Xenobiotics and their Biodegradation Antoine Lebel L Definition

Xenobiotics and their Bio-degradation Antoine Lebel L.

Definition 1948: 3 insecticides were in use in USA Foreign to the organism under study. Synthetic novel compounds(artificially produced). Pesticides – pesticide usage- persistence - no biological transformation. (Chlorophenoxy alkyl carboxylic acid, substituted ureas, nitrophenols, In 1975 , 1170 pesticides; triazines, phenyl carbamates, organochlorines, 425 herbicides; 410 organophosphates etc. , ). fungicides; 335 insecticides Organic pesticides and synthetic were registered for the use in that country pesticides

Environmental persistence of some pesticides Persistence Aldrin Chlordane DDT Endrin Heptachlor Lindane Parathion PCP Picloram Simazone 2, 4, 5 -T Toxaphene Persistence time > 15 years > 14 years > 15 years > 16 years > 5 years > 190 days > 14 years 2 years

Environmental persistence of some pesticides Organic pesticides are biodegradable: 3 major natural mechanisms 1. Auto-oxidation 2. Degradation by sunlight 3. Microbial degradation Microbes are unique tools for biodegradation or detoxification 1. Fermentation 2. Anaerobic metabolism 3. Chemo-lithotrophic metabolism 4. Metabolism through exo-enzymes 5. Adaptability to adverse conditions through mutation A Biomagnified pesticide may cause death or serious problem to the organism. They have higher lipo-solubility and lower water solubility Biomagnification Osprey 20 ppm L. Fish 3 ppm Small fish 0. 3 ppm Plankton 30 ppb Aquatic Environment 0. 3 ppb

Xenobiotics are recalcitrant-Reasons • • • Unusual bonds or bond sequences. Unusual substitutions(Chlorine and other halogens). Highly condensed aromatic ring. Excessive molecular size( as in plastics). Failure of a compound to induce synthesis of degrading enzymes. • Failure of a compound to enter the microbial cell for lack of suitable permeases. • Unavailability of the compound due to insolubility or adsoprtion phenomenon. • Excessive toxicity of the parent compound or its metabolic products.

Biodegradation -Objectives • How biodegradation activity arises , evolves and transferred among the members of soil micro-flora? • To device bioremediation methods for removing or detoxifying high concentration of dangerous pesticide residues.

