www hivnet ubc ca Aslam H Anis Ph

www. hivnet. ubc. ca Aslam H. Anis. Ph. D, FCAHS National Director, Professor, University of British Columbia

www. hivnet. ubc. ca CTN’S MISSION STATEMENT The CTN is a Canada-wide partnership of researchers, caregivers, governments, health advocates, the innovative pharmaceutical and biotechnology industry, and people living with HIV who are committed to developing treatments, preventions and a cure for HIV and related health conditions, through the conduct of scientifically sound and ethical trials. The CTN is committed to maximizing the impact of research and ultimately improving the health of Canadians by applying knowledge gained through research at home and abroad into applications and practice.

www. hivnet. ubc. ca Funding • Established in 1990 as a cornerstone of the Canadian federal AIDS Strategy • The CTN is funded by the Canadian Institutes of Health Research (CIHR) • The CTN is jointly sponsored by the University of British Columbia and St. Paul’s Hospital (Providence Health Care) in Vancouver • Recently renewed by CIHR in 2014 until 2019 which marks 29 years of uninterrupted funding

www. hivnet. ubc. ca CTN in Numbers § 9, 000 Canadians enrolled in 150+ CTN trials § 300 + trials reviewed; > 150 + trials implemented § Currently, supporting 60 studies (start-up, enrolling, ongoing, reporting) § 40 + sites in Canada § Contributed to the advances in HAART § > 29 years of uninterrupted grant renewals from CIHR § 79 postdoctoral fellows funded
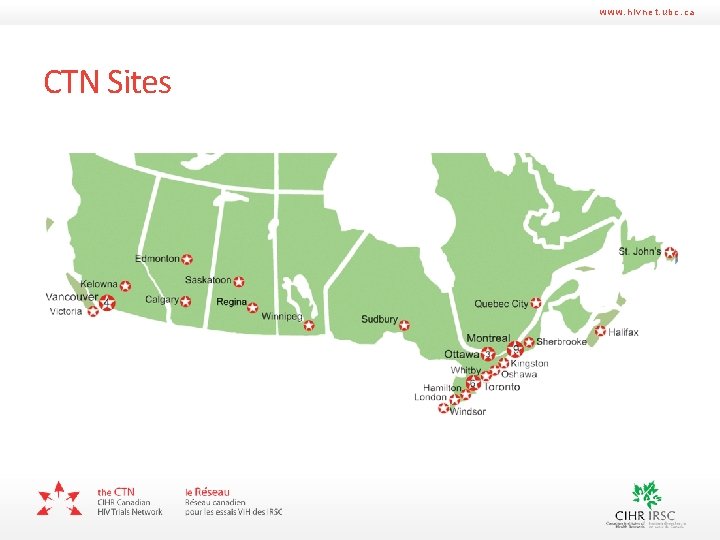
www. hivnet. ubc. ca CTN Sites

www. hivnet. ubc. ca RESEARCH CORE TEAMS • Responsible for: • Encouraging participation and idea/concept generation from investigators • Managing the flow of concepts through development to protocols • Supervision of ongoing study • Directing CTN research through the identification of new medical/scientific needs and emerging trends

www. hivnet. ubc. ca CTN STANDING COMMITTEES • External Advisory Committee (EAC) • Steering Committee (SC) • Scientific Review Committee (SRC) • Community Advisory Committee (CAC) • Data Safety Monitoring Committee (DSMC)
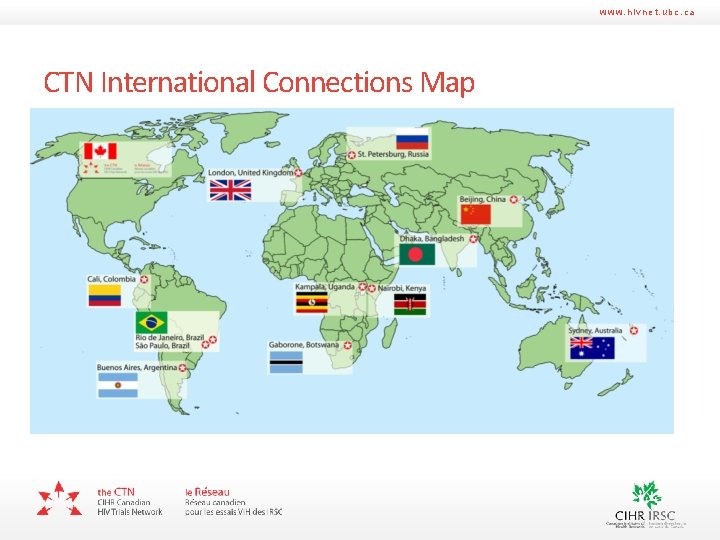
www. hivnet. ubc. ca CTN International Connections Map

www. hivnet. ubc. ca Collaborations • Research Partners • National Partners • ANRS, US NIAID/NIH, CANFAR • Canadian AIDS Society • Centre for Health Evaluation and Outcome • Canadian Aboriginal AIDS Network (also Sciences (CHÉOS), Centre Hospitalier de l’Université de Montréal, Medical Research Council, Kirby Institute, Women’s Health Research Institute • CHVI Research and Development Alliance Coordinating Office, CIHR Centre for REACH in HIV/AIDS, , Can. Cure, CANOC, Fonds de la recherche en santé du Quebec • Argos Therapeutics, GSK, Merck Canada, Wyeth Pharmaceuticals (Now a part of Pfizer), Gilead, Abbvie, BMS, Sanofi Pasteur, Vii. V Healthcare knowledge user on grant) • Canadian Association for HIV Research Canadian HIV/AIDS Legal Network • CATIE (also knowledge user on grant) • Canadian Treatment Action Council • Canadian Working Group on HIV and Rehabilitation (CWGHR) • Interagency Coalition on AIDS and Development (ICAD)

www. hivnet. ubc. ca Workshop Plan • CTN International Fellowship Awards Program, Jacquie Sas • Introduction to PMTC, Dr. Frederick Morfaw and Dr. Jason Brophy • Best Practices in PMTC, Dr. Amy Slogrove • Core Competencies: Posing a research question, Dr. Joel Singer • Core Competencies: Choosing a study design, Dr. Jackson Mukonzo • Core Competencies: Sample size: Why and how? Dr. Lawrence Mbuagbaw • Closing remarks by The Honourable Dr. Jane Philpott, Minister of Health, Canada
- Slides: 10