WORKSHEET Layers of Earths Geosphere and Atmosphere Layers






















![Dispersion Each of the colours of the spectrum [ROYGBIV] has a slightly different wave. Dispersion Each of the colours of the spectrum [ROYGBIV] has a slightly different wave.](https://slidetodoc.com/presentation_image_h2/af4ac5be9250cdbd7a23a8fa594c5f19/image-30.jpg)







- Slides: 49

WORKSHEET Layers of Earth’s Geosphere and Atmosphere

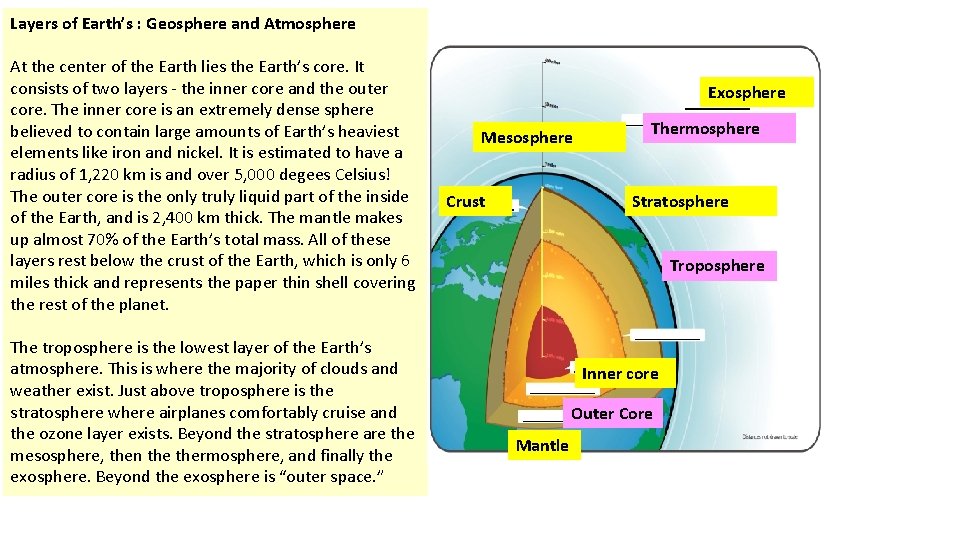
Layers of Earth’s : Geosphere and Atmosphere At the center of the Earth lies the Earth’s core. It consists of two layers - the inner core and the outer core. The inner core is an extremely dense sphere believed to contain large amounts of Earth’s heaviest elements like iron and nickel. It is estimated to have a radius of 1, 220 km is and over 5, 000 degees Celsius! The outer core is the only truly liquid part of the inside of the Earth, and is 2, 400 km thick. The mantle makes up almost 70% of the Earth’s total mass. All of these layers rest below the crust of the Earth, which is only 6 miles thick and represents the paper thin shell covering the rest of the planet. The troposphere is the lowest layer of the Earth’s atmosphere. This is where the majority of clouds and weather exist. Just above troposphere is the stratosphere where airplanes comfortably cruise and the ozone layer exists. Beyond the stratosphere are the mesosphere, then thermosphere, and finally the exosphere. Beyond the exosphere is “outer space. ” Exosphere Mesosphere Crust Thermosphere Stratosphere Troposphere Inner core Outer Core Mantle

Which layer is being described? Mesosphere Toposphere Inner core Crust A layer with very little air pressure, but still able to burn up most meteors coming in. where most of the air pressure and Earth’s weather is very hot, solid iron and nickel. It is solid due to the extreme pressures even though the temperatures here are well past what is needed to melt iron and nickel. solid, cool outer shell of the Earth. It is the thinnest of Earth’s layers and varies in thickness. Most of it is under water. Exosphere the last traces of our atmosphere are here; past this layer is outer space. Stratosphere a calm layer of the atmosphere with some air pressure, very little weather and the ozone layer Outer Core Thermosphere Mantle a layer made of iron and nickel, but is liquid due to the high temperatures. . A layer with so little air, we think of it as the beginnings of space. The ISS orbits here. starts about 4900 km deep and goes up to 30 km under the surface of the Earth. It has temperatures between 500 and 900 o. C. The high temperatures here allow it to flow in a similar way to a liquid

WORKSHEET Temperature, Pressure and Light

Temperature, Pressure, and Light 1. What is the definition of temperature? Ans: It is a measure of how much particles (such as atoms and molecules) are moving. 2. What is the definition of pressure? Ans: It is how much force air particles are pushing onto a surface, divided by the area of the surface (force per area) 3. What causes air pressure? Ans: The movement of air particles—the more particles there are, the more pressure there is; the more they bounce off an area, the more pressure there is.

Temperature, Pressure, and Light 4. How does air pressure change as we go farther away from Earth’s surface? Ans: Air pressure gets less and less as we go farther away from Earth’s surface. 5. When sunlight falls onto Earth’s surface, the light can do these three things: Ans: Absorb, reflect (bounce off), transmit (pass through)

6. Below, we have sunlight falling onto 4 different surfaces. Decide what will happen to the light in each of these cases: What will likely happen to the sunlight? Sunlight falling on the ocean Sunlight passes through at first, but is eventually absorbed by the water underneath Why I think this: Water is clear when it is shallow, but the deep ocean looks dark blue to us, showing that most sunlight is eventually absorbed when the water is deep.

6. Below, we have sunlight falling onto 4 different surfaces. Decide what will happen to the light in each of these cases: What will likely happen to the sunlight? Sunlight falls onto freshly fallen snow Sunlight reflects away. Why I think this: Snow is white and sparkly, showing that most colours of light are reflected

6. Below, we have sunlight falling onto 4 different surfaces. Decide what will happen to the light in each of these cases: What will likely happen to the sunlight? Sunlight falls onto black, volcanic rock Sunlight is absorbed. Why I think this: The black colour of the rock shows that very few colours of light are reflected back to our eyes, so they must be absorbed instead.