Genes for location. catabolic Breakdown – Understanding of the characters (genes) provides clues to the evolution of degradative pathways and makes the task of gene manipulation easier to construct the genetically engineered microbes capable of degrading the pollutants.
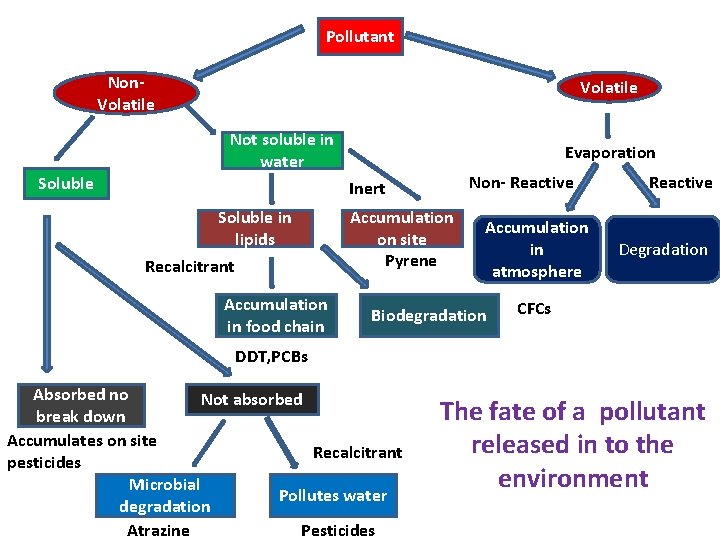
Pollutant Non. Volatile Not soluble in water Soluble Evaporation Non- Reactive Inert Soluble in lipids Recalcitrant Accumulation in food chain Accumulation on site Pyrene Accumulation in atmosphere Biodegradation Reactive Degradation CFCs DDT, PCBs Absorbed no Not absorbed break down Accumulates on site Recalcitrant pesticides Microbial Pollutes water degradation Atrazine Pesticides The fate of a pollutant released in to the environment
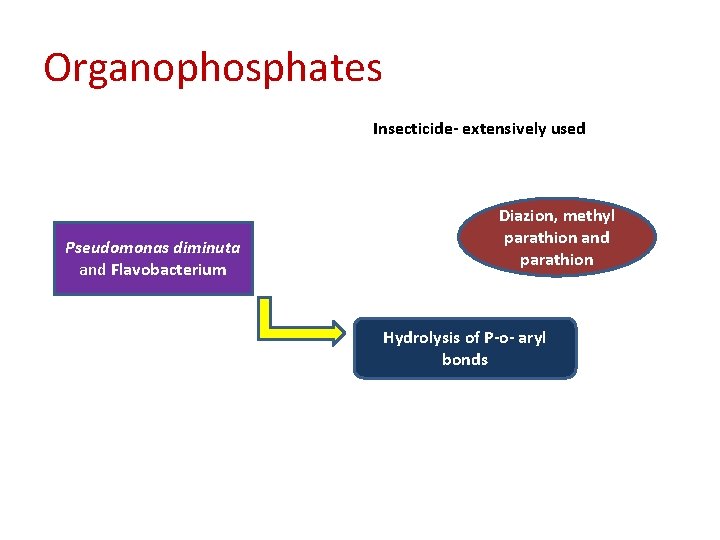
Organophosphates Insecticide- extensively used Pseudomonas diminuta and Flavobacterium Diazion, methyl parathion and parathion Hydrolysis of P-o- aryl bonds
