World of Buffers LO Learning Objectives v Understand

World of Buffers LO (Learning Objectives) v Understand the definition of a buffer v Understand buffer action v Calculate p. H of buffers and new p. H if acid or base added v. Salt hydrolysis

SALT HYDROLYSIS Many salts dissolve in water to produce solutions which are not neutral. This is because the ions formed react with the hydroxide and hydrogen ions formed when water dissociates. KNOW: Conjugate bases from strong acid, have essentially no ability to gain protons, so are not really bases at all. There are four distinct systems. All dissociated ions are aqueous ions. Some Salts are neutral: Na. Cl Na + + Cl - Some Salts form weak bases: Na. F Na + + F - Some Salts form weak acids: Some salts form both acids and bases - NH 4 Cl ——> NH 4+ + Cl¯ NH 4 F ——> NH 4+ + F¯ Kb (F ): 1. 4 x 10 -11 < Ka (NH 4 +) =: 5. 6 x 10 -10 Thus it is acid Neutral are bases are acids Acid and a base (biggest Ka or Kb wins)

Salt ID Identify each salt as neutral, acidic, or basic. 1) NH 4 I Acid because of NH 4+ I- from a conjugate base from strong acid 2) Base from the 3) Na. NO 2 Na+ is neutral but NO 2¯ is a base KNO 3 - from a conjugate base from strong I Neutral because K+ not acid or base, and acid 4) Base C 2 H 3 O 2¯ KC 2 H 3 O 2 5) NH 4 CH 3 COO (s) Neutral: Ka (NH 4+ ) = 5. 6 x 10 -10 and Kb = 5. 6 x 10 -10
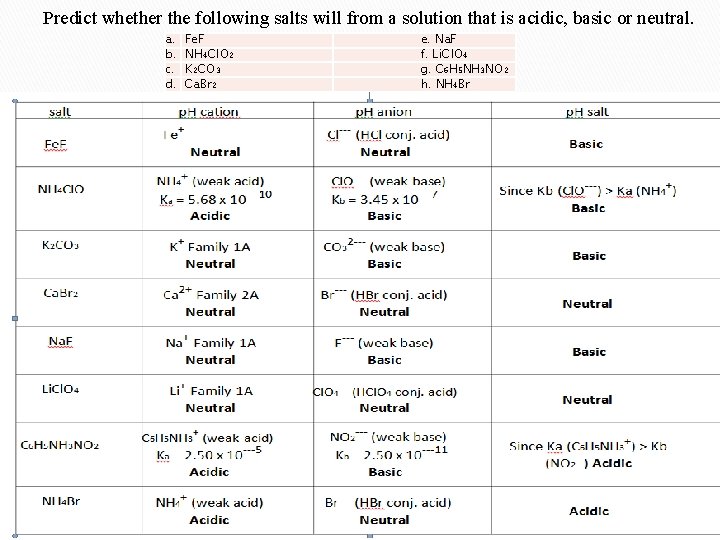
Predict whether the following salts will from a solution that is acidic, basic or neutral. a. b. c. d. Fe. F NH 4 Cl. O 2 K 2 CO 3 Ca. Br 2 e. Na. F f. Li. Cl. O 4 g. C 6 H 5 NH 3 NO 2 h. NH 4 Br

What is a Buffer? Contain weak acids or weak bases and their corresponding conjugate partners (common ions). Resist changes in p. H. Buffering capacity depends on the amount of weak acid or weak base and their common ions. Effective p. H buffering range ~ p. Ka 1. Depends on how much H 3 O+ or OH- the buffer can absorb so depends on the concentrations of HA and A ־.

Calculating the p. H of an acidic buffer solution Calculate the p. H of a buffer whose [HA] is 0. 1 mol dm-3 and [A¯] of 0. 1 mol dm-3. Ka = [H+(aq)] [A¯(aq)] [HA(aq)]

Calculating the p. H of an acidic buffer solution Calculate the p. H of a buffer whose [HA] is 0. 1 mol dm-3 and [A¯] of 0. 1 mol dm-3. Ka = [H+(aq)] [A¯(aq)] [HA(aq)] re-arrange [H+(aq)] = Ka x [HA(aq)] [A¯(aq)]

Calculating the p. H of an acidic buffer solution Calculate the p. H of a buffer whose [HA] is 0. 1 mol dm-3 and [A¯] of 0. 1 mol dm-3. Ka = [H+(aq)] [A¯(aq)] [HA(aq)] re-arrange [H+(aq)] = [HA(aq)] x Ka [A¯(aq)] from information given [A¯] = 0. 1 mol dm-3 [HA] = 0. 1 mol dm-3

