World Federation of Haemophilia 2014 World Congress Melbourne

World Federation of Haemophilia 2014 World Congress Melbourne, Australia An Indirect Comparison of the Efficacy of Prophylactic Use of r. FIXFc and r. FIX Products and Simulation of the Effect of Compliance on Effectiveness Alfonso Iorio, MD, Ph. D 13 May 2014 Alfonso Iorio, 1 Sangeeta Krishnan, 2 Lynn Huynh, 3 Paul Karner, 3 Mei Sheng Duh, 3 Sander Yermakov 3 1 Mc. Master University, Hamilton, Ontario, Canada; 2 Biogen Idec, Cambridge, MA, USA; 3 Analysis Group, Boston, MA, USA.

Presenter Disclosures for Alfonso Iorio In compliance with the EACCME* policy, WFH requires the following disclosures to be made at each presentation: CONFLICT DISCLOSURE – IF CONFLICT OF INTEREST EXISTS RESEARCH SUPPORT Baxter, Biogen Idec, and Novo Nordisk DIRECTOR, OFFICER, EMPLOYEE SHAREHOLDER HONORARIA ADVISORY COMITTEE CONSULTANT Baxter, Bayer, Biogen Idec, Novo Nordisk, and Pfizer This research was supported by Biogen Idec *European Accreditation Council for Continuing Medical Education

Introduction and Objective § § Prophylaxis for hemophilia B requires 2 -3 infusions/week. r. FIXFc may require fewer infusions. Compliance may be affected by number of infusions. No head-to-head clinical studies of r. FIXFc and r. FIX have been conducted. Objectives: Ø To indirectly compare the efficacy of r. FIXFc and r. FIX products, using published data. Ø To model the potential impact of improved compliance. 3 r. FIX, recombinant factor IX, r. FIXFc, recombinant factor IX Fc fusion protein.
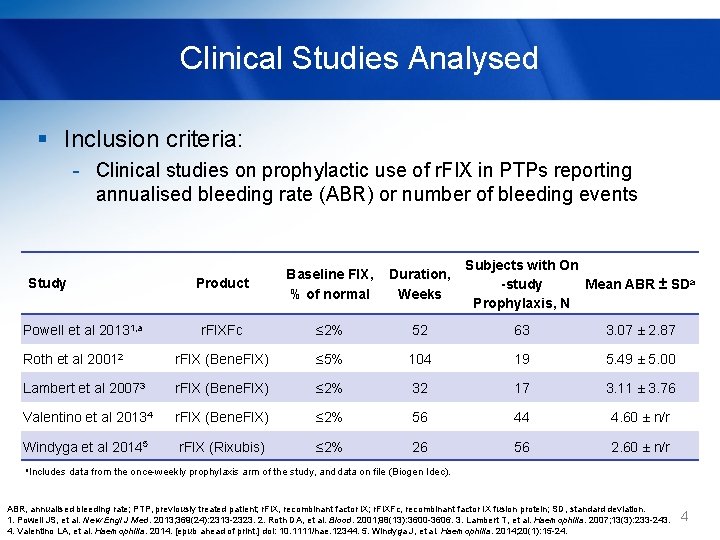
Clinical Studies Analysed § Inclusion criteria: - Clinical studies on prophylactic use of r. FIX in PTPs reporting annualised bleeding rate (ABR) or number of bleeding events Subjects with On -study Mean ABR ± SDa Prophylaxis, N Product Baseline FIX, % of normal Duration, Weeks r. FIXFc ≤ 2% 52 63 3. 07 ± 2. 87 Roth et al 20012 r. FIX (Bene. FIX) ≤ 5% 104 19 5. 49 ± 5. 00 Lambert et al 20073 r. FIX (Bene. FIX) ≤ 2% 32 17 3. 11 ± 3. 76 Valentino et al 20134 r. FIX (Bene. FIX) ≤ 2% 56 44 4. 60 ± n/r Windyga et al 20145 r. FIX (Rixubis) ≤ 2% 26 56 2. 60 ± n/r Study Powell et al 20131, a a. Includes data from the once-weekly prophylaxis arm of the study, and data on file (Biogen Idec). ABR, annualised bleeding rate; PTP, previously treated patient; r. FIX, recombinant factor IX; r. FIXFc, recombinant factor IX fusion protein; SD, standard deviation. 1. Powell JS, et al. New Engl J Med. 2013; 369(24): 2313 -2323. 2. Roth DA, et al. Blood. 2001; 98(13): 3600 -3606. 3. Lambert T, et al. Haemophilia. 2007; 13(3): 233 -243. 4. Valentino LA, et al. Haemophilia. 2014. [epub ahead of print. ] doi: 10. 1111/hae. 12344. 5. Windyga J, et al. Haemophilia. 2014; 20(1): 15 -24. 4