6. Below, we have sunlight falling onto 4 different surfaces. Decide what will happen to the light in each of these cases: What will likely happen to the sunlight? Sunlight falls onto light-coloured sand Sunlight is mostly reflected. Why I think this: Sand is light-coloured, showing that most colours of sunlight are reflected to our eyes, however the sand still has SOME colour, showing that a bit of light is absorbed









WORKSHEET Carbon cycle and the Green house Effect

Carbon Cycle and The Greenhouse Effect 1. Which gases can absorb infrared light? Ans: Water vapour, carbon dioxide, methane, and others 2. What is the carbon cycle? Ans: The way that carbon moves around our planet through different processes

Carbon Cycle and The Greenhouse Effect 3. How do plants and oceans remove carbon dioxide from the atmosphere? Ans: Plants do photosynthesis by taking carbon dioxide through their leaves and water from the soil. When this happens, the carbon is stored in glucose in the plant. 4. How do animals add carbon into the atmosphere? Ans: Animals carry out respiration, which uses oxygen and glucose to create carbon dioxide and water. The carbon dioxide is then breathed out into the atmosphere.

Carbon Cycle and The Greenhouse Effect 5. Where is carbon stored for a very long time? Ans: In the ground and deep in the ocean. 6. Explain why burning fossil fuels changes the balance of the carbon cycle. Ans: Fossil fuels are normally store carbon in the ground for millions of years before natural processes will move the carbon elsewhere. When we burn fossil fuels, we use this stored carbon and quickly release it back into the atmosphere as carbon dioxide.

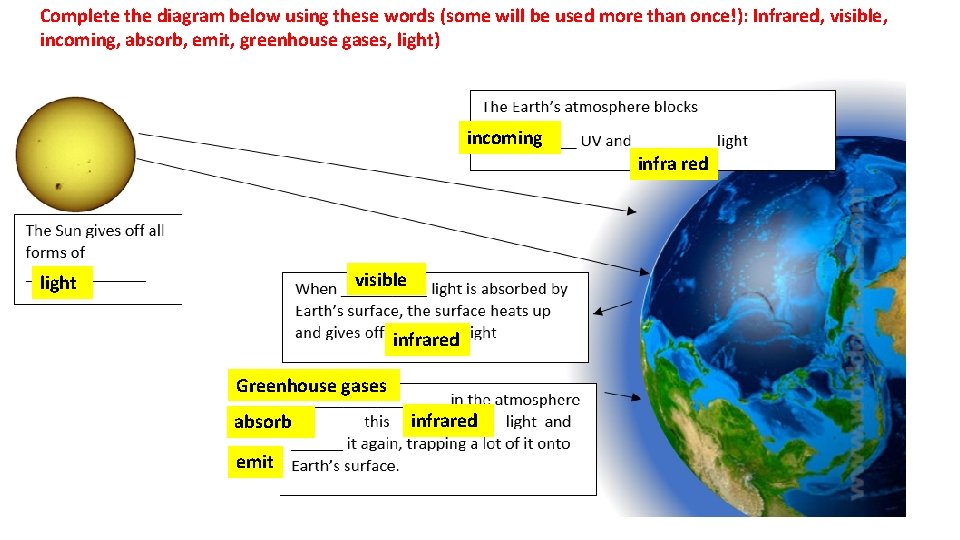
Complete the diagram below using these words (some will be used more than once!): Infrared, visible, incoming, absorb, emit, greenhouse gases, light) incoming infra red visible light infrared Greenhouse gases absorb emit infrared

Carbon Cycle and The Greenhouse Effect 7. Explain why we NEED greenhouse gases here on Earth. Ans: Without greenhouse gases, our planet would be much colder than it currently is, and the temperatures would not be suitable for life. 8. What would happen if we didn’t have enough greenhouse gases? Ans: Earth would be colder, and more of the oceans may freeze. Plants would struggle to survive or die out completely. This would affect the food web of the entire planet and therefore affect all life on the planet.

Carbon Cycle and The Greenhouse Effect 9. What would happen if we had too many greenhouse gases? Ans: Earth would be hotter, and more of the oceans may evaporate / ice caps would melt. Depending on how much hotter it was, some plants may prefer the warmer temperature, but many would struggle to adapt if the change happened quickly.

Carbon Cycle and The Greenhouse Effect 10. Explain why the carbon cycle plays an important role in Earth’s global temperature. Ans: The carbon cycle keeps the levels of carbon dioxide roughly the same over time. Since carbon dioxide is a greenhouse gas, it is one of the gases responsible for trapping infrared light onto our planet. The trapping of infrared light is what helps keep our planet at its current temperature. If the carbon cycle changed so that more carbon dioxide was in the atmosphere, Earth have hotter temperatures overall.


Life without colour Imagine you could only see in black and white. How might this affect your life? Would it rule out any careers for you? What dangers could there be? Working in groups, each person has two minutes to give a presentation to the rest of the group about their ideas. 1 28 ofof 20 84 © Boardworks Ltd 2005 2004