Calculating the p. H of an acidic buffer solution Calculate the p. H of a buffer whose [HA] is 0. 1 mol dm-3 and [A¯] of 0. 1 mol dm-3. Ka = [H+(aq)] [A¯(aq)] [HA(aq)] re-arrange [H+(aq)] = Ka from information given [HA(aq)] [A¯] = 0. 1 mol dm-3 [HA] = 0. 1 mol dm-3 If the Ka of the weak acid HA is 2 x 10 -4 mol dm-3. [H+(aq)] = 0. 1 x 2 x 10 -4 0. 1 = 2 x 10 -4 mol dm-3

Calculating the p. H of an acidic buffer solution Calculate the p. H of a buffer whose [HA] is 0. 1 mol dm-3 and [A¯] of 0. 1 mol dm-3. Ka = [H+(aq)] [A¯(aq)] [HA(aq)] re-arrange [H+(aq)] = [HA(aq)] x Ka [A¯(aq)] from information given [A¯] = 0. 1 mol dm-3 [HA] = 0. 1 mol dm-3 If the Ka of the weak acid HA is 2 x 10 -4 mol dm-3. [H+(aq)] = 0. 1 x 2 x 10 -4 = 2 x 10 -4 mol dm-3 0. 1 p. H = - log 10 [H+(aq)] = 3. 699

Buffer solutions It is essential to have a weak acid for an equilibrium to be present so that conjugate base ions are also present and they can go back and forth CH 3 COOH(aq) CH 3 COO¯(aq) + H+(aq) A strong acid can’t be used as it is fully dissociated and cannot remove H +(aq), we have no effective conjugate base and no back and forth either. HCl(aq) ——> Cl¯(aq) + H+(aq) Adding acid Any H+ added is removed by reacting with CH 3 COO¯ ions to form CH 3 COOH Adding alkali This adds OH¯ ions which react with H+ ions Removal of H+ according to Le Chatelier’s Principle we shift right

