Writing Chemical Formulas General Chemistry Mrs Amy Nare


















- Slides: 21

Writing Chemical Formulas General Chemistry Mrs. Amy Nare 1 http: //chem. pdx. edu/~wamserc/C 335 W 00/gifs/MW 2. gif

Objectives n n n Distinguish between molecular and ionic compounds Contrast molecular formulas and formula units Use the periodic table to determine the charge on an ion Define monatomic and polyatomic ion and name the charges of common polyatomic ions Write the formulas and names for binary and ternary ionic compounds Write formulas and names for molecular compounds 2

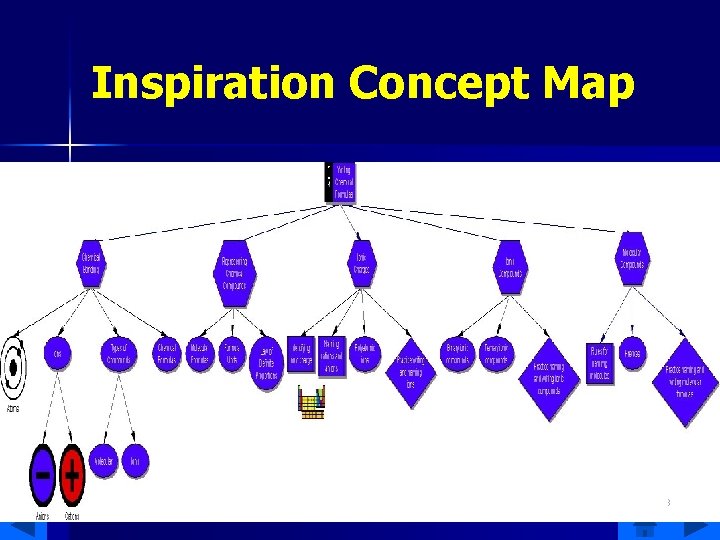
Inspiration Concept Map 3

Chemical Bonding Atoms – same number of protons (+) and electrons (-); electrically neutral n Ions – atoms w/ a (+) or (-) charge; have lost or gained electrons (e-) * Cations: (+) charge; has lost e-; metals n * Anions: (-) charge; has gained e-; nonmetals 4

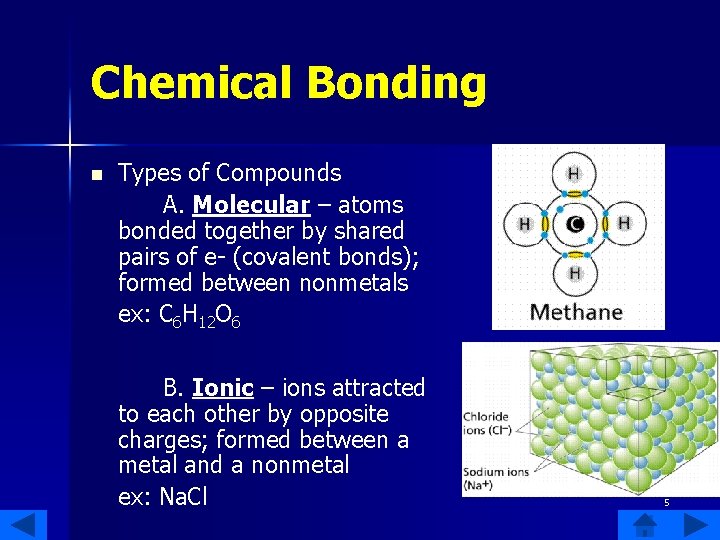
Chemical Bonding n Types of Compounds A. Molecular – atoms bonded together by shared pairs of e- (covalent bonds); formed between nonmetals ex: C 6 H 12 O 6 B. Ionic – ions attracted to each other by opposite charges; formed between a metal and a nonmetal ex: Na. Cl 5

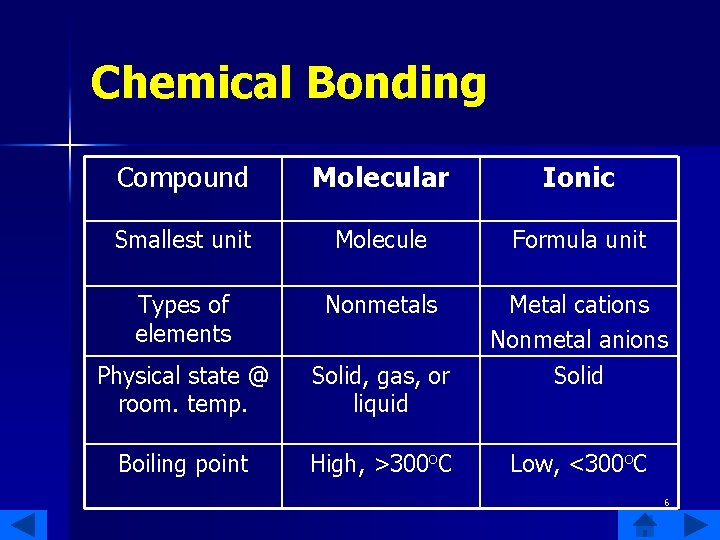
Chemical Bonding Compound Molecular Ionic Smallest unit Molecule Formula unit Types of elements Nonmetals Physical state @ room. temp. Solid, gas, or liquid Metal cations Nonmetal anions Solid Boiling point High, >300 o. C Low, <300 o. C 6