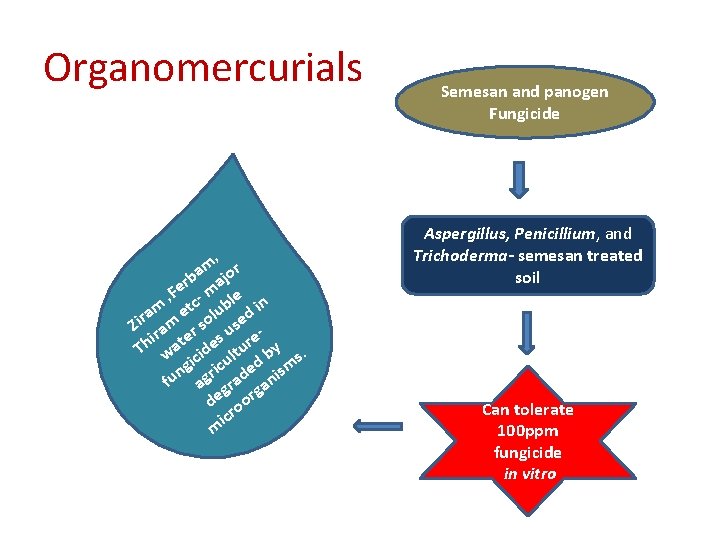
Organomercurials , m r ba ajo r , Fe c- m le n m et lub d i a r Zi ram r so use i e s e Th wat cide ltur by s. gi ricu ed ism n fu ag grad an g de oor icr m Semesan and panogen Fungicide Aspergillus, Penicillium, and Trichoderma- semesan treated soil Can tolerate 100 ppm fungicide in vitro
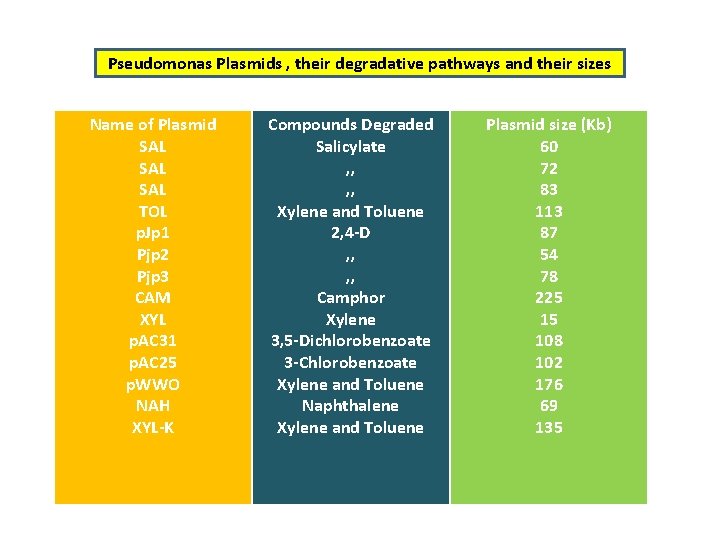
Pseudomonas Plasmids , their degradative pathways and their sizes Name of Plasmid SAL SAL TOL p. Jp 1 Pjp 2 Pjp 3 CAM XYL p. AC 31 p. AC 25 p. WWO NAH XYL-K Compounds Degraded Salicylate , , Xylene and Toluene 2, 4 -D , , Camphor Xylene 3, 5 -Dichlorobenzoate 3 -Chlorobenzoate Xylene and Toluene Naphthalene Xylene and Toluene Plasmid size (Kb) 60 72 83 113 87 54 78 225 15 108 102 176 69 135
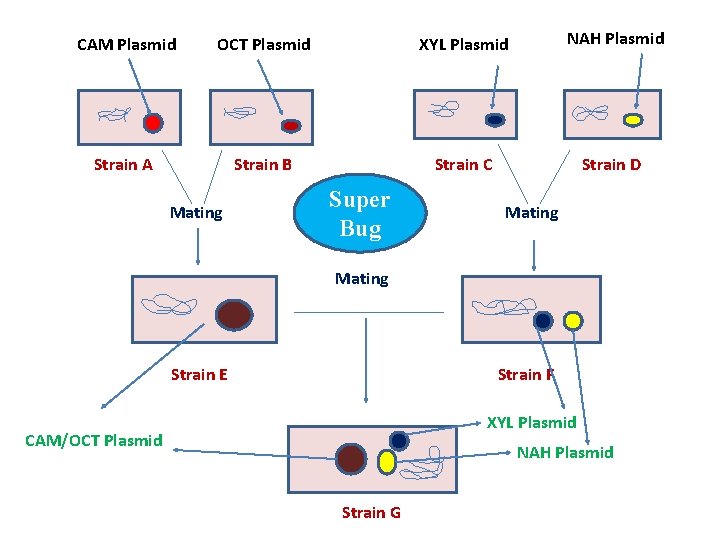
CAM Plasmid OCT Plasmid XYL Plasmid NAH Plasmid Strain A Strain B Strain C Strain D Mating Super Bug Mating Strain E Strain F XYL Plasmid CAM/OCT Plasmid NAH Plasmid Strain G

