WORKING WITH RECOMBINANT AAV VECTORS BIOSAFETY CONSIDERATIONS OBJECTIVES

WORKING WITH RECOMBINANT AAV VECTORS: BIOSAFETY CONSIDERATIONS

OBJECTIVES This training session will inform you on the following topics: • • The history and current state of human gene therapy Criteria used to categorize biological risk Criteria used to ensure appropriate containment procedures Risk management when working with adeno-associated virus (AAV) vectors

HUMAN GENE THERAPY OVERVIEW & INTRODUCTION TO GENE REPLACEMENT THERAPY
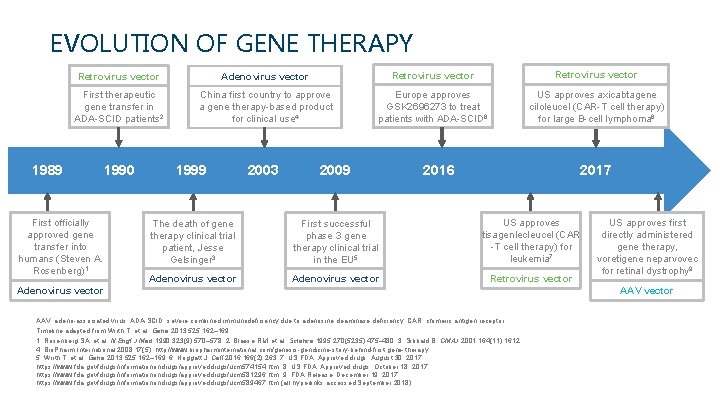
EVOLUTION OF GENE THERAPY 1989 Retrovirus vector Adenovirus vector Retrovirus vector First therapeutic gene transfer in ADA-SCID patients 2 China first country to approve a gene therapy-based product for clinical use 4 Europe approves GSK 2696273 to treat patients with ADA-SCID 6 US approves axicabtagene ciloleucel (CAR-T cell therapy) for large B-cell lymphoma 8 1990 First officially approved gene transfer into humans (Steven A. Rosenberg)1 1999 2003 2009 2016 2017 The death of gene therapy clinical trial patient, Jesse Gelsinger 3 First successful phase 3 gene therapy clinical trial in the EU 5 US approves tisagenlecleucel (CAR -T cell therapy) for leukemia 7 Adenovirus vector Retrovirus vector Adenovirus vector AAV, adeno-associated virus; ADA-SCID, severe combined immunodeficiency due to adenosine deaminase deficiency; CAR, chimeric antigen receptor. Timeline adapted from Wirth T, et al. Gene 2013; 525: 162– 169. 1. Rosenberg SA, et al. N Engl J Med 1990; 323(9): 570– 578. 2. Blaese RM, et al. Science 1995; 270(5235): 475– 480. 3. Sibbald B. CMAJ 2001; 164(11): 1612. 4. Bio. Pharm International 2008; 17(5). http: //www. biopharminternational. com/genesis-gendicine-story-behind-first-gene-therapy. 5. Wirth T, et al. Gene 2013; 525: 162– 169. 6. Hoggatt J. Cell 2016; 166(2): 263. 7. US FDA. Approved drugs. August 30, 2017. https: //www. fda. gov/drugs/informationondrugs/approveddrugs/ucm 574154. htm. 8. US FDA. Approved drugs. October 18, 2017. https: //www. fda. gov/drugs/informationondrugs/approveddrugs/ucm 581296. htm. 9. FDA Release. December 19, 2017. https: //www. fda. gov/drugs/informationondrugs/approveddrugs/ucm 589467. htm (all hyperlinks accessed September 2018). US approves first directly administered gene therapy, voretigene neparvovec for retinal dystrophy 9 AAV vector
