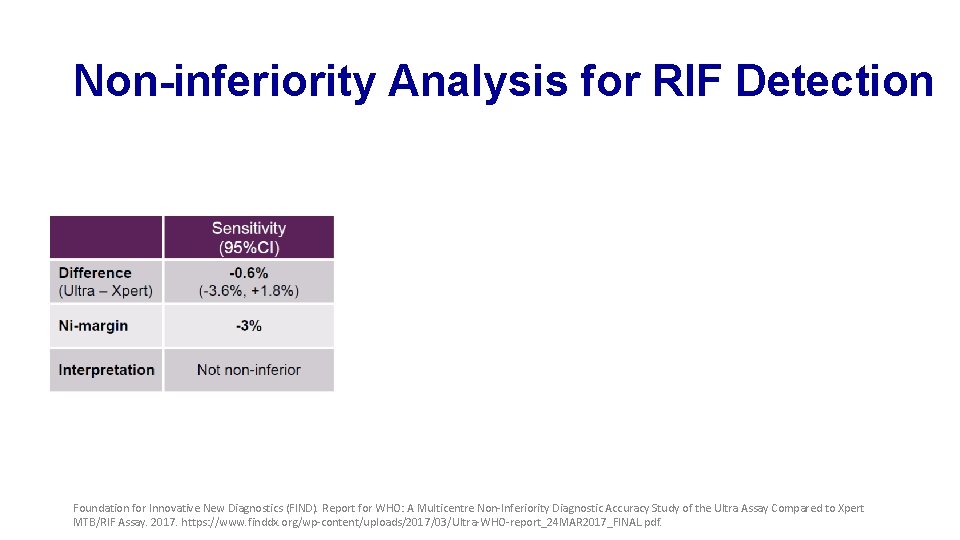
Non-inferiority Analysis for RIF Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
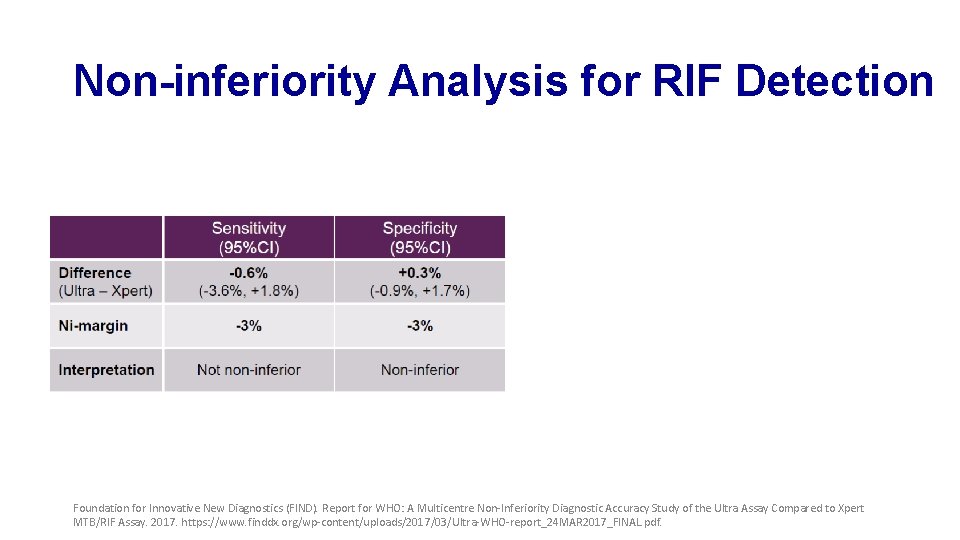
Non-inferiority Analysis for RIF Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
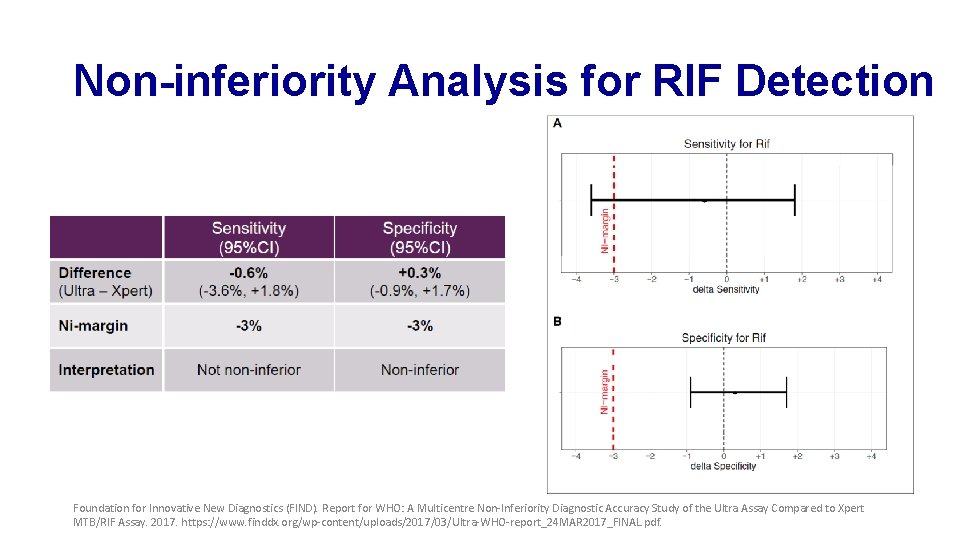
Non-inferiority Analysis for RIF Detection Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
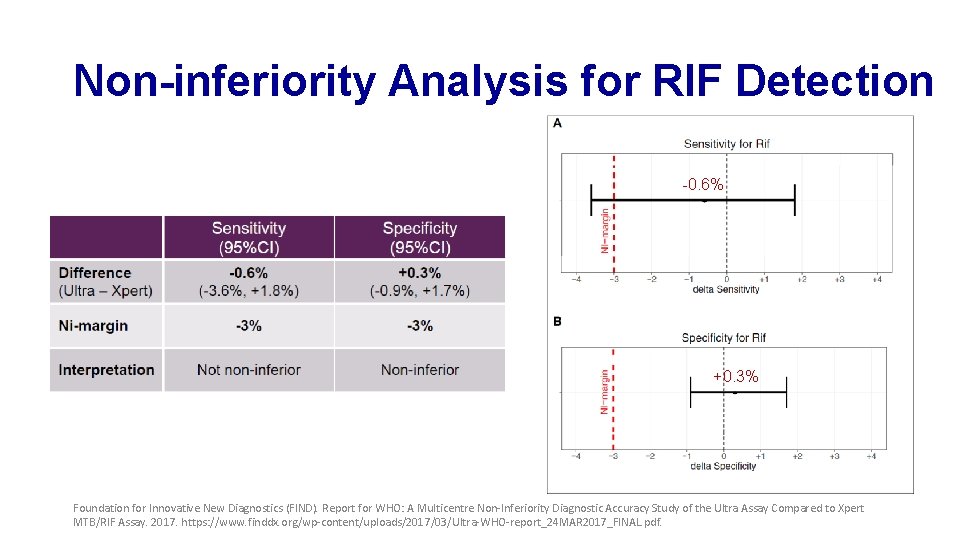
Non-inferiority Analysis for RIF Detection -0. 6% +0. 3% Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Primary Analysis Findings • MTB detection • Ultra shows improved sensitivity but reduced specificity • Performance for RIF detection is very similar • Non-inferiority met for specificity, but missed by a small margin for sensitivity Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Primary Analysis Findings • MTB detection • Ultra shows improved sensitivity but reduced specificity • Performance for RIF detection is very similar • Non-inferiority met for specificity, but missed by a small margin for sensitivity Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
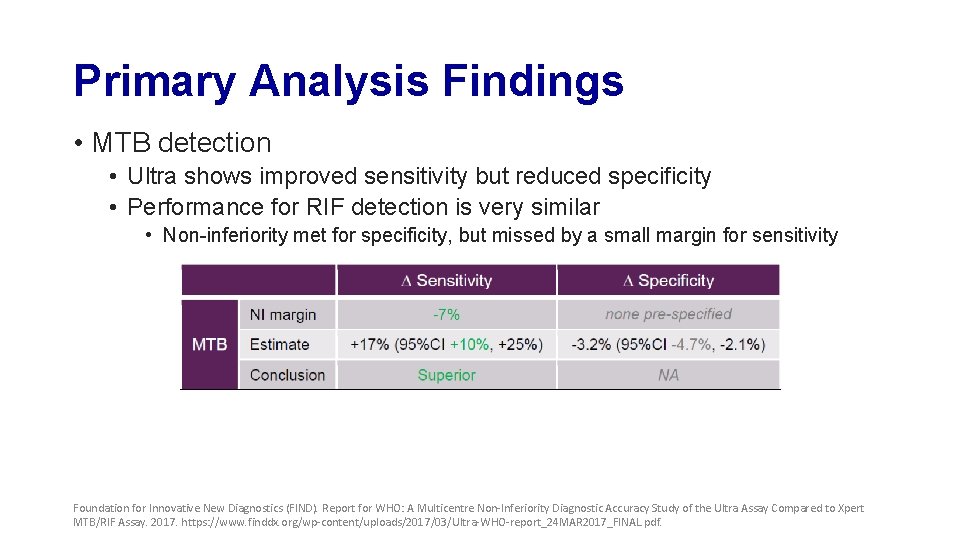
Primary Analysis Findings • MTB detection • Ultra shows improved sensitivity but reduced specificity • Performance for RIF detection is very similar • Non-inferiority met for specificity, but missed by a small margin for sensitivity Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
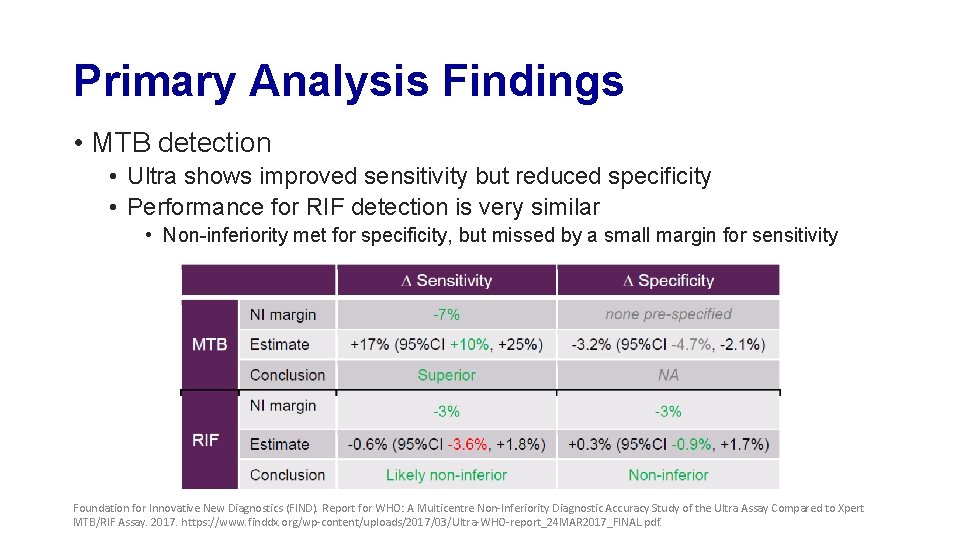