Phenols Biodegradation

Phenols v Aromatic – Recalcitrant Contaminants. v Imparts objectionable taste and odour to the drinking water when it combines with chlorine to form chlorophenol. v Phenol have high oxygen demand – in high concentration can deplete the oxygen of the receiving water body. v Sources: Oil refineries, chemical plants, explosive manufacturing, resin manufacturing, and cake ovens are the sources of phenol, polyphenols, chlorophenols and phenoxyacids. v Rubber reclamation plant, fibre glass manufacturing and textile mills are also the source for phenol contamination. v Oil and cyanide are common in phenolic waste

Aerobic degradation and pathways of aromatic ring cleavage • The basic reaction sequences involved are 1. Transformation of side chains. 2. Formation of ring fission substrate. 3. Ring cleavage and transformation of the products of ring fission in to common compounds of intermediary metabolism.

Biodegradation of Phenol Aerobic condition Absence of oxygen • Molecular oxygen is required as the terminal electron acceptor during respiration. • Microorganisms necessarily have evolved different mechanisms • And also for inserting the compounds for ring fission. during hydroxylation and ring cleavage (Oxygen is incorporated in the aromatic ring). • Oxygenase enzymes(mono and dioxygenases) mediate these reactions.

ØBacteria, Fungi and Actinomycetes – Responsible organisms. ØHow? By producing mono or dioxygenases. ØWhat is the role of mono and dioxygenases? ØIncorporate single or two oxygen molecule to form reduced central intermediary, catechol. ØSpores of Fusarium flocciferum can be used to degrade phenol following immobilization on diatomic particles. ØPseudomonas putida isolated from mangrove forest soil and Nocardia sp. and Bacillus circulans were selected as a known representative of the aerobic degraders of aromatics

Cleavage of Aromatic Ring • The aerobic cleavage of aromatic ring is achieved through dioxygenases by three mechanisms 1. Ortho cleavage 2. Meta cleavage 3. Gentisic acid pathways
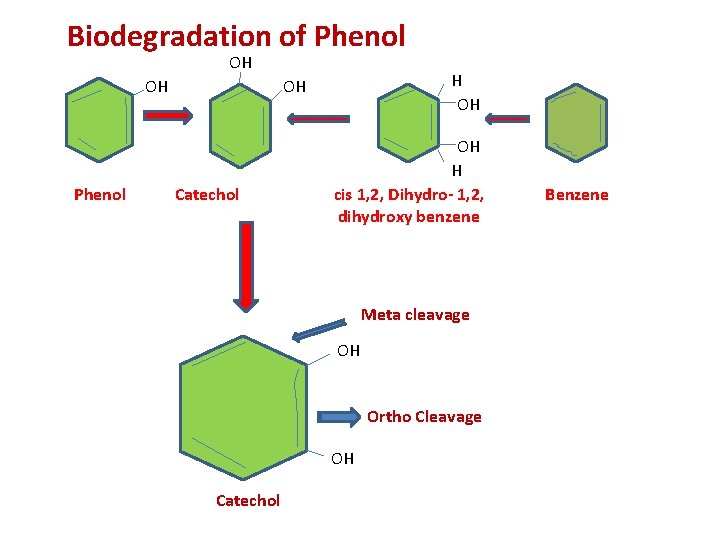
Biodegradation of Phenol OH OH Phenol H OH OH Catechol OH H cis 1, 2, Dihydro- 1, 2, dihydroxy benzene Meta cleavage OH Ortho Cleavage OH Catechol Benzene
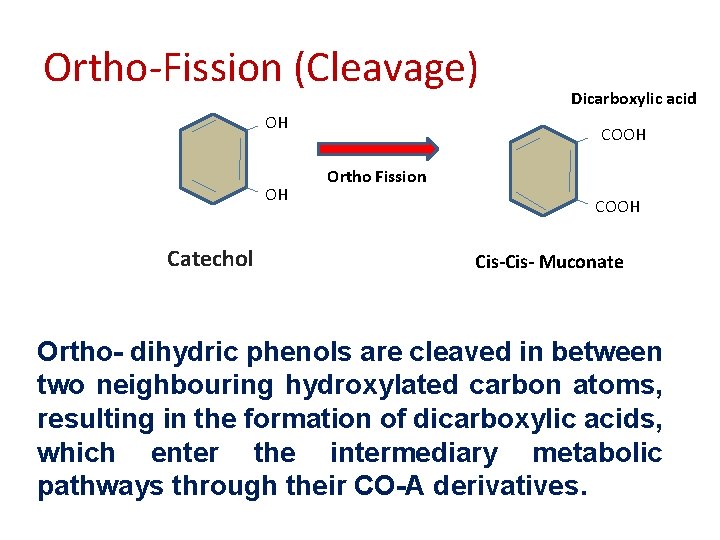
Ortho-Fission (Cleavage) OH OH Catechol Dicarboxylic acid COOH Ortho Fission COOH Cis- Muconate Ortho- dihydric phenols are cleaved in between two neighbouring hydroxylated carbon atoms, resulting in the formation of dicarboxylic acids, which enter the intermediary metabolic pathways through their CO-A derivatives.
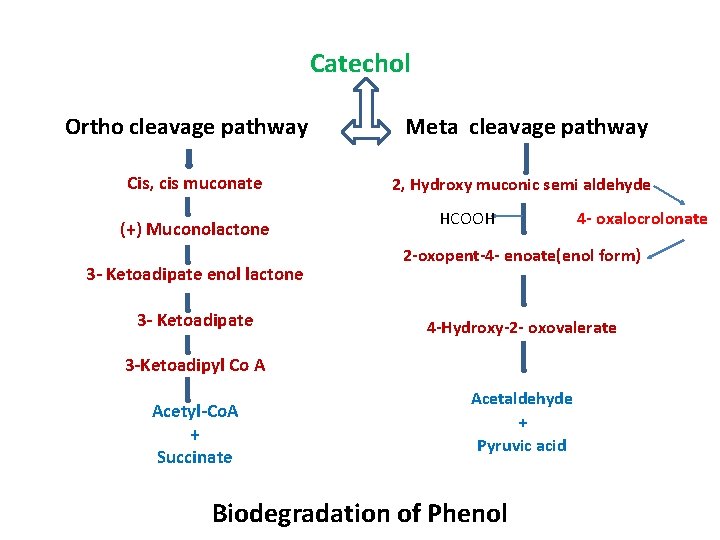
Catechol Ortho cleavage pathway Cis, cis muconate (+) Muconolactone 3 - Ketoadipate enol lactone 3 - Ketoadipate Meta cleavage pathway 2, Hydroxy muconic semi aldehyde HCOOH 2 -oxopent-4 - enoate(enol form) 4 -Hydroxy-2 - oxovalerate 3 -Ketoadipyl Co A Acetyl-Co. A + Succinate 4 - oxalocrolonate Acetaldehyde + Pyruvic acid Biodegradation of Phenol