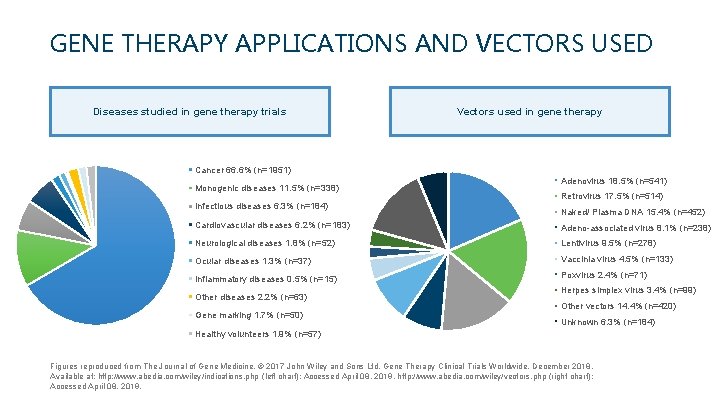
GENE THERAPY APPLICATIONS AND VECTORS USED Diseases studied in gene therapy trials Vectors used in gene therapy Cancer 66. 6% (n=1951) Monogenic diseases 11. 5% (n=338) Infectious diseases 6. 3% (n=184) Adenovirus 18. 5% (n=541) Retrovirus 17. 5% (n=514) Naked/ Plasma DNA 15. 4% (n=452) Cardiovascular diseases 6. 2% (n=183) Adeno-associated virus 8. 1% (n=238) Neurological diseases 1. 8% (n=52) Lentivirus 9. 5% (n=278) Ocular diseases 1. 3% (n=37) Vaccinia virus 4. 5% (n=133) Inflammatory diseases 0. 5% (n=15) Poxvirus 2. 4% (n=71) Other diseases 2. 2% (n=63) Gene marking 1. 7% (n=50) Herpes simplex virus 3. 4% (n=99) Other vectors 14. 4% (n=420) Unknown 6. 3% (n=184) Healthy volunteers 1. 9% (n=57) Figures reproduced from The Journal of Gene Medicine, © 2017 John Wiley and Sons Ltd. Gene Therapy Clinical Trials Worldwide, December 2018. Available at: http: //www. abedia. com/wiley/indications. php (left chart); Accessed April 08, 2019. http: //www. abedia. com/wiley/vectors. php (right chart); Accessed April 08, 2019.
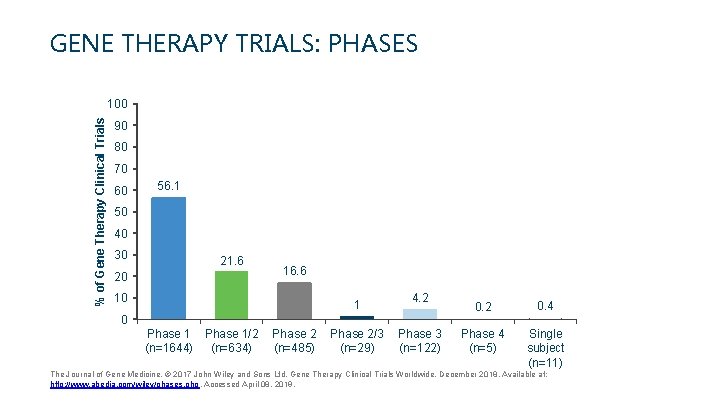
GENE THERAPY TRIALS: PHASES % of Gene Therapy Clinical Trials 100 90 80 70 60 56. 1 50 40 30 20 10 21. 6 16. 6 1 4. 2 0 Phase 1/2 Phase 2/3 Phase 3 (n=1644) (n=634) (n=485) (n=29) (n=122) 0. 2 0. 4 Phase 4 (n=5) Single subject (n=11) The Journal of Gene Medicine, © 2017 John Wiley and Sons Ltd. Gene Therapy Clinical Trials Worldwide, December 2018. Available at: http: //www. abedia. com/wiley/phases. php. Accessed April 08, 2019.
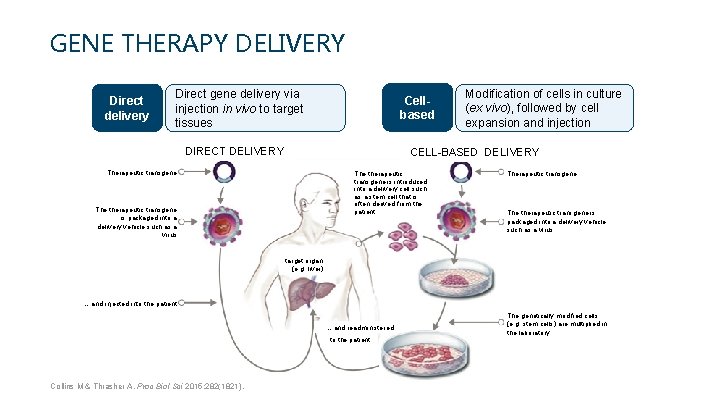
GENE THERAPY DELIVERY Direct delivery Direct gene delivery via injection in vivo to target tissues Cellbased DIRECT DELIVERY Modification of cells in culture (ex vivo), followed by cell expansion and injection CELL-BASED DELIVERY Therapeutic transgene The therapeutic transgene is introduced into a delivery cell such as a stem cell that is often derived from the patient The therapeutic transgene is packaged into a delivery vehicle such as a virus Therapeutic transgene The therapeutic transgene is packaged into a delivery vehicle such as a virus target organ (e. g. liver) … and injected into the patient … and readministered to the patient Collins M & Thrasher A. Proc Biol Sci 2015; 282(1821). The genetically modified cells (e. g. stem cells) are multiplied in the laboratory