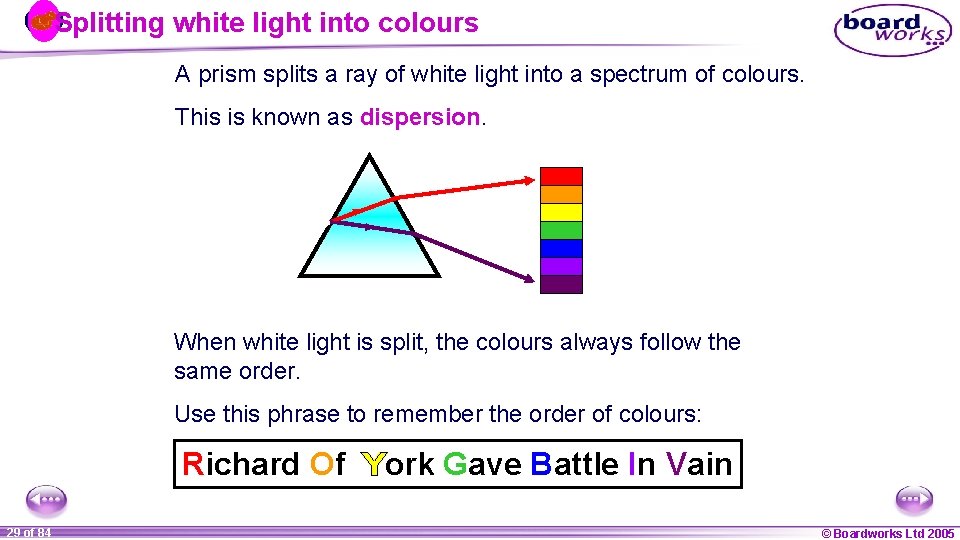
Splitting white light into colours A prism splits a ray of white light into a spectrum of colours. This is known as dispersion. When white light is split, the colours always follow the same order. Use this phrase to remember the order of colours: Richard Of York Gave Battle In Vain 1 29 ofof 20 84 © Boardworks Ltd 2005 2004
![Dispersion Each of the colours of the spectrum ROYGBIV has a slightly different wave Dispersion Each of the colours of the spectrum [ROYGBIV] has a slightly different wave.](https://slidetodoc.com/presentation_image_h2/af4ac5be9250cdbd7a23a8fa594c5f19/image-30.jpg)
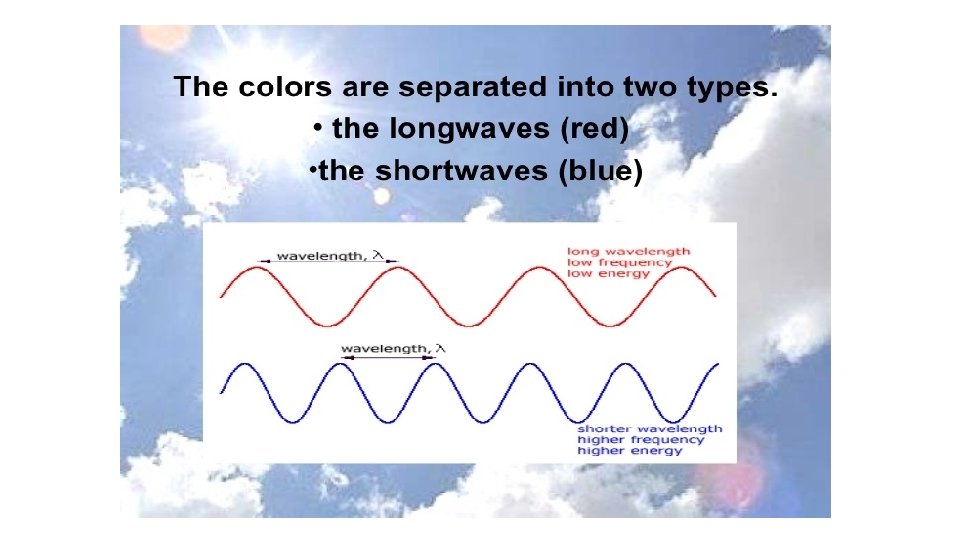
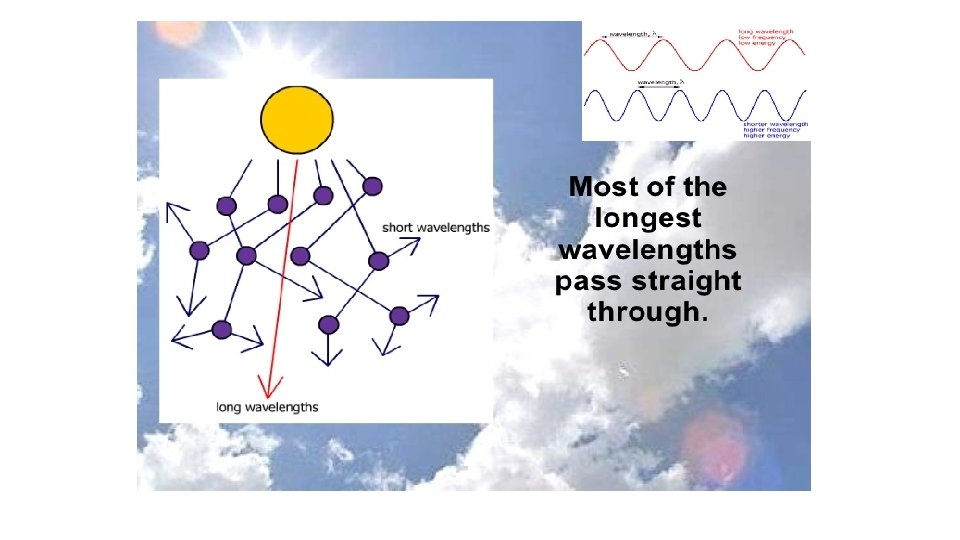
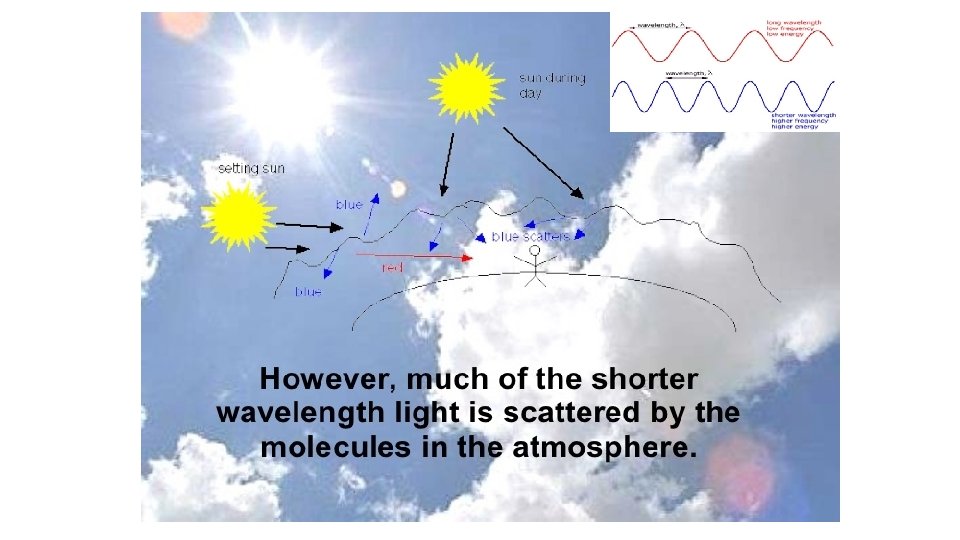
Dispersion Each of the colours of the spectrum [ROYGBIV] has a slightly different wave. What is the difference? Each colour has a different wavelength ( ). 1 30 ofof 20 84 © Boardworks Ltd 2005 2004