Meta Fission(Cleavage) Meta fission is the cleavage of the ring between the hydroxylated and non- hydroxylated carbon atoms. Gentisic Acid Pathway It is cleaved at the carbon- carbon bond between the carboxyl groups and the neighboring hydroxyl group to give maleyl pyruvic acid followed by a glutathione dependent isomerisation to form fumaric and pyruvic acids.

Pentachloropheno l

OH Cl Cl PENTACHLOROPHENOL Cl Cl (Chlorinated aromatic compound) Cl Broad spectrum Biocide. Introduced as wood preservatives during the year 1930 s. General purpose killer.

ØToxicity and degree of chlorination. ØDegrading microorganisms: Arthrobacter sp. Pseudomonas cepacia and Flavobacterium sp. ØCan degrade few not all chlorinated phenol. ØFlavobacterium sp. from PCP contaminated dump sites.

v. Bioreactors - Alginate immobilized + Polyurethane foam immobilized PCP degrading Flavobacterium (ATCC 39723). v. PCP from contaminated water. v. Adsorption of PCP by immobilized matrixreducing the toxicity. v. Carbon- chlorine bond is very strong. Cell extracts of Arthrobacter- Dehalogenate PCP. Actinomycetes and fungal organisms- metabolize PCP.
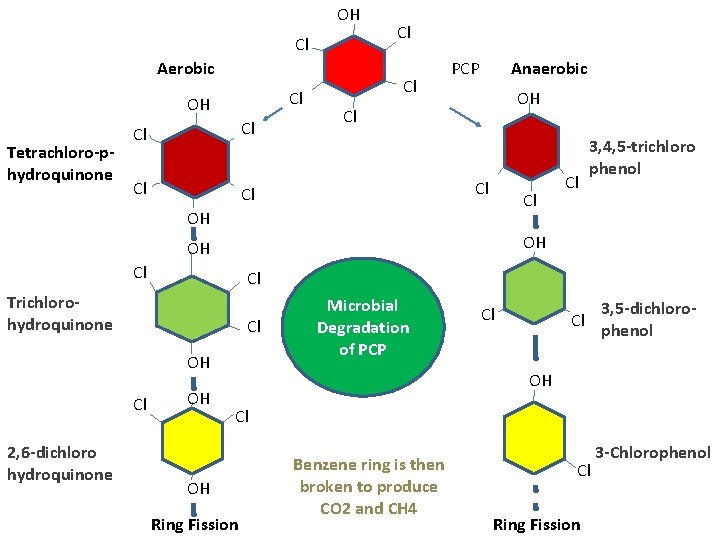
OH Cl Aerobic Cl OH Tetrachloro-phydroquinone Cl Cl Cl PCP Anaerobic OH Cl Cl Cl OH OH OH Cl Cl Trichlorohydroquinone Cl OH Cl 2, 6 -dichloro hydroquinone Cl 3, 4, 5 -trichloro phenol OH Microbial Degradation of PCP Cl Cl 3, 5 -dichlorophenol OH Cl OH Ring Fission Benzene ring is then broken to produce CO 2 and CH 4 Cl Ring Fission 3 -Chlorophenol