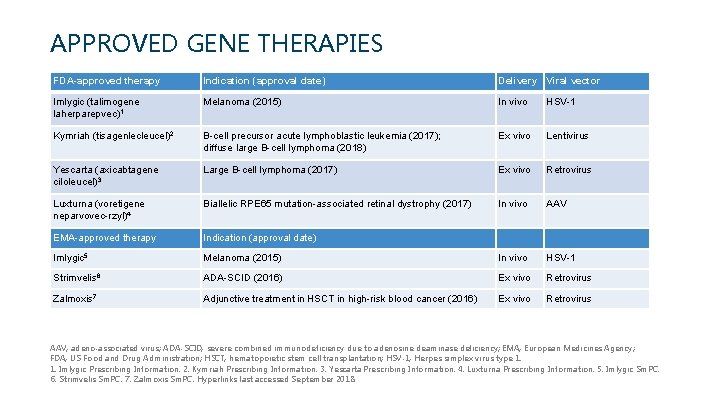
APPROVED GENE THERAPIES FDA-approved therapy Indication (approval date) Delivery Viral vector Imlygic (talimogene laherparepvec)1 Melanoma (2015) In vivo HSV-1 Kymriah (tisagenlecleucel)2 B-cell precursor acute lymphoblastic leukemia (2017); diffuse large B-cell lymphoma (2018) Ex vivo Lentivirus Yescarta (axicabtagene ciloleucel)3 Large B-cell lymphoma (2017) Ex vivo Retrovirus Luxturna (voretigene neparvovec-rzyl)4 Biallelic RPE 65 mutation-associated retinal dystrophy (2017) In vivo AAV EMA-approved therapy Indication (approval date) Imlygic 5 Melanoma (2015) In vivo HSV-1 Strimvelis 6 ADA-SCID (2016) Ex vivo Retrovirus Zalmoxis 7 Adjunctive treatment in HSCT in high-risk blood cancer (2016) Ex vivo Retrovirus AAV, adeno-associated virus; ADA-SCID, severe combined immunodeficiency due to adenosine deaminase deficiency; EMA, European Medicines Agency; FDA, US Food and Drug Administration; HSCT, hematopoietic stem cell transplantation; HSV-1, Herpes simplex virus type 1. 1. Imlygic Prescribing Information. 2. Kymriah Prescribing Information. 3. Yescarta Prescribing Information. 4. Luxturna Prescribing Information. 5. Imlygic Sm. PC. 6. Strimvelis Sm. PC. 7. Zalmoxis Sm. PC. Hyperlinks last accessed September 2018.
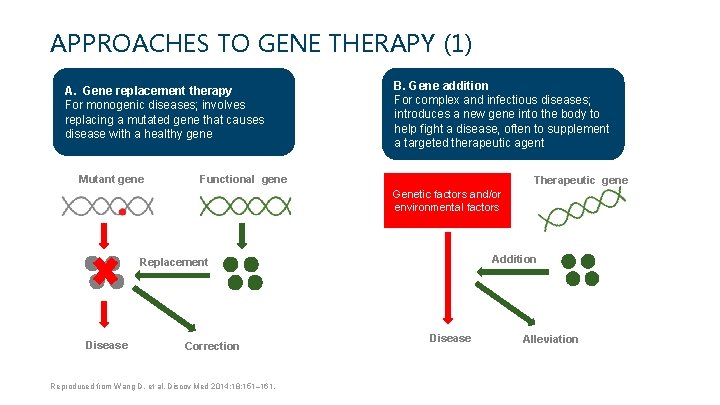
APPROACHES TO GENE THERAPY (1) A. Gene replacement therapy For monogenic diseases; involves replacing a mutated gene that causes disease with a healthy gene Mutant gene B. Gene addition For complex and infectious diseases; introduces a new gene into the body to help fight a disease, often to supplement a targeted therapeutic agent Functional gene Therapeutic gene Genetic factors and/or environmental factors Addition Replacement Disease Correction Reproduced from Wang D, et al. Discov Med 2014; 18: 151– 161. Disease Alleviation
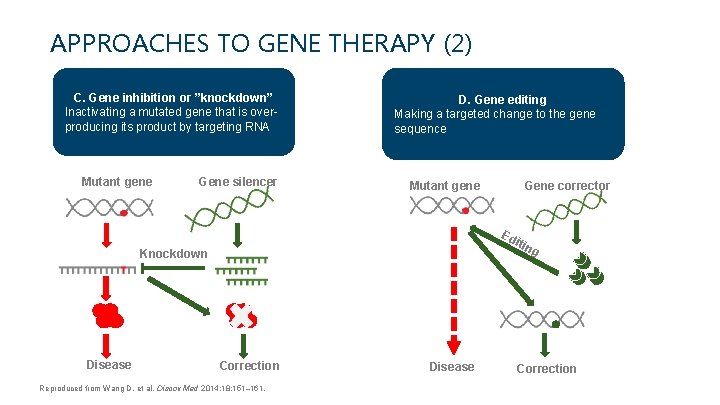
APPROACHES TO GENE THERAPY (2) C. Gene inhibition or ”knockdown” Inactivating a mutated gene that is overproducing its product by targeting RNA Mutant gene Gene silencer D. Gene editing Making a targeted change to the gene sequence Mutant gene Gene corrector Ed itin Knockdown Disease Correction Reproduced from Wang D, et al. Discov Med 2014; 18: 151– 161. Disease g Correction