Chemical Bonding Chemical Formulas – show the type and number of atoms in smallest unit of substance n Molecular Formula – type and number of atoms joined to form a molecule ex: C 2 H 5 OH n Formula Unit – smallest ratio of ions in an ionic compound ex: Ca. F 2 7

Representing Chemical Compounds Law of Definite Proportions – in any sample of a compound, the elements are always combined in the same proportions ex: H 2 O and H 2 O 2 H 2 O – water – H: O ratio always 2: 1 H 2 O 2 – hydrogen peroxide – H: O ratio always 1: 1 n 8

Identifying Ionic Charges Group A elements – use the periodic table to determine ionic charge * elements in same group have same ionic charge * Group 4 A and Noble gases – almost never form ions n Group B elements – many have more than one ionic charge n 9

Identifying Ionic Charges http: //wps. prenhall. com/wps/media/objects/476/488316/ch 04. html Charge on cations corresponds to group #. Charge on anions is found by subtracting 8 by group number 10 the number 8 is used b/c it represents # of valence e- in Noble gases

Naming Cations and Anions Monatomic Ions n n n Ions formed by one element Cations * for Group A elements – just write element’s name ex: calcium ion (Ca 2+) = calcium * for Group B elements – write element’s name, then Roman numerals in parentheses to denote charge ex: Fe 2+ = Iron (II) and Fe 3+ = Iron (III) Anions – drop the end of the element’s name & add “–ide” ending ex: chlorine ion (Cl 1 -) = chloride 11

Naming Cations and Anions Polyatomic Ions n n Ions formed by more than one type of element Atoms of different elements held together by covalent bonds Atoms always stay together and collectively have a single charge Do not always have “-ide” ending ex: NH 41 - = ammonium ion CO 32 - = carbonate ion Learn names, formulas, and charges of polyatomic ions! 12

Naming Cations and Anions n n n n Potassium ion Copper (II) ion Chloride ion Oxide ion Ba 2+ S 2 Au 3+ n n n Nitrite ion Hydroxide ion Phosphate ion SO 42 Cr. O 42 Cl. O 3213

Binary Ionic Compounds n n n Compounds composed of 2 different monatomic elements To write binary formulas – write cation first, then anion *criss-cross charges to determine how many of each ion you need *use subscripts to denote number of ions ex: Ca 2+ + Cl 1 Ca. Cl 2 Na 1+ + Cl 1 Na. Cl To name binary compounds – write name of cation first, then anion (-ide) ex: Ca. Cl 2 = calcium chloride 14 Li 2 O = lithium oxide

Ternary Ionic Compounds n n n Compounds containing at least one polyatomic ion; at least 3 different elements To write ternary formulas: write cation first, then anion *criss-cross charges to determine how many of each ion you need *use subscripts to denote number of ions *must use parentheses around polyatomic if more than one is needed!!! ex: Na 1+ + SO 32 Na 2 SO 3 Mg 2+ + OH 1 Mg(OH)2 [not same as Mg. OH 2] To name ternary compounds: write name of cation, then name of anion (not all end in “-ide”) **be careful with transition metals (more than one charge)** ex: Ca. CO 3 = calcium carbonate 15 Pb. SO 4 = lead (II) sulfate Ag 2 Cr. O 4 = silver chromate

Ionic Compounds Na. NO 3 n Ca. SO 4 n (NH 4)2 O n Cu. SO 3 n Fe(OH)3 n Na. F n n n n Lithium sulfide Iron (III) phosphide Magnesium fluoride Barium nitrate Aluminum hydroxide Potassium phosphate Practice making ionic compounds! 16

Binary Molecular Compounds n n 1 2 3 4 5 Two nonmetals joined by covalent bonds Use prefixes for naming = = = monoditritetrapenta- 6 = hexa 7 = hepta 8 = octa 9 = nona 10 = deca- 17

Binary Molecular Compounds n n To name binary molecular compounds: *first element gets a prefix if there is more than one *second element ALWAYS gets prefix, and “-ide” ending ex: N 2 O 3 = dinitrogen trioxide CO = carbon monoxide (not monocarbon) If element begins with vowel and prefix ends in “a” or “o”, then drop last vowel on prefix to form the name ex: Cl 2 O 7 = dichlorine heptoxide (not heptaoxide) 18

Molecular Compounds P 2 O 5 n N 2 O n NO 2 n CBr 4 n CO 2 n tetraiodine nonoxide n sulfur hexafluoride n nitrogen trioxide n carbon tetrahydride n phosphorus trifluoride n 19

n Assess what you learned. Log on to the Internet and take the quiz. http: //school. discovery. com/quizzes 31/amy_trauth/F ormulas. Quiz. html 20

Websites on Writing Formulas 21