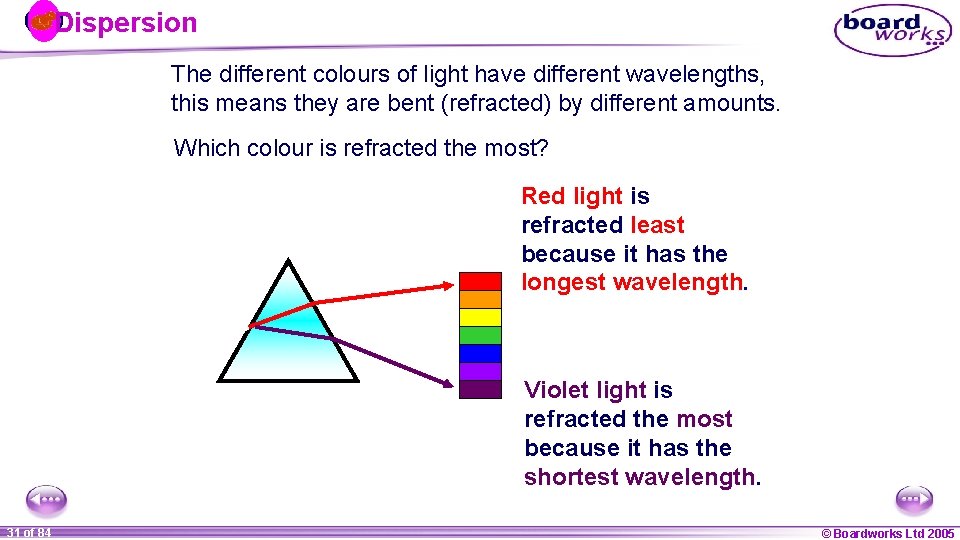
Dispersion The different colours of light have different wavelengths, this means they are bent (refracted) by different amounts. Which colour is refracted the most? Red light is refracted least because it has the longest wavelength. Violet light is refracted the most because it has the shortest wavelength. 1 31 ofof 20 84 © Boardworks Ltd 2005 2004

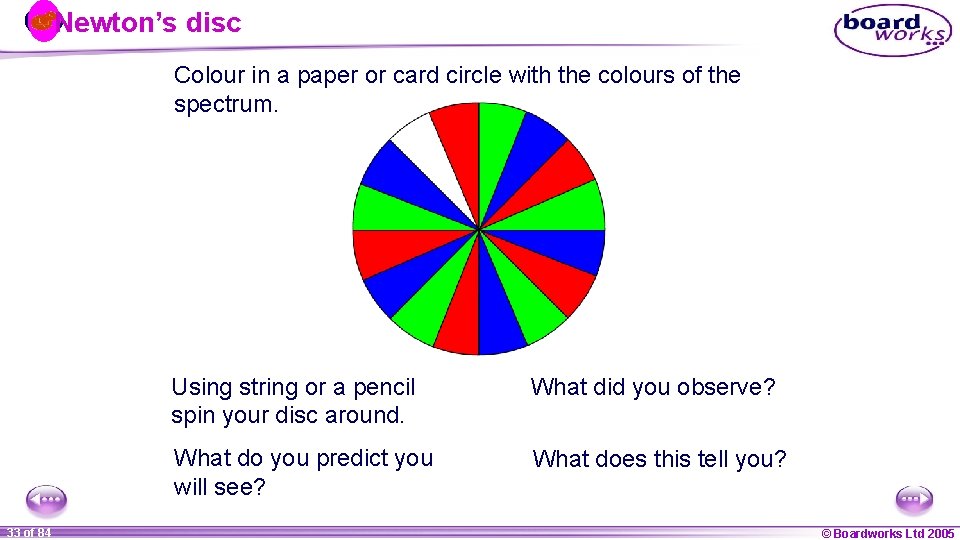
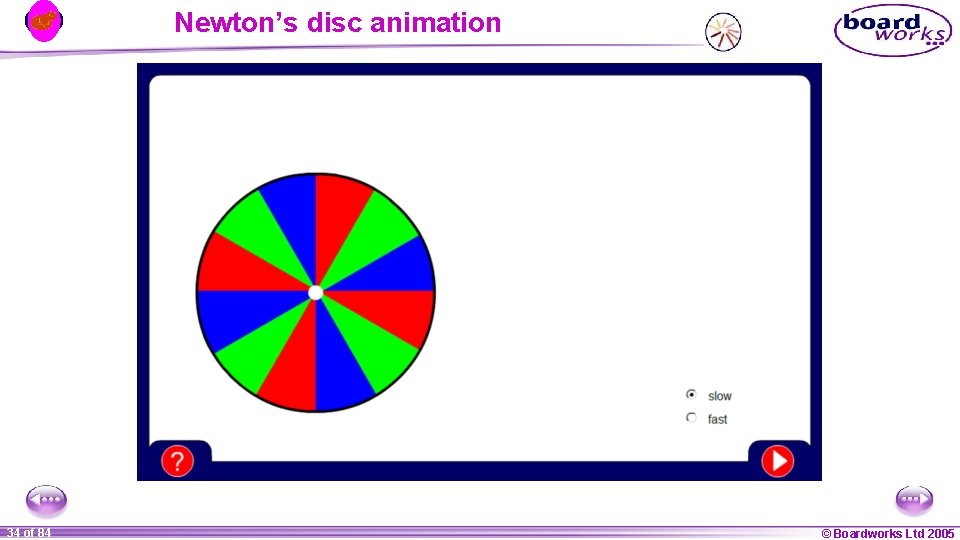
Recombining colours Remember how white light can be dispersed to give a spectrum of colours? To do the opposite – two prisms are needed! A similar effect can also be seen using a colour wheel (or Newton’s disc). 1 32 ofof 20 84 © Boardworks Ltd 2005 2004