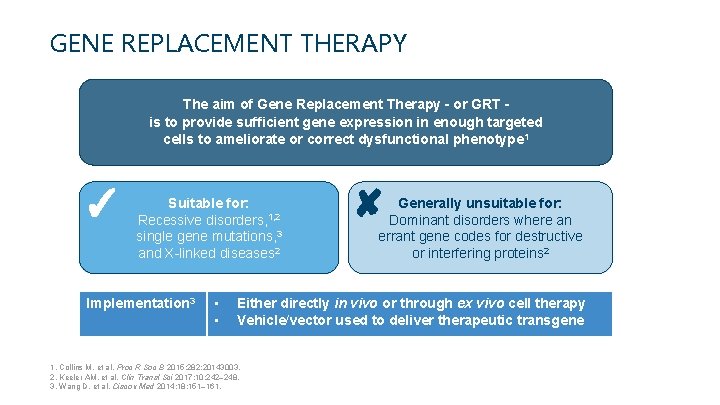
GENE REPLACEMENT THERAPY The aim of Gene Replacement Therapy – or GRT – is to provide sufficient gene expression in enough targeted cells to ameliorate or correct dysfunctional phenotype 1 ✓ Suitable for: Recessive disorders, 1, 2 single gene mutations, 3 and X-linked diseases 2 Implementation 3 • • ✘ Generally unsuitable for: Dominant disorders where an errant gene codes for destructive or interfering proteins 2 Either directly in vivo or through ex vivo cell therapy Vehicle/vector used to deliver therapeutic transgene 1. Collins M, et al. Proc R Soc B 2015; 282: 20143003. 2. Keeler AM, et al. Clin Transl Sci 2017; 10: 242– 248. 3. Wang D, et al. Discov Med 2014; 18: 151– 161.
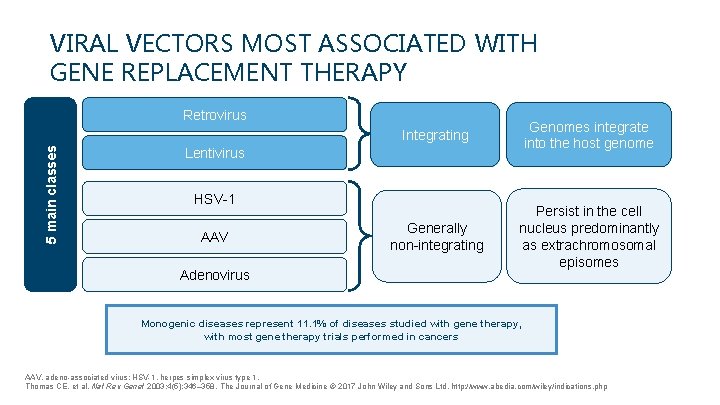
VIRAL VECTORS MOST ASSOCIATED WITH GENE REPLACEMENT THERAPY 5 main classes Retrovirus Integrating Genomes integrate into the host genome Generally non-integrating Persist in the cell nucleus predominantly as extrachromosomal episomes Lentivirus HSV-1 AAV Adenovirus Monogenic diseases represent 11. 1% of diseases studied with gene therapy, with most gene therapy trials performed in cancers AAV, adeno-associated virus; HSV-1, herpes simplex virus type 1. Thomas CE, et al. Nat Rev Genet 2003; 4(5): 346– 358. The Journal of Gene Medicine © 2017 John Wiley and Sons Ltd. http: //www. abedia. com/wiley/indications. php
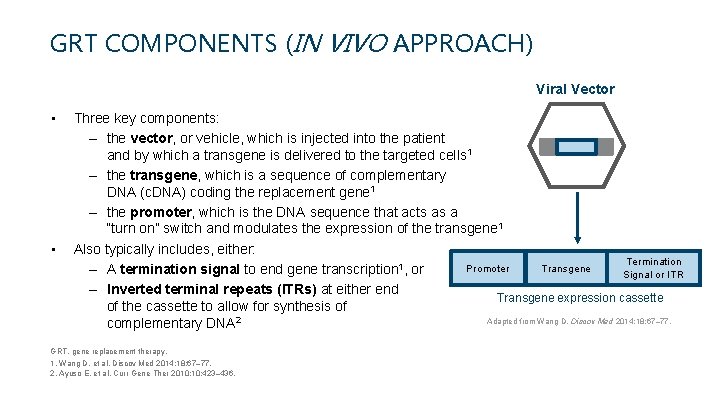
GRT COMPONENTS (IN VIVO APPROACH) Viral Vector • • Three key components: – the vector, or vehicle, which is injected into the patient and by which a transgene is delivered to the targeted cells 1 – the transgene, which is a sequence of complementary DNA (c. DNA) coding the replacement gene 1 – the promoter, which is the DNA sequence that acts as a “turn on” switch and modulates the expression of the transgene 1 Also typically includes, either: Termination Transgene Promoter – A termination signal to end gene transcription 1, or Signal or ITR – Inverted terminal repeats (ITRs) at either end Transgene expression cassette of the cassette to allow for synthesis of Adapted from Wang D. Discov Med 2014; 18: 67– 77. complementary DNA 2 GRT, gene replacement therapy. 1. Wang D, et al. Discov Med 2014; 18: 67– 77. 2. Ayuso E, et al. Curr Gene Ther 2010; 10: 423– 436.