Buffer Exercise Indicate whether each of the following solution mixtures will make a buffer solution. Explain. (a) 50. 0 m. L of 0. 10 M NH 3 + 50. 0 m. L of 0. 10 M NH 4 NO 3; Yes (b) 50. 0 m. L of 0. 10 M NH 3 + 50. 0 m. L of 0. 10 M HNO 3; NO (c) 50. 0 m. L of 0. 10 M NH 3 + 25. 0 m. L of 0. 10 M HNO 3; YES (d) 50. 0 m. L of 0. 10 M NH 4 NO 3 + 25. 0 m. L of 0. 10 M Na. OH; YES (e) 50. 0 m. L of 0. 10 M NH 4 NO 3 + 50. 0 m. L of 0. 10 M Na. OH; NO
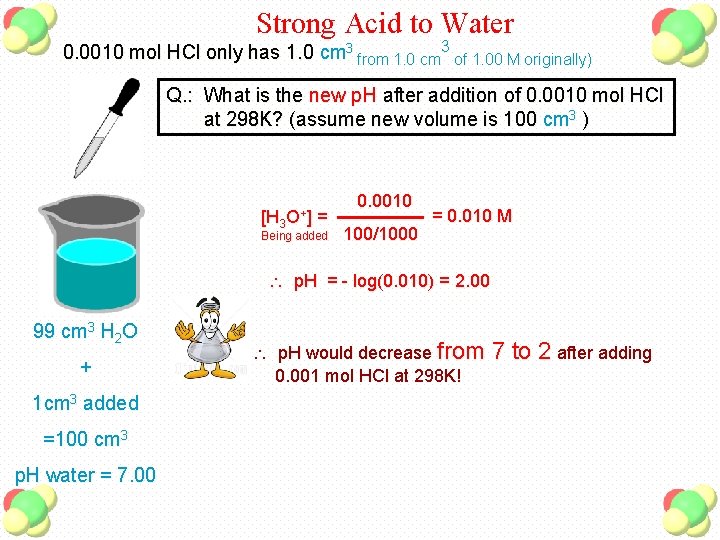
Strong Acid to Water 0. 0010 mol HCl only has 1. 0 cm 3 from 1. 0 cm 3 of 1. 00 M originally) Q. : What is the new p. H after addition of 0. 0010 mol HCl at 298 K? (assume new volume is 100 cm 3 ) [H 3 O+] = Being added 0. 0010 100/1000 = 0. 010 M p. H = - log(0. 010) = 2. 00 99 cm 3 H 2 O + 1 cm 3 added =100 cm 3 p. H water = 7. 00 p. H would decrease from 0. 001 mol HCl at 298 K! 7 to 2 after adding
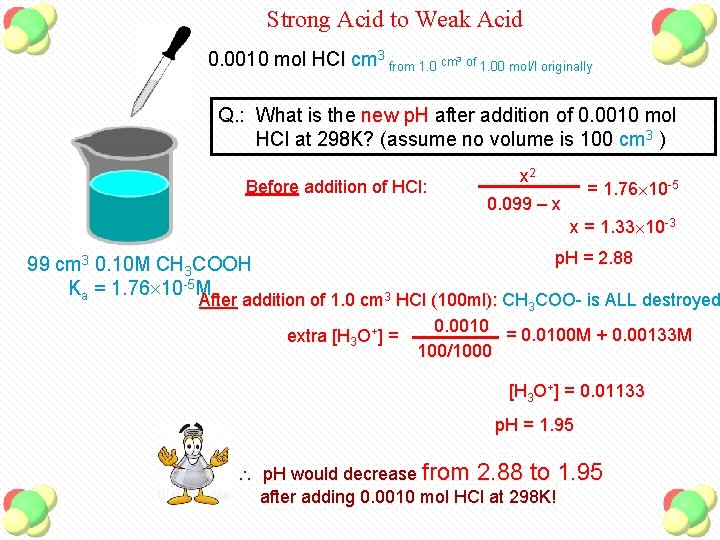
Strong Acid to Weak Acid 0. 0010 mol HCl cm 3 from 1. 0 cm 3 of 1. 00 mol/l originally Q. : What is the new p. H after addition of 0. 0010 mol HCl at 298 K? (assume no volume is 100 cm 3 ) Before addition of HCl: x 2 0. 099 – x = 1. 76 10 -5 x = 1. 33 10 -3 99 cm 3 0. 10 M CH 3 COOH Ka = 1. 76 10 -5 M p. H = 2. 88 After addition of 1. 0 cm 3 HCl (100 ml): CH 3 COO- is ALL destroyed 0. 0010 = 0. 0100 M + 0. 00133 M extra [H 3 O+] = 100/1000 [H 3 O+] = 0. 01133 p. H = 1. 95 p. H would decrease from 2. 88 to 1. 95 after adding 0. 0010 mol HCl at 298 K!
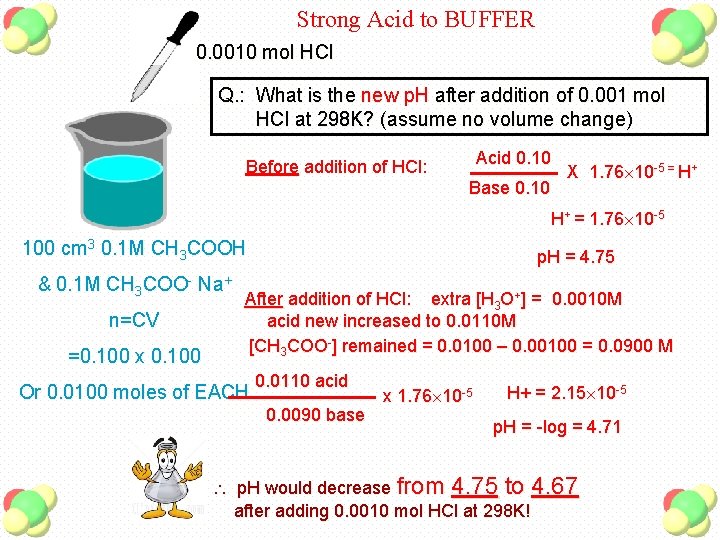
Strong Acid to BUFFER 0. 0010 mol HCl Q. : What is the new p. H after addition of 0. 001 mol HCl at 298 K? (assume no volume change) Acid 0. 10 Before addition of HCl: Base 0. 10 X 1. 76 10 -5 = H+ H+ = 1. 76 10 -5 100 cm 3 0. 1 M CH 3 COOH & 0. 1 M CH 3 COO- Na+ n=CV =0. 100 x 0. 100 p. H = 4. 