Primary Analysis Findings • MTB detection • Ultra shows improved sensitivity but reduced specificity • Performance for RIF detection is very similar • Non-inferiority met for specificity, but missed by a small margin for sensitivity Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Secondary Analysis Findings • Specificity of both Xpert and Ultra are affected by TB history • Stronger for Ultra and for recent history • Trade-off of higher sensitivity is lower specificity of Ultra • “trace-calls” • Reclassification of “trace-calls” as ‘TB-negative’ mitigates specificity losses while maintaining some of the sensitivity gains • To be considered for all patients or for patients with TB history • Re-testing of “trace-call” results with a second Ultra is another possibility to mitigate specificity losses leading to similar results as reclassification state above Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Other Secondary Analysis Findings • Analyses by site: • Expected variability of sensitivity-estimates between sites due to small sample • Specificity-estimates broadly consistent • Root cause analysis: • FPs on sample 1 mainly due to non-viable bacilli prior TB • Lower specificity-estimates for samples 2 and 3 versus sample 1 is likely due cross-contamination • Cross-contamination was due to procedures that were performed in certain sites (i. e. , beads homogenization) Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Conclusions • Ultra offers opportunities for increased TB detection • Ultra has increased sensitivity for TB detection including patients with paucibacillary disease • Ultra has decreased specificity in patients with prior history of TB • Ultra implementation needs to be considered carefully due to reduced specificity Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Additional Data Published with Main Study: CE-mark, Extrapulmonary TB, Elimination in non-HBDC, and Pediatric Cases