Meta-Analysis and Indirect Comparison § Unadjusted indirect comparison of efficacy - r. FIXFc: mean ABR = 3. 071 - r. FIX: pooled mean ABR based on random effects meta-analysis = 3. 84 (I 2 = 57. 5%) - Unreported standard deviations were estimated assuming a Poisson distribution and corrected for overdispersion. 1. Powell JS, et al. New Engl J Med. 2013; 369(24): 2313 -2323. 2. Roth DA, et al. Blood. 2001; 98(13): 3600 -3606. 3. Lambert T, et al. Haemophilia. 2007; 13(3): 233 -243. 4. Valentino LA, et al. Haemophilia. 2014. [epub ahead of print. ] doi: 10. 1111/hae. 12344. 5. Windyga J, et al. Haemophilia. 2014; 20(1): 15 -24. 6. Der. Simonian R, Laird N. Controlled Clin Trials. 1986; 7(3): 177 -188 5
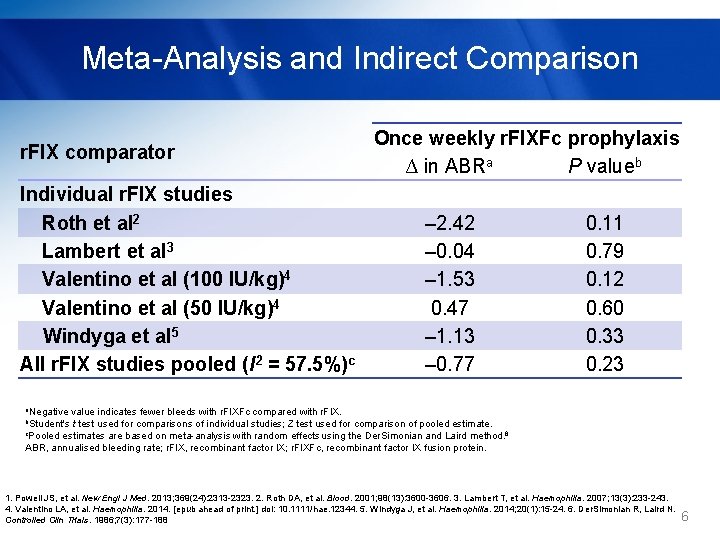
Meta-Analysis and Indirect Comparison r. FIX comparator Individual r. FIX studies Roth et al 2 Lambert et al 3 Valentino et al (100 IU/kg)4 Valentino et al (50 IU/kg)4 Windyga et al 5 All r. FIX studies pooled (I 2 = 57. 5%)c Once weekly r. FIXFc prophylaxis ∆ in ABRa P valueb – 2. 42 0. 11 – 0. 04 0. 79 – 1. 53 0. 12 0. 47 0. 60 – 1. 13 0. 33 – 0. 77 0. 23 a. Negative value indicates fewer bleeds with r. FIXFc compared with r. FIX. b. Student's t test used for comparisons of individual studies; Z test used for comparison of pooled estimate. c. Pooled estimates are based on meta-analysis with random effects using the Der. Simonian and Laird method. 6 ABR, annualised bleeding rate; r. FIX, recombinant factor IX; r. FIXFc, recombinant factor IX fusion protein. 1. Powell JS, et al. New Engl J Med. 2013; 369(24): 2313 -2323. 2. Roth DA, et al. Blood. 2001; 98(13): 3600 -3606. 3. Lambert T, et al. Haemophilia. 2007; 13(3): 233 -243. 4. Valentino LA, et al. Haemophilia. 2014. [epub ahead of print. ] doi: 10. 1111/hae. 12344. 5. Windyga J, et al. Haemophilia. 2014; 20(1): 15 -24. 6. Der. Simonian R, Laird N. Controlled Clin Trials. 1986; 7(3): 177 -188 6
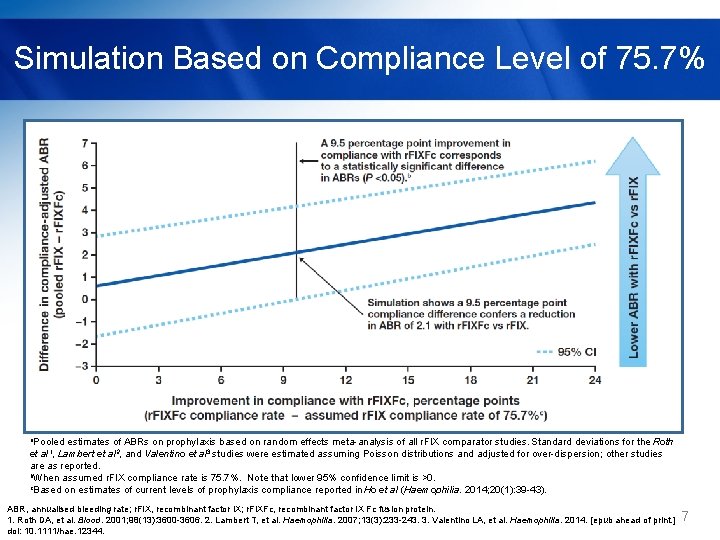