Newton’s disc Colour in a paper or card circle with the colours of the spectrum. 1 33 ofof 20 84 Using string or a pencil spin your disc around. What did you observe? What do you predict you will see? What does this tell you? © Boardworks Ltd 2005 2004

Newton’s disc animation 1 34 ofof 20 84 © Boardworks Ltd 2005 2004

Seeing colours How do you see non-luminous objects such as a book? You see a non-luminous object when light hits the object and is then reflected into your eyes. So how do we see different colours? Why does a red dress look red? Why does a green apple look green? 1 35 ofof 20 84 © Boardworks Ltd 2005 2004

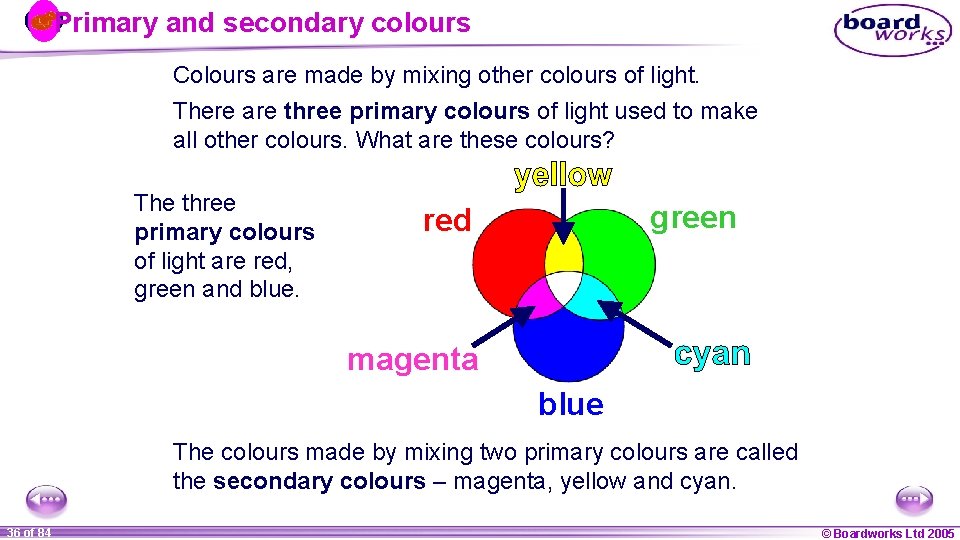
Primary and secondary colours Colours are made by mixing other colours of light. There are three primary colours of light used to make all other colours. What are these colours? The three primary colours of light are red, green and blue. green red magenta blue The colours made by mixing two primary colours are called the secondary colours – magenta, yellow and cyan. 1 36 ofof 20 84 © Boardworks Ltd 2005 2004

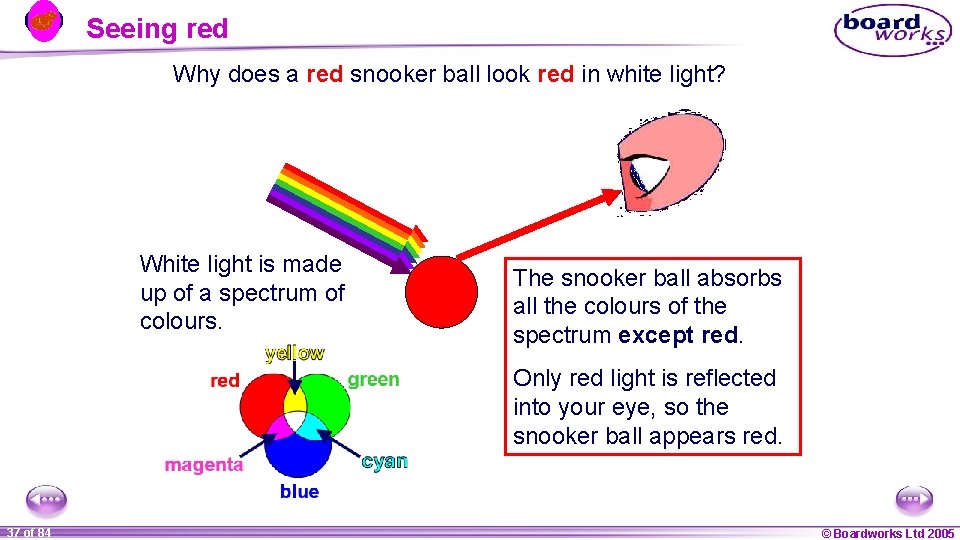
Seeing red Why does a red snooker ball look red in white light? White light is made up of a spectrum of colours. The snooker ball absorbs all the colours of the spectrum except red. Only red light is reflected into your eye, so the snooker ball appears red. 1 37 ofof 20 84 © Boardworks Ltd 2005 2004