75 After addition of HCl: extra [H 3 O+] = 0. 0010 M acid new increased to 0. 0110 M [CH 3 COO-] remained = 0. 0100 – 0. 00100 = 0. 0900 M Or 0. 0100 moles of EACH 0. 0110 acid 0. 0090 base x 1. 76 10 -5 H+ = 2. 15 10 -5 p. H = -log = 4. 71 p. H would decrease from 4. 75 to 4. 67 after adding 0. 0010 mol HCl at 298 K!
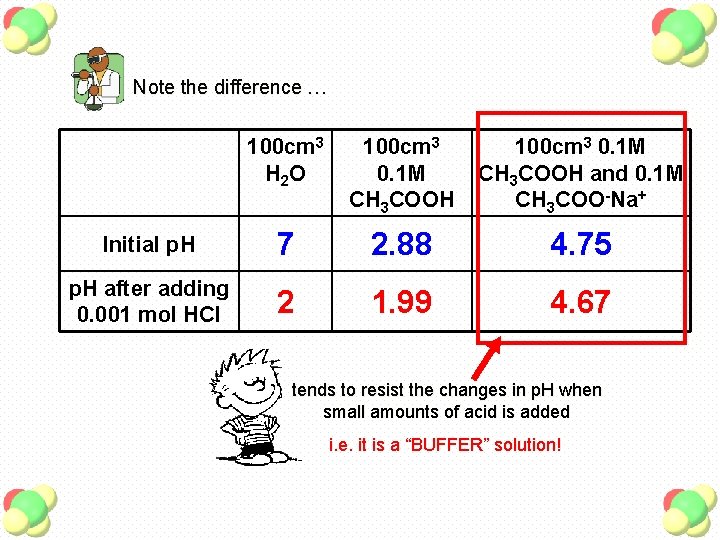
Note the difference … 100 cm 3 H 2 O 100 cm 3 0. 1 M CH 3 COOH and 0. 1 M CH 3 COO-Na+ Initial p. H 7 2. 88 4. 75 p. H after adding 0. 001 mol HCl 2 1. 99 4. 67 tends to resist the changes in p. H when small amounts of acid is added i. e. it is a “BUFFER” solution!
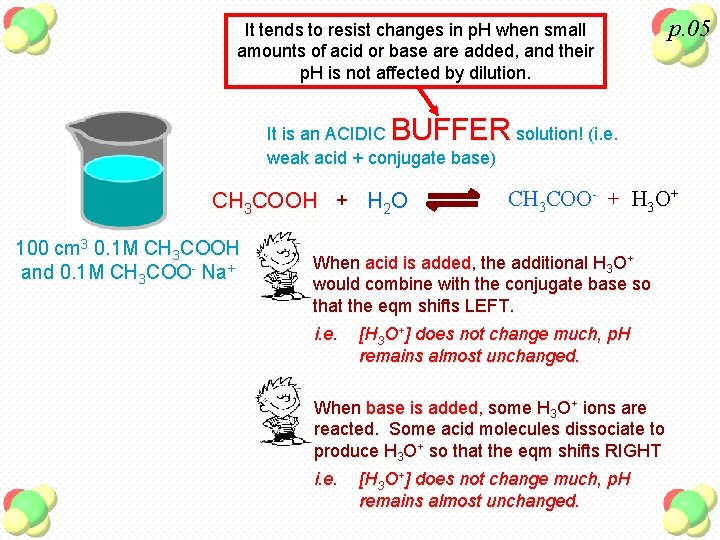
It tends to resist changes in p. H when small amounts of acid or base are added, and their p. H is not affected by dilution. p. 05 BUFFER solution! (i. e. It is an ACIDIC weak acid + conjugate base) CH 3 COOH + H 2 O 100 cm 3 0. 1 M CH 3 COOH and 0. 1 M CH 3 COO- Na+ CH 3 COO- + H 3 O+ When acid is added, the additional H 3 O+ would combine with the conjugate base so that the eqm shifts LEFT. i. e. [H 3 O+] does not change much, p. H remains almost unchanged. When base is added, some H 3 O+ ions are reacted. Some acid molecules dissociate to produce H 3 O+ so that the eqm shifts RIGHT i. e. [H 3 O+] does not change much, p. H remains almost unchanged.
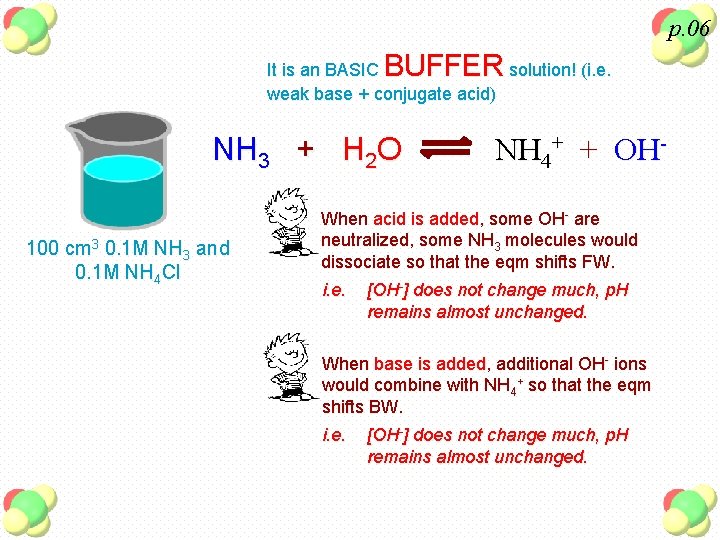
p. 06 BUFFER It is an BASIC solution! (i. e. weak base + conjugate acid) NH 3 + H 2 O 100 cm 3 0. 1 M NH 3 and 0. 1 M NH 4 Cl NH 4+ + OH- When acid is added, some OH- are neutralized, some NH 3 molecules would dissociate so that the eqm shifts FW. i. e. [OH-] does not change much, p. H remains almost unchanged. When base is added, additional OH- ions would combine with NH 4+ so that the eqm shifts BW. i. e. [OH-] does not change much, p. H remains almost unchanged.