AAV VECTORS

BIOLOGICAL PROPERTIES OF AAV 1 AAV is a member of the parvovirus family of single-stranded small DNA viruses AAV Requires a helper virus such as adenovirus or herpes simplex virus for replication 1 Considered a Risk Group 1 microorganism – infection with AAV is not associated with disease in humans 2 Has several serotypes that impact tropism (susceptible tissues), but all appear to be non-pathogenic 3 • • • r. AAV-based vectors used in GRT have wildtype viral genes removed and typically exist as extrachromosomal episomes 1, 3 AAV can efficiently infect both non-dividing and dividing cells 1 AAV is typically transmitted by respiratory and gastrointestinal routes 1, 4 AAV, adeno-associated virus; GRT, gene replacement therapy. 1. Deyle DR, Russell DW. Curr Opin Mol Ther 2009; 11(4): 442– 447. 2. NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Molecules. April 2016. https: //osp. od. nih. gov/wp-content/uploads/2013/06/NIH_Guidelines. pdf. 3. Gonçalves MA, et al. Virol J 2005; 2: 43– 60. 4. Gombash SE. Postdoc J 2015; 3: 1– 12; Environmental Health & Safety. Adeno-Associated Virus and Adeno-Associated Viral Vectors. https: //ehs. research. uiowa. edu/adeno-associated-virus-and-adeno-associatedviral-vectors (hyperlinks last accessed September 2018).
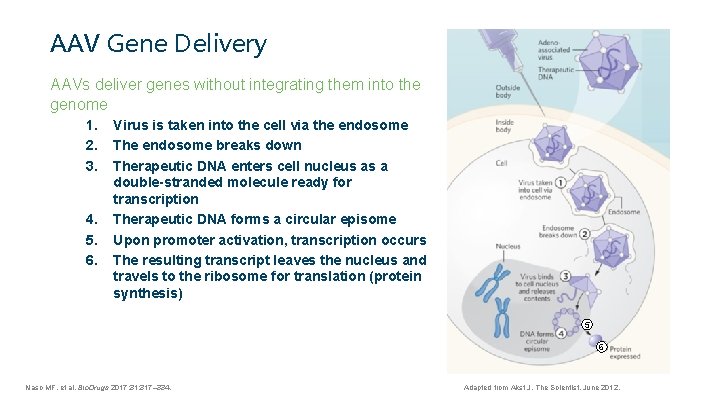
AAV Gene Delivery AAVs deliver genes without integrating them into the genome 1. 2. 3. 4. 5. 6. Virus is taken into the cell via the endosome The endosome breaks down Therapeutic DNA enters cell nucleus as a double-stranded molecule ready for transcription Therapeutic DNA forms a circular episome Upon promoter activation, transcription occurs The resulting transcript leaves the nucleus and travels to the ribosome for translation (protein synthesis) 5 6 Naso MF, et al. Bio. Drugs 2017; 31: 317– 334. Adapted from Akst J. The Scientist. June 2012.
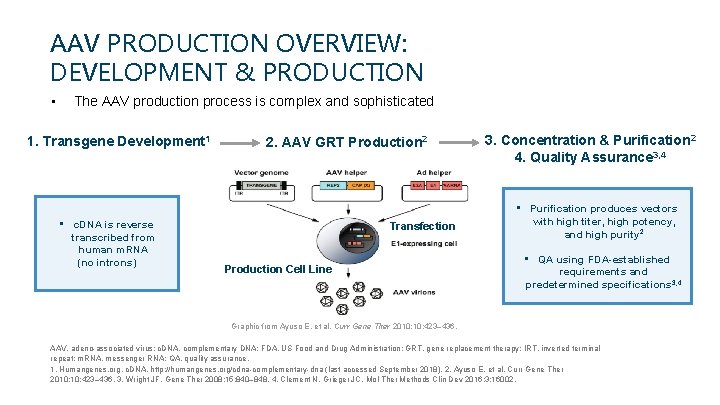
AAV PRODUCTION OVERVIEW: DEVELOPMENT & PRODUCTION • The AAV production process is complex and sophisticated 1. Transgene Development 1 • c. DNA is reverse transcribed from human m. RNA (no introns) 2. AAV GRT Production 2 Transfection Production Cell Line 3. Concentration & Purification 2 4. Quality Assurance 3, 4 • Purification produces vectors with high titer, high potency, and high purity 2 • QA using FDA-established requirements and predetermined specifications 3, 4 Graphic from Ayuso E, et al. Curr Gene Ther 2010; 10: 423– 436. AAV, adeno-associated virus; c. DNA, complementary DNA; FDA, US Food and Drug Administration; GRT, gene replacement therapy; IRT, inverted terminal repeat; m. RNA, messenger RNA; QA, quality assurance. 1. Humangenes. org. c. DNA. http: //humangenes. org/cdna-complementary-dna (last accessed September 2018). 2. Ayuso E, et al. Curr Gene Ther 2010; 10: 423– 436. 3. Wright JF. Gene Ther 2008; 15: 840– 848. 4. Clement N, Grieger JC. Mol Ther Methods Clin Dev 2016; 3: 16002.