Polychlorinated Biphenyls (PCBs)

Polychlorinated Biphenyls(PCBs) • Biphenyls with one to ten chlorine atom per molecule. • Properties: Oily fluids with high boiling points Great chemical resistance Low electrical conductivity High refractive index First described in 1881 PCBs , a group of 209 synthetic halogenated aromatic hydrocarbons

Solubility Substance Biphenyl Monochlorobiphenyls Dichlorobiphenyls Hexachlorobiphenyls Solubility (mg/l) 7. 5 1 -6 0. 085 -5 0. 00095 PCBs are water insoluble , lipid soluble and inert fluids. Any chlorine substituent decreases the water solubility.
Uses eat h – ble eat a m flam oils in h n o N nt ms a e t t s s i y res er s f s n tra Plasticizers in food packaging material Hydraulic fluids nts a c i br Lu eum l o r Pet itives add Tran sfo fluid rmer cap s in acit ors
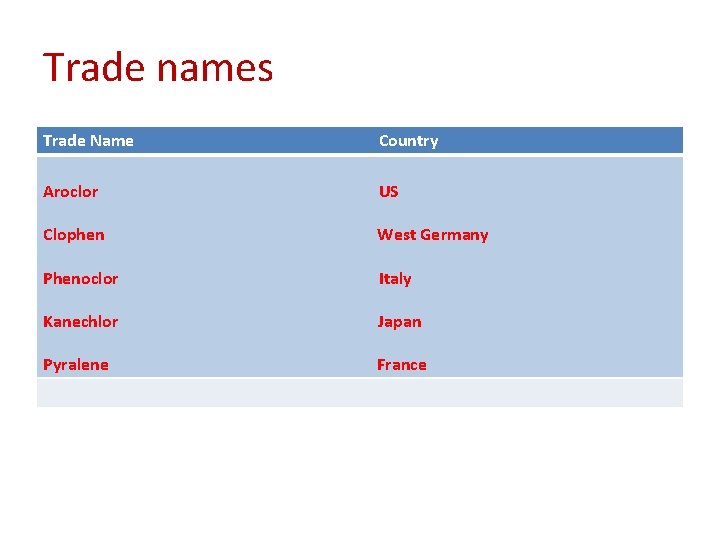
Trade names Trade Name Country Aroclor US Clophen West Germany Phenoclor Italy Kanechlor Japan Pyralene France
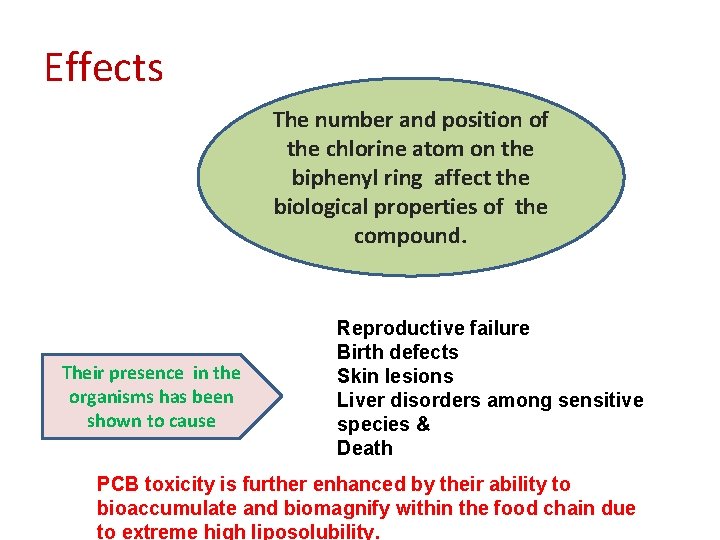
Effects The number and position of the chlorine atom on the biphenyl ring affect the biological properties of the compound. Their presence in the organisms has been shown to cause Reproductive failure Birth defects Skin lesions Liver disorders among sensitive species & Death PCB toxicity is further enhanced by their ability to bioaccumulate and biomagnify within the food chain due to extreme high liposolubility.