Simulation Based on Compliance Level of 75. 7% a. Pooled estimates of ABRs on prophylaxis based on random effects meta-analysis of all r. FIX comparator studies. Standard deviations for the Roth et al 1, Lambert et al 2, and Valentino et al 3 studies were estimated assuming Poisson distributions and adjusted for over-dispersion; other studies are as reported. b. When assumed r. FIX compliance rate is 75. 7%. Note that lower 95% confidence limit is >0. c. Based on estimates of current levels of prophylaxis compliance reported in Ho et al (Haemophilia. 2014; 20(1): 39 -43). ABR, annualised bleeding rate; r. FIX, recombinant factor IX; r. FIXFc, recombinant factor IX Fc fusion protein. 1. Roth DA, et al. Blood. 2001; 98(13): 3600 -3606. 2. Lambert T, et al. Haemophilia. 2007; 13(3): 233 -243. 3. Valentino LA, et al. Haemophilia. 2014. [epub ahead of print. ] doi: 10. 1111/hae. 12344. 7

Study Limitations § This comparison is indirect. § A random-effects meta-analysis approach was used to account for between-study variance. § The effect of changes in compliance are based on the assumption that ABR is correlated to compliance over the range of values reported in clinical trials for patients treated with on-demand or prophylaxis regimens. 8

Conclusions § Based on unadjusted indirect comparison of 6 clinical studies for r. FIXFc and r. FIX products - The efficacy of prophylaxis treatment with once-weekly r. FIXFc is comparable to more frequently infused r. FIX. - Less frequent infusions with r. FIXFc may enhance compliance and consequently effectiveness, as suggested by compliance modeling. - Simulations suggest improvements in compliance of ≥ 9 to 14% with r. FIXFc would yield a statistically significant reduction in mean ABR. § Additional studies are necessary to validate these findings and assess the true impact of r. FIXFc on realworld effectiveness. ABR, annualised bleeding rate; r. FIX, recombinant factor IX; r. FIXFc, recombinant factor IX Fc fusion protein. 9
- Slides: 9