AAV SAFETY CONSIDERATIONS Transgenes remain transcriptionally active indefinitely, making AAV vectors efficient one-time delivery vehicles, but transcription cannot subsequently be deactivated 1 Although wildtype viral genes have been deleted from the AAV vector, therapeutic genes present in AAV vectors may have unexpected properties (if non-self)2, 3 Prior exposure to wild-type AAV virus has the potential to result in an immune response to the AAV capsid; however, little to no capsid-specific cellular response has been seen in immuneprivileged tissues, such as the CNS 4 AAV, adeno-associated virus. 1. Negrete A, Kotin RM. Brief Funct Genomic Proteomic 2008; 7: 303– 311. 2. Naso FN, et al. Bio. Drugs 2017; 31: 317– 334. 3. Watakabe A, et al. Neurosci Res 2015; 93: 145– 157. 3. NIH Guidelines. https: //osp. od. nih. gov/wp-content/uploads/2013/06/NIH_Guidelines. pdf (last accessed September 2018). 4. Vandamme C et al. Hum Gene Ther 2017; 28: 1061– 1074.

SUMMARY • Gene therapy is a novel technique that uses manufactured transgenes to address the underlying genetic cause of a disease 1 – GRT aims to provide sufficient gene expression in enough targeted cells to ameliorate or correct a disease phenotype 2 • Recent accomplishments in gene delivery techniques have advanced safety and efficacy with viral vectors 3 – AAVs are one of the most promising vectors for gene transfer and expression in monogenic disorders AAV, adeno-associated virus; GRT, gene replacement therapy. 1. Foust KD & Kaspar BK. In: Spinal Muscular Atrophy: Disease Mechanisms and Therapy. London, UK: Academic Press. 2017; 313– 323. 2. Collins M, et al. Proc R Soc B 2015; 282: 20143003. 3. Wang D, et al. Mol Ther Methods Clin Dev 2018; 9: 234– 246.

ESTABLISHING BIOLOGICAL RISK & APPROPRIATE CONTAINMENT FOR GENE THERAPY AGENTS

RISK GROUPS The NIH Guidelines and WHO categorize wildtype infectious agents into risk groups: 1, 2 Risk Group 1 Risk Group 2 Risk Group 3 Risk Group 4 Agents that are not associated with disease in healthy adult humans Agents that are associated with human disease which is rarely serious and for which preventative or therapeutic interventions are often available Agents that are associated with serious or lethal human disease for which preventive or therapeutic may be available (high individual risk but low community risk) Agents that are likely to cause serious or lethal human disease for which preventive or therapeutic interventions are not usually available (high individual risk and high community risk) NIH, National Institutes of Health; WHO, World Health Organization. 1. NIH Guidelines. https: //osp. od. nih. gov/wp-content/uploads/2013/06/NIH_Guidelines. pdf. 2. WHO LSM. http: //www. who. int/csr/resources/publications/biosafety/en/Biosafety 7. pdf. (hyperlinks last accessed September 2018).
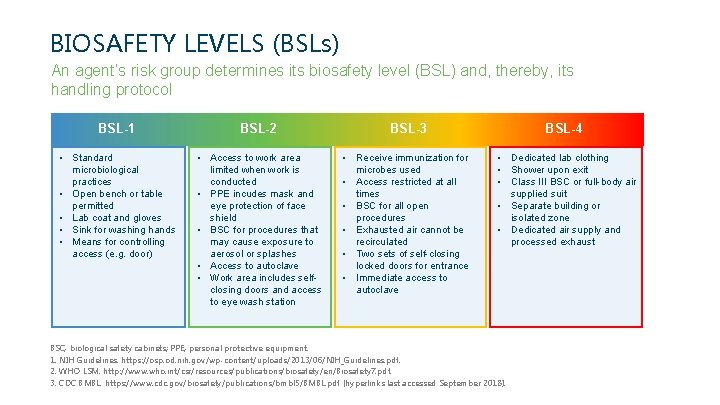
BIOSAFETY LEVELS (BSLs) An agent’s risk group determines its biosafety level (BSL) and, thereby, its handling protocol BSL-1 BSL-2 BSL-3 • Standard microbiological practices • Open bench or table permitted • Lab coat and gloves • Sink for washing hands • Means for controlling access (e. g. door) • Access to work area limited when work is conducted • PPE incudes mask and eye protection of face shield • BSC for procedures that may cause exposure to aerosol or splashes • Access to autoclave • Work area includes selfclosing doors and access to eye wash station • Receive immunization for microbes used • Access restricted at all times • BSC for all open procedures • Exhausted air cannot be recirculated • Two sets of self-closing locked doors for entrance • Immediate access to autoclave BSL-4 • Dedicated lab clothing • Shower upon exit • Class III BSC or full-body air supplied suit • Separate building or isolated zone • Dedicated air supply and processed exhaust BSC, biological safety cabinets; PPE, personal protective equipment. 1. NIH Guidelines. https: //osp. od. nih. gov/wp-content/uploads/2013/06/NIH_Guidelines. pdf. 2. WHO LSM. http: //www. who. int/csr/resources/publications/biosafety/en/Biosafety 7. pdf. 3. CDC BMBL. https: //www. cdc. gov/biosafety/publications/bmbl 5/BMBL. pdf (hyperlinks last accessed September 2018).