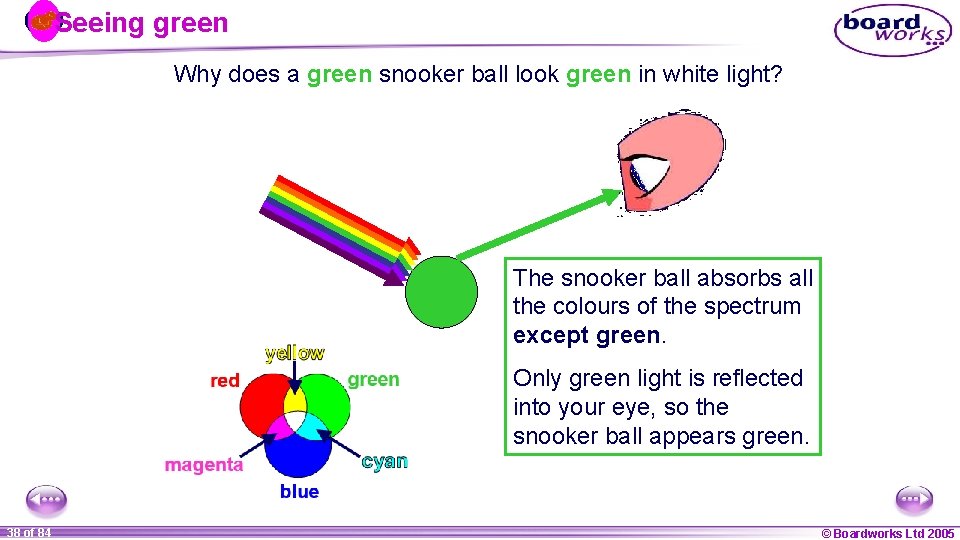
Seeing green Why does a green snooker ball look green in white light? The snooker ball absorbs all the colours of the spectrum except green. Only green light is reflected into your eye, so the snooker ball appears green. 1 38 ofof 20 84 © Boardworks Ltd 2005 2004

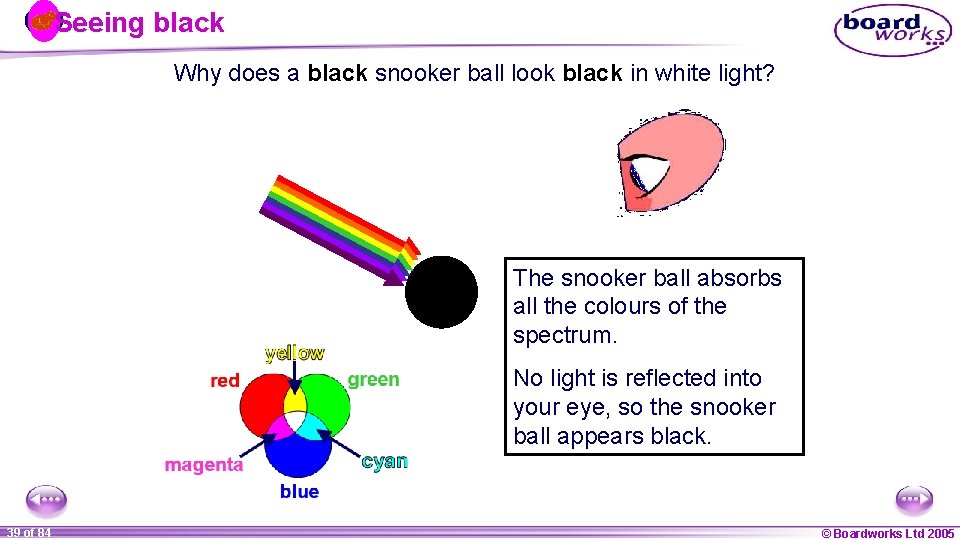
Seeing black Why does a black snooker ball look black in white light? The snooker ball absorbs all the colours of the spectrum. No light is reflected into your eye, so the snooker ball appears black. 1 39 ofof 20 84 © Boardworks Ltd 2005 2004

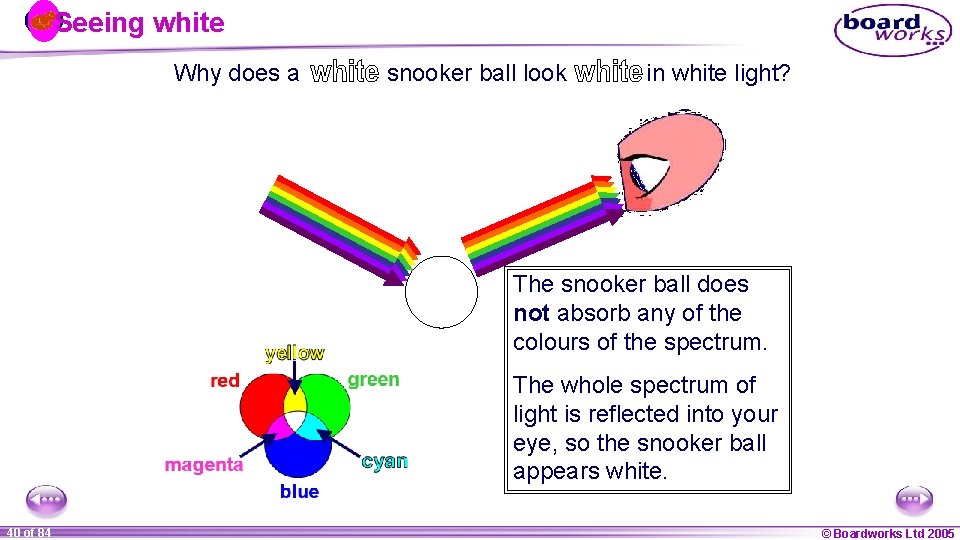
Seeing white Why does a snooker ball look in white light? The snooker ball does not absorb any of the colours of the spectrum. The whole spectrum of light is reflected into your eye, so the snooker ball appears white. 1 40 ofof 20 84 © Boardworks Ltd 2005 2004