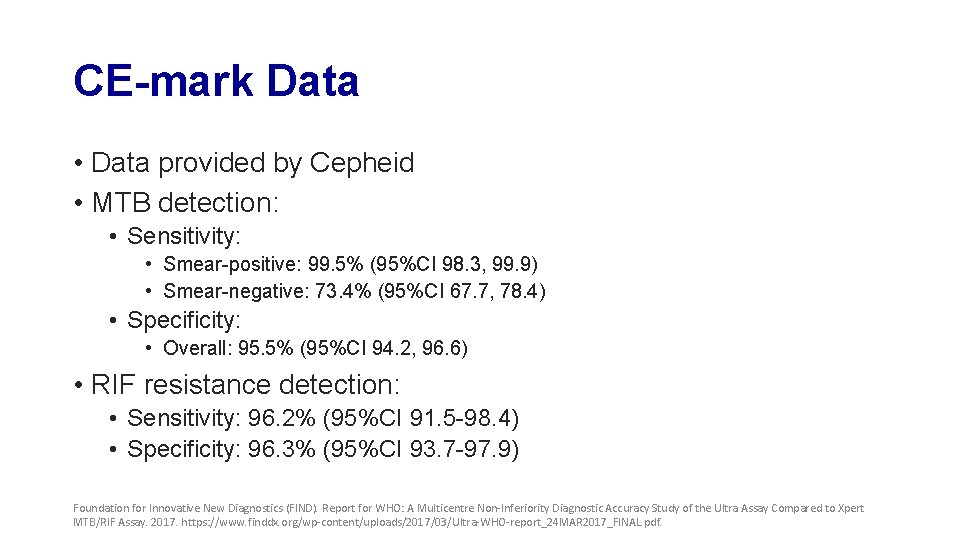
CE-mark Data • Data provided by Cepheid • MTB detection: • Sensitivity: • Smear-positive: 99. 5% (95%CI 98. 3, 99. 9) • Smear-negative: 73. 4% (95%CI 67. 7, 78. 4) • Specificity: • Overall: 95. 5% (95%CI 94. 2, 96. 6) • RIF resistance detection: • Sensitivity: 96. 2% (95%CI 91. 5 -98. 4) • Specificity: 96. 3% (95%CI 93. 7 -97. 9) Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Extra-pulmonary TB • TB meningitis • Sensitivity: • Ultra: 95% • Xpert 45% • Culture 45% • Conclusion: • Ultra detected significantly more TB meningitis than either Xpert or culture • Adequate CSF testing volume is critical Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Pediatric Study South Africa • Culture and Ultra • Sensitivity: 75. 7% • Specificity: 96. 5% • HIV-infected • Sensitivity: 70. 6% • Specificity: 98. 2% • HIV-uninfected • Conclusion: • Ultra provides rapid detection of MTB from a single induced sputum in most children with culture confirmed TB • Ultra may detect an additional group of children with TB, who are not detected by culture • Sensitivity: 77. 2% • Specificity: 96. 1% Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Evaluation of the Ultra Assay in a Pediatric TB Study from Tanzania Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Evaluation of the Ultra Assay in a Pediatric TB Study from Tanzania Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.
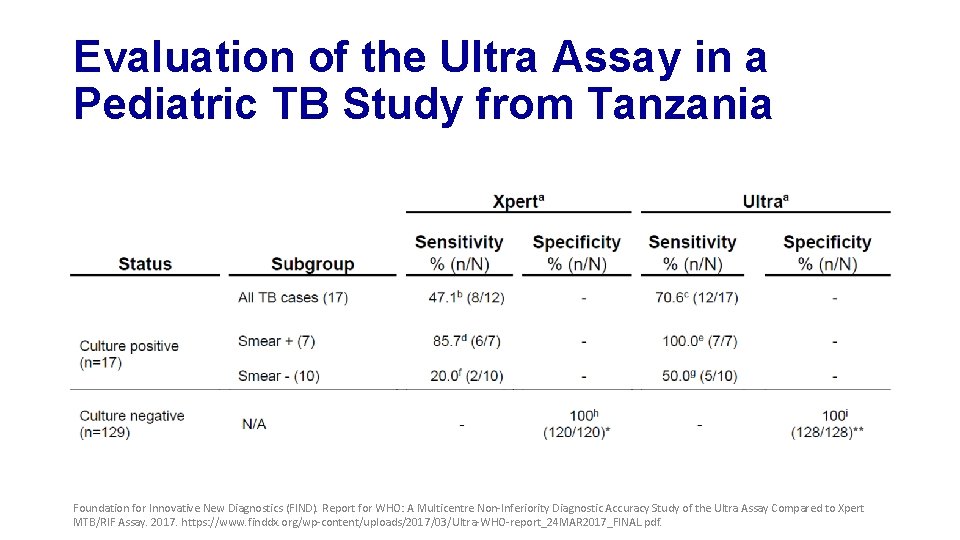
Evaluation of the Ultra Assay in a Pediatric TB Study from Tanzania Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