AAV IS CLASSED AS BIOSAFETY LEVEL 1 (BSL-1) • • Microbes that are not known to cause disease in healthy adults Practices – Standard microbiological practices – Open bench or table permitted – Decontaminate work surfaces; use disinfectant following spills; decontaminate materials before disposal using an effective method Equipment – Lab coat and gloves Facilities – Sink for washing hands – Means for controlling access (e. g. door) AAV, adeno-associated virus. CDC BMBL. https: //www. cdc. gov/biosafety/publications/bmbl 5/BMBL. pdf (last accessed September 2018). BSL-4 High-risk microbes BSL-3 BSL-2 BSL-1 Low-risk microbes
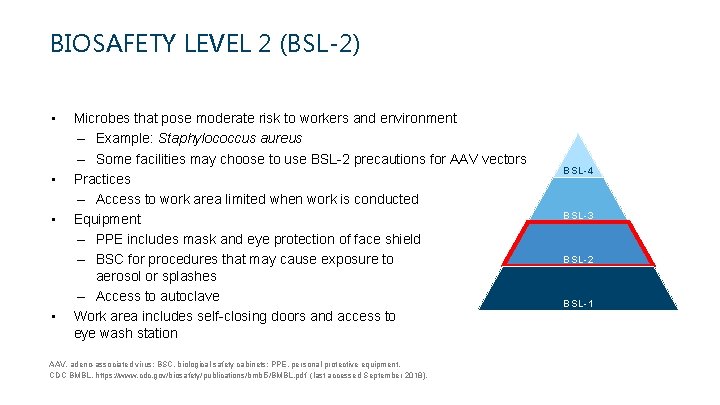
BIOSAFETY LEVEL 2 (BSL-2) • • Microbes that pose moderate risk to workers and environment – Example: Staphylococcus aureus – Some facilities may choose to use BSL-2 precautions for AAV vectors Practices – Access to work area limited when work is conducted Equipment – PPE includes mask and eye protection of face shield – BSC for procedures that may cause exposure to aerosol or splashes – Access to autoclave Work area includes self-closing doors and access to eye wash station AAV, adeno-associated virus; BSC, biological safety cabinets; PPE, personal protective equipment. CDC BMBL. https: //www. cdc. gov/biosafety/publications/bmbl 5/BMBL. pdf (last accessed September 2018). BSL-4 BSL-3 BSL-2 BSL-1
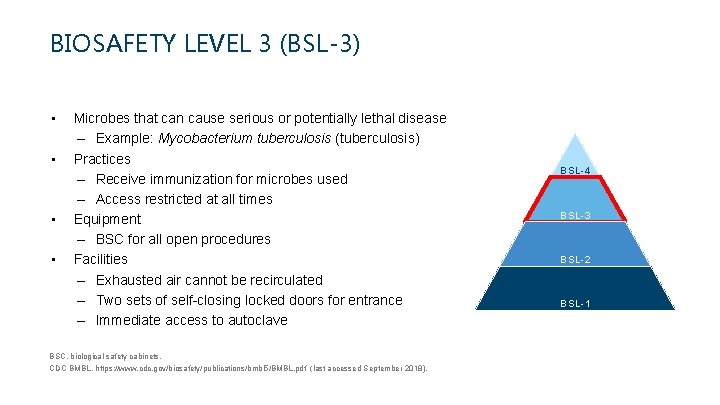
BIOSAFETY LEVEL 3 (BSL-3) • • Microbes that can cause serious or potentially lethal disease – Example: Mycobacterium tuberculosis (tuberculosis) Practices – Receive immunization for microbes used – Access restricted at all times Equipment – BSC for all open procedures Facilities – Exhausted air cannot be recirculated – Two sets of self-closing locked doors for entrance – Immediate access to autoclave BSC, biological safety cabinets. CDC BMBL. https: //www. cdc. gov/biosafety/publications/bmbl 5/BMBL. pdf (last accessed September 2018). BSL-4 BSL-3 BSL-2 BSL-1

BIOSAFETY LEVEL 4 (BSL-4) • • • Very few labs in the world Most exotic and dangerous microbes (e. g. Ebola virus) Practices – Dedicated lab clothing – Shower upon exit Equipment – Class III BSC or full-body, air-supplied suit Facilities – Separate building or isolated zone – Dedicated air supply and processed exhaust BSC, biological safety cabinets. CDC BMBL. https: //www. cdc. gov/biosafety/publications/bmbl 5/BMBL. pdf (last accessed September 2018). BSL-4 BSL-3 BSL-2 BSL-1

SAFETY FEATURES OF AAV VECTORS r. AAV* is considered Risk Group 1 – infection with AAV is not associated with disease in humans 1 r. AAV vectors are replication-incompetent (non-infectious) by design 2 Absent of hazardous genes*, r. AAV vectors can be handled at BSL-1 containment 1, 3 r. AAV vectors are susceptible to common disinfectants approved for bloodborne pathogens (e. g. EPA Lists B, D, and E)4 AAV, adeno-associated virus; BSL, biosafety level; EPA, Environmental Protection Agency. *In which the transgene does not encode either a potentially tumorigenic gene product or a toxin molecule, and construct is produced in the absence of a helper virus. 1. IBC Guidance on Biosafety Level Assignment for AAV. https: //www. pittsburgh. va. gov/Research/docs/AAV-Guidance. pdf. 2. Gonçalves MA, et al. Virol J 2005; 2: 43– 60. 3. Vector Safety Information. https: //gtp. med. upenn. edu/uploads/attachments/cjgo 427 wq 00 oit 1 gd 35 bjpgmi-vector-safety-information. pdf; 4. United States EPA. Selected EPA-registered Disinfectants. https: //www. epa. gov/pesticide-registration/selected-epa-registered-disinfectants (hyperlinks last accessed September 2018).