Rice oil Factory Accident in Japan in 1968 brought PCBs international attention. The heat exchanger pipes used to process rice oil contained PCBs as the heat exchange fluid. Unnoticed, a heat exchange pipe broke and leaked PCBs into a batch of rice oil. Packaged and consumed by the local population. The contaminated rice oil poisoned over 1000 people, producing a spectrum of symptoms including v v v and v Gum and nail bed discoloration Joint swelling Emission of waxy secretions from eyelid glands Lethargy

Rules governing the use of PCBs Food and Drug Administration (FDA- US) Issued tolerance levels for PCBs in food and packaging products Environmental Protection Agency (EPA) Issued rules governing the use of PCBs This has drastically reduced domestic production and use of PCBs. Despite, decreased use, PCBs still pose an environmental Problem – chemically stable and also resist biodegradation. Because Past use of PCBs has been high- accumulated in the environment.
DDT PCBs

Biodegradation of PCBs • Microbial degradation of PCBs depends on the degree of chlorination and the position of the chlorine atom on the biphenyl molecule. • Lower chlorinated biphenyls-readily transformed by bacteria. • Higher chlorinated compounds (with 5 or more chlorine) –persistent in the environment. • Tetrachloro biphenyl compounds- intermediate in persistence.

Oxidative and Reductive metabolic mechanisms The gene specifying the oxidative catabolic pathways of PCBs include four enzymes(Bacteria). Biphenyl dioxygenase Dihydrodiol dehydrogenase 3, 3 - phenylcatechol dioxygenase 4, 2 - hydroxyl-6 -oxo-6 -phenyl hexa-2, 4 diconate hydrolase
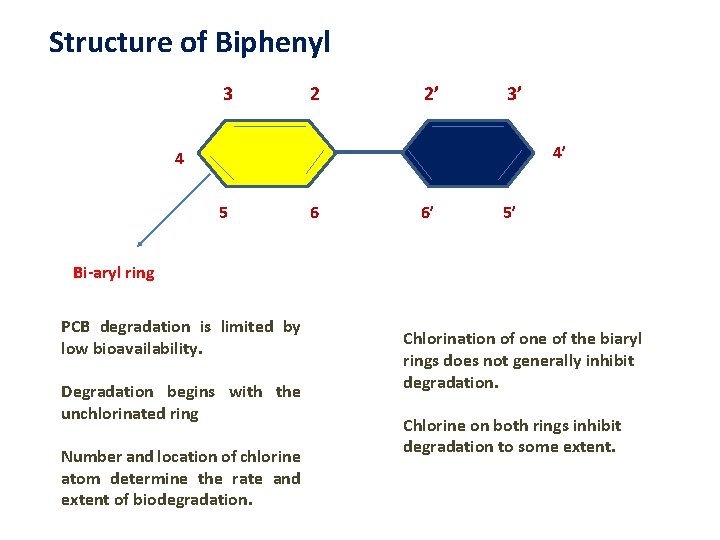
Structure of Biphenyl 3 2 2’ 3’ 4’ 4 5 6 6’ 5’ Bi-aryl ring PCB degradation is limited by low bioavailability. Degradation begins with the unchlorinated ring Number and location of chlorine atom determine the rate and extent of biodegradation. Chlorination of one of the biaryl rings does not generally inhibit degradation. Chlorine on both rings inhibit degradation to some extent.