Summary of Findings • Shows a significant increase of sensitivity for Ultra in TB meningitis • Suggests an increase in sensitivity in paucibacillary disease (i. e. , children and active case finding in asylum seekers) • Sensitivity increase is attributable to Ultra “trace call” Foundation for Innovative New Diagnostics (FIND). Report for WHO: A Multicentre Non-Inferiority Diagnostic Accuracy Study of the Ultra Assay Compared to Xpert MTB/RIF Assay. 2017. https: //www. finddx. org/wp-content/uploads/2017/03/Ultra-WHO-report_24 MAR 2017_FINAL. pdf.

WHO Position Regarding Xpert® MTB/RIF Ultra • Ultra assay is non-inferior to Xpert assay for the diagnosis of MTB and detection of RIF resistance • Ultra can be used as an alternative to Xpert in all settings • Ultra can be used as the initial diagnostic test for all adults and children with signs and symptoms of TB • Ultra can be used in the testing of selected extra pulmonary specimens (CSF, lymph nodes and tissue specimens) • Interpretation of Ultra is similar to Xpert except for “trace call” WHO meeting report of a technical expert consultation: non-inferiority analysis of Xpert MTF/RIF Ultra compared to Xpert MTB/RIF. Geneva: World Health Organization; 2017 (WHO/HTM/TB/2017. 04). Licence: CC BY-NCSA 3. 0 IGO.