Calculating the p. H of an acidic buffer solution Calculate the p. H of the solution formed when 500 cm 3 of 0. 1 mol dm-3 of weak acid HX is mixed with 500 cm 3 of a 0. 2 mol dm-3 solution of its salt Na. X. Ka = 4 x 10 -5 mol dm-3. Ka = [H+(aq)] [X¯(aq)] [HX(aq)]

Calculating the p. H of an acidic buffer solution Calculate the p. H of the solution formed when 500 cm 3 of 0. 1 mol dm-3 of weak acid HX is mixed with 500 cm 3 of a 0. 2 mol dm-3 solution of its salt Na. X. Ka = 4 x 10 -5 mol dm-3. Ka = [H+(aq)] [X¯(aq)] [HX(aq)] re-arrange [H+(aq)] = [HX(aq)] Ka [X¯(aq)]

Calculating the p. H of an acidic buffer solution Calculate the p. H of the solution formed when 500 cm 3 of 0. 1 mol dm-3 of weak acid HX is mixed with 500 cm 3 of a 0. 2 mol dm-3 solution of its salt Na. X. Ka = 4 x 10 -5 mol dm-3. Ka = [H+(aq)] [X¯(aq)] [HX(aq)] re-arrange [H+(aq)] = [HX(aq)] Ka [X¯(aq)] The solutions have been mixed; the volume is now 1 dm 3 therefore [HX] = 0. 05 mol dm-3 [X¯] = 0. 10 mol dm-3 and

Calculating the p. H of an acidic buffer solution Calculate the p. H of the solution formed when 500 cm 3 of 0. 1 mol dm-3 of weak acid HX is mixed with 500 cm 3 of a 0. 2 mol dm-3 solution of its salt Na. X. Ka = 4 x 10 -5 mol dm-3. [H+(aq)] = Ka [HX(aq)] [X¯(aq)] The solutions have been mixed; the volume is now 1 dm 3 therefore Substituting [HX] = 0. 05 mol dm-3 [X¯] = 0. 10 mol dm-3 [H+(aq)] = 4 x 10 -5 0. 05 and = 2 x 10 -5 mol dm-3 = 4. 699 0. 1 p. H = - log 10 [H+(aq)]

Buffer with Acid Added A 50. 00 m. L buffer solution which is 2. 00 M of both HC 2 H 3 O 2 and Na. C 2 H 3 O 2 It has a p. H = 4. 74. What is the new p. H after 2. 00 m. L of 6. 00 M HCl is added Initial moles in buffer= (2. 00 mol/L)(0. 500 L) = 0. 100 mol (same for acid & base) ADDED H+ moles=(6. 00 mol/L)(0. 00200 L) = 0. 0120 HCl mol The H+ ion reacts quantitatively with the conjugate base, C 2 H 3 O 2 - , form of the buffer, changing the base to acid ratio and thus the p. H. Equation H+ + HC 2 H 3 O 2 Initial moles 0. 012 Change in moles -0. 012 Final moles 0. 000 0. 088 0. 0 1. 69 New molarity 0. 100 C 2 H 3 O 2 0. 100 -0. 012 +0. 012 0. 112 2. 15
- Slides: 23