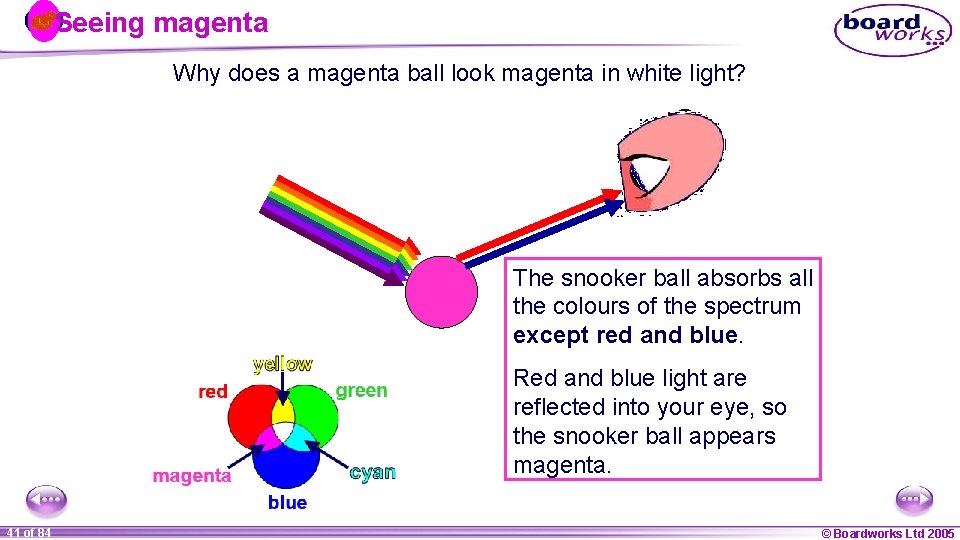
Seeing magenta Why does a magenta ball look magenta in white light? The snooker ball absorbs all the colours of the spectrum except red and blue. Red and blue light are reflected into your eye, so the snooker ball appears magenta. 1 41 ofof 20 84 © Boardworks Ltd 2005 2004

Seeing different colours What colours are absorbed by this frog’s skin? What colours are reflected into your eyes? This part of the skin absorbs all the colours of the spectrum except red, and so reflects red light. 1 42 ofof 20 84 This part of the skin absorbs all the colours of the spectrum and none are reflected. © Boardworks Ltd 2005 2004

Seeing different colours What colours are absorbed by this flower? What colours are reflected into your eyes? This part of the flower absorbs all colours except red and green. It reflects red and green light, and so appears yellow. 1 43 ofof 20 84 This part of the flower absorbs no colours. It reflects them all and so appears white. © Boardworks Ltd 2005 2004

WORKSHEET Biological, Chemical, Physical Processes of the Carbon Cycle

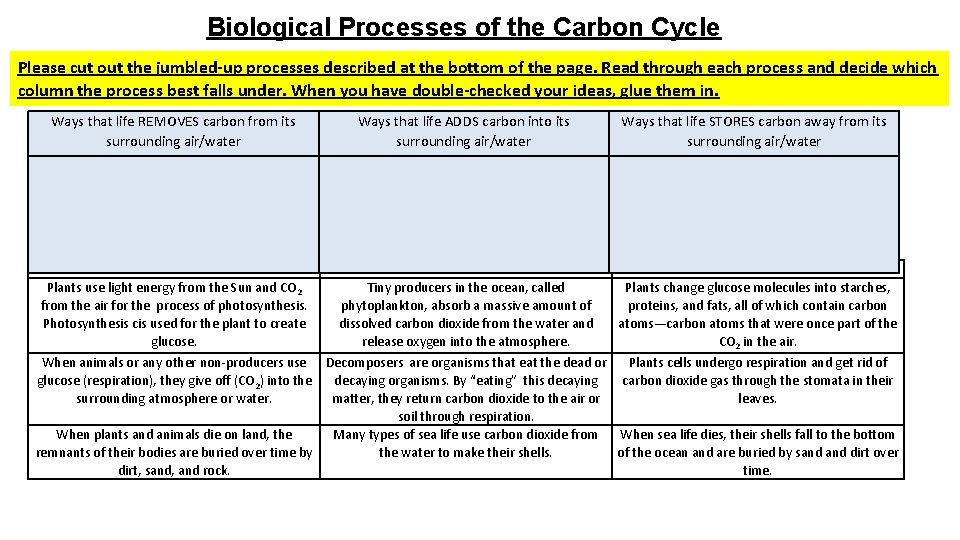
Biological Processes of the Carbon Cycle Please cut out the jumbled-up processes described at the bottom of the page. Read through each process and decide which column the process best falls under. When you have double-checked your ideas, glue them in. Ways that life REMOVES carbon from its surrounding air/water Ways that life ADDS carbon into its surrounding air/water Ways that life STORES carbon away from its surrounding air/water Plants use light energy from the Sun and CO 2 from the air for the process of photosynthesis. Photosynthesis cis used for the plant to create glucose. Tiny producers in the ocean, called phytoplankton, absorb a massive amount of dissolved carbon dioxide from the water and release oxygen into the atmosphere. Plants change glucose molecules into starches, proteins, and fats, all of which contain carbon atoms—carbon atoms that were once part of the CO 2 in the air. When animals or any other non-producers use Decomposers are organisms that eat the dead or Plants cells undergo respiration and get rid of glucose (respiration), they give off (CO 2) into the decaying organisms. By “eating” this decaying carbon dioxide gas through the stomata in their surrounding atmosphere or water. matter, they return carbon dioxide to the air or leaves. soil through respiration. When plants and animals die on land, the Many types of sea life use carbon dioxide from When sea life dies, their shells fall to the bottom remnants of their bodies are buried over time by the water to make their shells. of the ocean and are buried by sand dirt over dirt, sand, and rock. time.