Recent Published Studies ® Using the Xpert MTB/RIF Ultra Assay

Lancet Infect Dis. 2018 Jan; 18(1): 76 -84. doi: 10. 1016/S 1473 -3099(17)30691 -6. Epub 2017 Nov 30.

Lancet Infect Dis. 2018 Jan; 18(1): 76 -84. doi: 10. 1016/S 1473 -3099(17)30691 -6. Epub 2017 Nov 30.
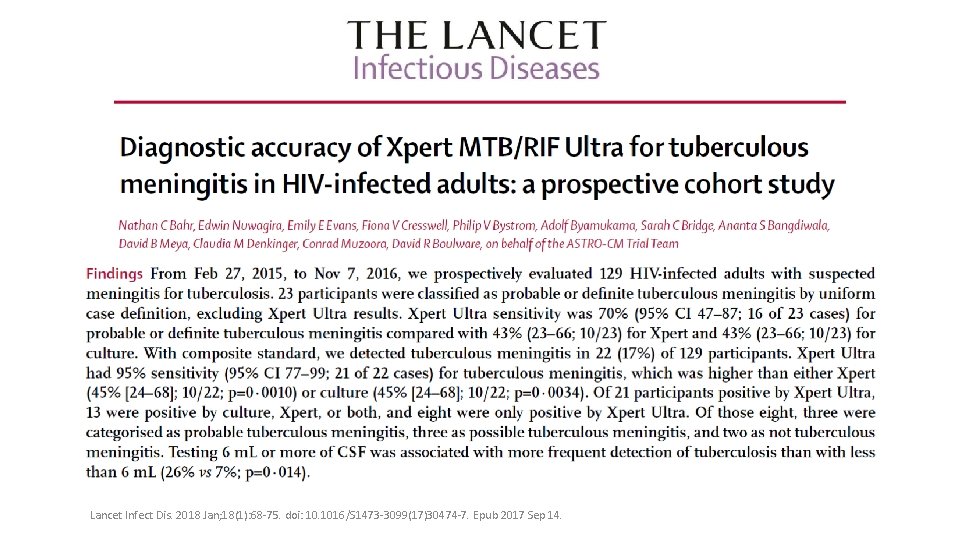
Lancet Infect Dis. 2018 Jan; 18(1): 68 -75. doi: 10. 1016/S 1473 -3099(17)30474 -7. Epub 2017 Sep 14.

Lancet Infect Dis. 2018 Jan; 18(1): 68 -75. doi: 10. 1016/S 1473 -3099(17)30474 -7. Epub 2017 Sep 14.

PLo. S Med. 2017 Dec 14; 14(12): e 1002472. doi: 10. 1371/journal. pmed. 1002472. e. Collection 2017 Dec.

PLo. S Med. 2017 Dec 14; 14(12): e 1002472. doi: 10. 1371/journal. pmed. 1002472. e. Collection 2017 Dec.

Pediatr Infect Dis J. 2018 Feb 22. doi: 10. 1097/INF. 0000001960. [Epub ahead of print]

Pediatr Infect Dis J. 2018 Feb 22. doi: 10. 1097/INF. 0000001960. [Epub ahead of print]

Xpert® MTB/RIF Ultra: The Next-Generation Assay for Tuberculosis Diagnosis and Rifampin Resistance Detection Diego P. Peralta, M. D. , FACP Assistant Professor Division of Infectious Diseases March 22, 2018 Texas Association for Clinical Laboratory Science
- Slides: 78