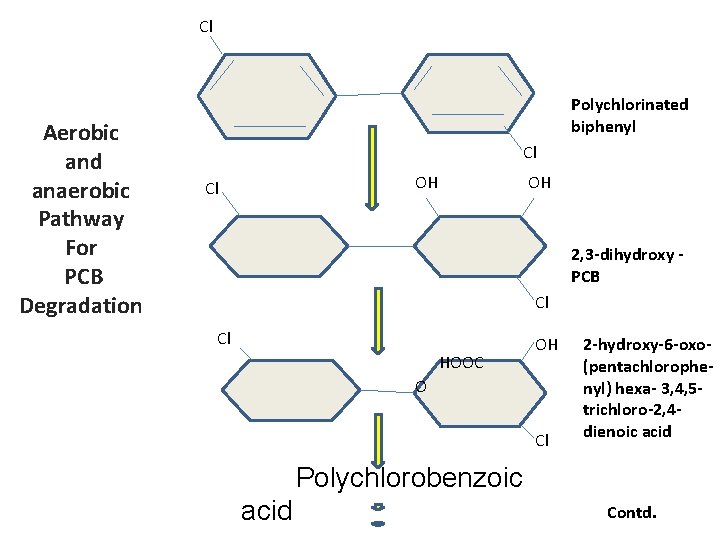
Cl Aerobic and anaerobic Pathway For PCB Degradation Polychlorinated biphenyl Cl OH 2, 3 -dihydroxy PCB Cl Cl HOOC OH O Cl 2 -hydroxy-6 -oxo(pentachlorophenyl) hexa- 3, 4, 5 trichloro-2, 4 dienoic acid Polychlorobenzoic acid Contd.
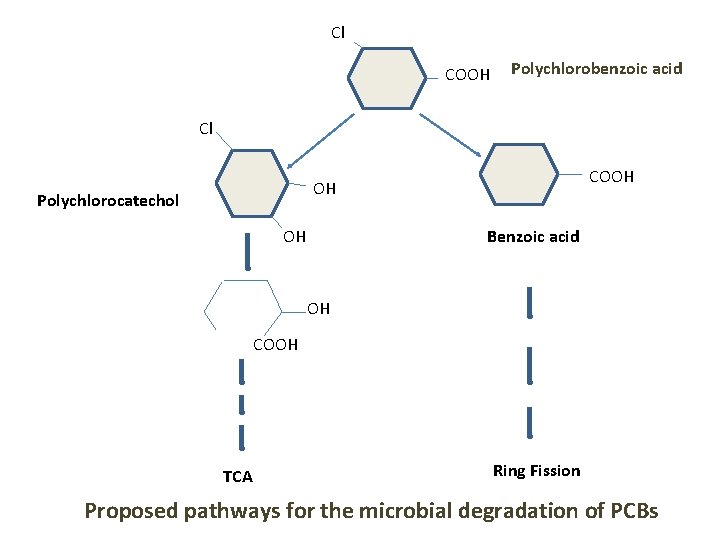
Cl COOH Polychlorobenzoic acid Cl COOH OH Polychlorocatechol OH Benzoic acid OH COOH TCA Ring Fission Proposed pathways for the microbial degradation of PCBs

Biodegradation -evidence Passage of PCBs through ASP in sewage treatment plants for 48 hrs. resulted in 81% degradation for Aroclor Dmochewitz et al. , reported as Aspergillus niger that was capable of utilizing mixtures of PCBs Eaton showed mineralization of PCBs by a lignocellulosic fungus Takase et a ioslate l. , d Pseud o cruciv monas ia that co e strain u on mo ld grow re biphen than 10 yl r compo elated unds. raded g e d s a way ther w h e t l a y p n e e g Biph cleava o h t r ugh a o o r n h a t h d g e throu egrad d s a w l way Bipheny cleavage path meta

Chaudhry and Chapalamaduger reported several microbes that are capable of degrading PCBs. Anaerobic degradation of PCB is carried out by the white rust fungus (Phanerochaete), Acinetobacter and Alcaligenes. Degradation of PCB typically is by cometabolism and is enhanced by the addition of less chlorinated analogs such as dichlorophenyl.

Strategies for promoting biodegradation of PCBs The use of a sequential anaerobic – aerobic process to remove chlorines and then allow mineralization of the less chlorinated isomers. The addition of a co-substrate , such as the parent PCB compound biphenyl to stimulate PCB degrading population……. Rationale for this strategy. Bio-augmentation or addition of specific PCB degrading microorganisms to contaminated areas.

Your Questions? ? ? . . . Thank you
- Slides: 45