AAV BIOSAFETY CONSIDERATIONS: STAFF CONCERNS • • Use good standard microbiological practices and BSL-1 containment 1 Conduct all open procedures with AAV vectors inside of BSC (class not specified for BSL-1) 1 � Use of BSC at higher levels may be used to ensure sterility of product • • Wear lab coat, gloves, and mask/face shield when working with AAV vectors 2 Wash hands with soap and water after working with AAV vectors 2 • In the event of exposure: 3 • Wash exposed area with soap and water In event of eye exposure, flush in eye wash for 15 minutes • Report incident per institutional policy AAV, adeno-associated virus; BSC, biological safety cabinets; BSL, biosafety level. 1. NIH Guidelines. https: //osp. od. nih. gov/wp-content/uploads/2013/06/NIH_Guidelines. pdf. 2. CDC BMBL. https: //www. cdc. gov/biosafety/publications/bmbl 5/BMBL. pdf. 3. Environmental Health & Safety. Adeno-Associated Virus and Adeno-Associated Viral Vectors. https: //ehs. research. uiowa. edu/adeno-associated-virusand-adeno-associated-viral-vectors (hyperlinks last accessed September 2018).

AAV BIOSAFETY CONSIDERATIONS: AVOIDANCE OF CROSS-CONTAMINATION Clean work surfaces after all open procedures with AAV vectors 1 • • Use disinfectants approved for bloodborne pathogens (EPA Lists B, D, E)2 10% dilution of household bleach is effective (freshly prepared) Dispose of all contaminated materials (including gloves) as biohazardous waste 3, 4 • • • Red containers with biohazard symbol Yellow chemo/biohazard mixed waste containers Ask your hazardous waste disposal company if you are not sure Limit access to work area by untrained staff while working with AAV vectors 3 AAV, adeno-associated virus; EPA, Environmental Protection Agency. 1. Biological Safety. https: //ehs. uky. edu/docs/pdf/bio_ia_uk_viral_vector_guidance_0001. pdf. 2. United States EPA. Selected EPA-registered Disinfectants. https: //www. epa. gov/pesticide-registration/selected-epa-registered-disinfectants. 3. NIH Guidelines. https: //osp. od. nih. gov/wp-content/uploads/2013/06/NIH_Guidelines. pdf. 4. CDC BMBL. https: //www. cdc. gov/biosafety/publications/bmbl 5/BMBL. pdf. 5. Pharmaceutical compounding-sterile preparations (general information chapter 797). In: The United States pharmacopeia, 27 th rev. , and The national formulary, 22 nd ed. Rockville, MD: The United States Pharmacopeial Convention, 2004: 2350– 2370 (hyperlinks accessed September 2018).

SUMMARY • • • Basic biological risk and containment criteria have been established by the NIH 1, CDC 2, WHO 3, and OSHA 4 The biological risks from working with AAV vectors are considered very low 5 AAV vectors can be handled at BSL-1, the lowest biosafety level 6 Care should nevertheless be taken to mitigate exposure to AAV vectors 7 All materials contaminated with AAV vectors should be disposed of as biohazardous waste 7 AAV, adeno-associated virus; BSL, biosafety level; CDC, Centers for Disease Control and Prevention; NIH, National Institutes of Health; OSHA, Occupational Safety & Health Administration; WHO, World Health Organization. 1. NIH Guidelines. https: //osp. od. nih. gov/wp-content/uploads/2013/06/NIH_Guidelines. pdf. 2. CDC BMBL. https: //www. cdc. gov/biosafety/publications/ bmbl 5/BMBL. pdf. 3. WHO LSM. http: //www. who. int/csr/resources/publications/biosafety/en/Biosafety 7. pdf. 4. OSHA. Healthcare Wide Hazards. (Lack of) Universal Precautions. https: //www. osha. gov/SLTC/etools/hospital/hazards/univprec/univ. html. 5. Gonçalves MA, et al. Virol J 2005; 2: 43. 6. IBC Guidance on Biosafety Level Assignment for AAV. https: //www. pittsburgh. va. gov/Research/docs/AAV-Guidance. pdf. 7. Environmental Health & Safety. Adeno-Associated Virus and Adeno-Associated Viral Vectors. https: //ehs. research. uiowa. edu/adeno-associated-virus-and-adeno-associated-viral-vectors (hyperlinks last accessed September 2018).
- Slides: 30