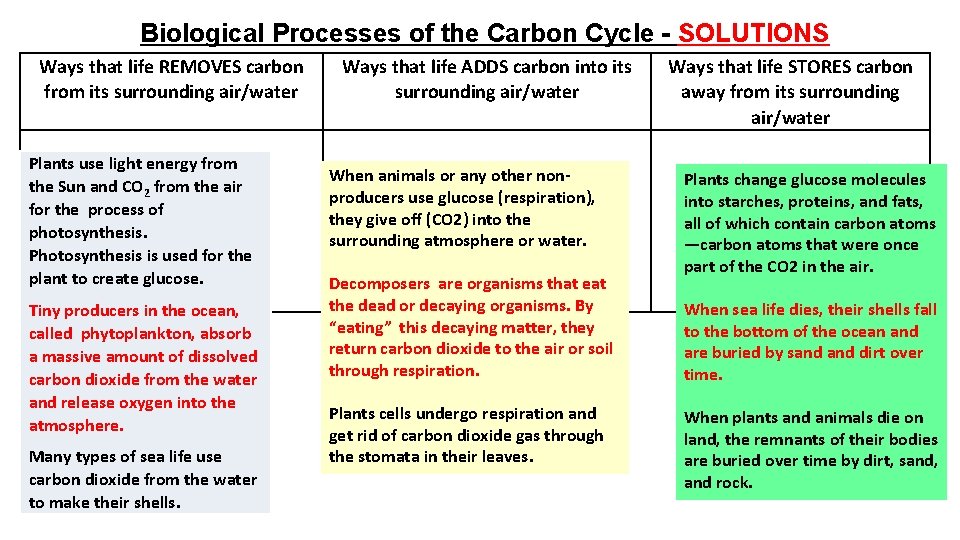
Biological Processes of the Carbon Cycle - SOLUTIONS Ways that life REMOVES carbon from its surrounding air/water Plants use light energy from the Sun and CO 2 from the air for the process of photosynthesis. Photosynthesis is used for the plant to create glucose. Tiny producers in the ocean, called phytoplankton, absorb a massive amount of dissolved carbon dioxide from the water and release oxygen into the atmosphere. Many types of sea life use carbon dioxide from the water to make their shells. Ways that life ADDS carbon into its surrounding air/water When animals or any other nonproducers use glucose (respiration), they give off (CO 2) into the surrounding atmosphere or water. Decomposers are organisms that eat the dead or decaying organisms. By “eating” this decaying matter, they return carbon dioxide to the air or soil through respiration. Plants cells undergo respiration and get rid of carbon dioxide gas through the stomata in their leaves. Ways that life STORES carbon away from its surrounding air/water Plants change glucose molecules into starches, proteins, and fats, all of which contain carbon atoms —carbon atoms that were once part of the CO 2 in the air. When sea life dies, their shells fall to the bottom of the ocean and are buried by sand dirt over time. When plants and animals die on land, the remnants of their bodies are buried over time by dirt, sand, and rock.

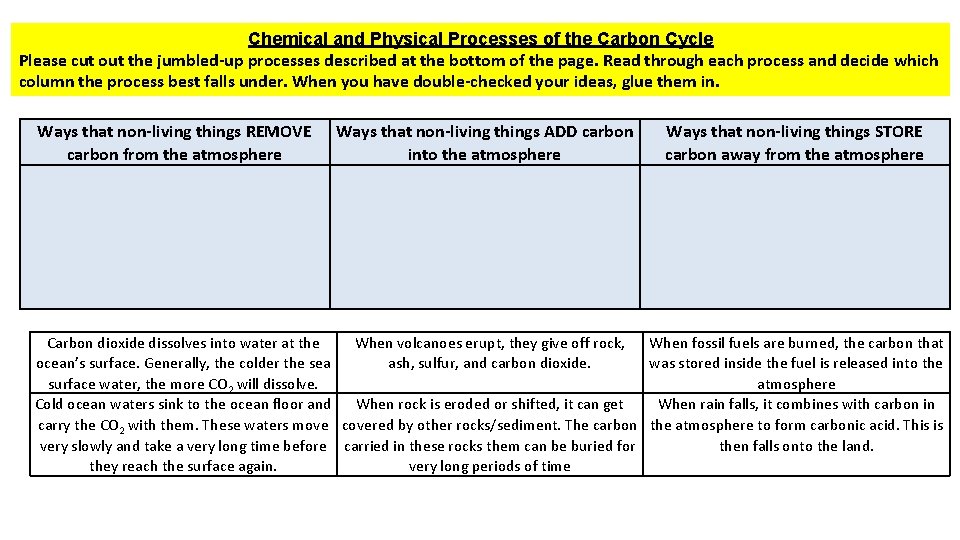
Chemical and Physical Processes of the Carbon Cycle Please cut out the jumbled-up processes described at the bottom of the page. Read through each process and decide which column the process best falls under. When you have double-checked your ideas, glue them in. Ways that non-living things REMOVE carbon from the atmosphere Ways that non-living things ADD carbon into the atmosphere Ways that non-living things STORE carbon away from the atmosphere Carbon dioxide dissolves into water at the When volcanoes erupt, they give off rock, When fossil fuels are burned, the carbon that ocean’s surface. Generally, the colder the sea ash, sulfur, and carbon dioxide. was stored inside the fuel is released into the surface water, the more CO 2 will dissolve. atmosphere Cold ocean waters sink to the ocean floor and When rock is eroded or shifted, it can get When rain falls, it combines with carbon in carry the CO 2 with them. These waters move covered by other rocks/sediment. The carbon the atmosphere to form carbonic acid. This is very slowly and take a very long time before carried in these rocks them can be buried for then falls onto the land. they reach the surface again. very long periods of time

Chemical & Physical Processes of the Carbon Cycle SOLUTIONS Ways that non-living things REMOVE carbon from the atmosphere When rain falls, it combines with carbon in the atmosphere to form carbonic acid. This is then falls onto the land. Carbon dioxide dissolves into water at the ocean’s surface. Generally, the colder the sea surface water, the more CO 2 will dissolve. Ways that non-living things ADD carbon into the atmosphere Ways that non-living things STORE carbon away from the atmosphere When fossil fuels are burned, the carbon that was stored inside the fuel is released into the atmosphere Cold ocean waters sink to the ocean floor and carry the CO 2 with them. These waters move very slowly and take a very long time before they reach the surface again. When volcanoes erupt, they give off rock, ash, sulfur, and carbon dioxide. When rock is eroded or shifted, it can get covered by other rocks/sediment. The carbon carried in these rocks them can be buried for very long periods of time

WORKSHEET