Working with Genetically Modified Organisms GMOs Genetically Modified
































- Slides: 37

Working with Genetically Modified Organisms (GMO’s)

Genetically Modified Organism (GMO) • An organism modified by gene technology. • An organism that has inherited particular traits from an organism (the initial organism) being traits that occurred in the initial organism because of gene technology. • Anything declared by the regulations to be a genetically modified organism, or that belongs to a class of things declared by the regulations to be a genetically modified organism.

ENIGMA’S! If DNA is taken from an organism, spliced to a vector, and replaced in the same organism, then that organism is a GMO. Just because an organism is not considered to be a GMO in another jurisdiction (USA, . EU) does not mean it is not a GMO in Australia. Just because someone else has permission to work with a GMO does not mean that you do. You still have to apply for permission. Although you may not have constructed the GMO, if you are working with it you still have to apply for permission. Plant, animal, or fungal cells containing recombinant DNA are considered to be GMO’s.

What is not a GMO? • Does not include – – – mutants. fusion of animal cells unless the product can form an animal. plants formed x protoplast fusion. plants formed by embryo rescue or invitro fertilisation or zygote implantation. organisms formed by natural DNA transfer.

History of GMO regulation in Australia. • Genetic Manipulation Advisory Committee (GMAC) 1980’s-2001 • Established regulatory framework for regulating research with GMO’s. • Carried out assessment of proposals. • Provided advice on containment conditions. • No statutory powers

The Gene Technology Act 2000 • Establishes a uniform regulatory system throughout the states and territories. • The object of this Act is to protect the health and safety of people, and to protect the environment, by identifying risks posed by or as a result of gene technology, and by managing those risks through regulating certain dealings with GMOs.

Purpose • To ensure that all work with Genetically Manipulated Organisms (GMO’s) within Australia is carried out in such a way that threats to human health and the environment are minimised.

The Regulator. • To administer the regulations under the act, the Federal government established the Office of the Gene Technology Regulator (OGTR). • The OGTR is a statutory office-holder independent from commercial or political influences. • The regulator cannot be directed by anyone when making a decision on whether to grant a licence. • Reports direct to Fed Health Minister. • Currently Dr Sue Meek.

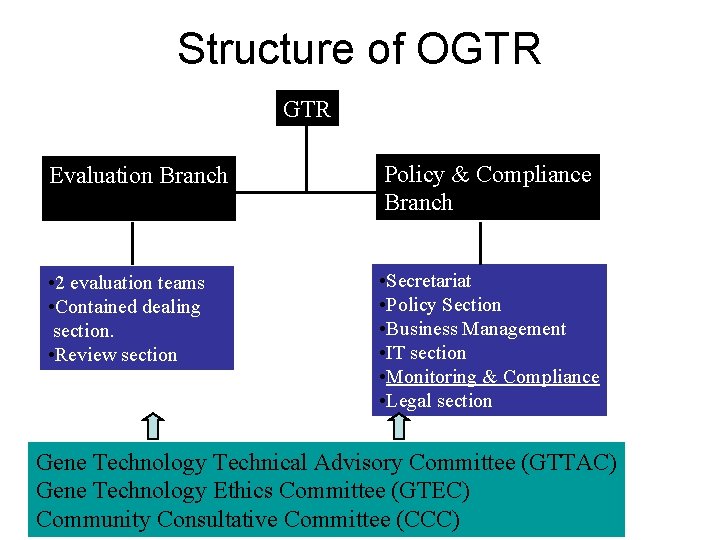
Structure of OGTR Evaluation Branch Policy & Compliance Branch • 2 evaluation teams • Contained dealing section. • Review section • Secretariat • Policy Section • Business Management • IT section • Monitoring & Compliance • Legal section Gene Technology Technical Advisory Committee (GTTAC) Gene Technology Ethics Committee (GTEC) Community Consultative Committee (CCC)

Function of GTTAC • To provide scientific and technical advice on – Gene technology, GMO and GMO products – Applications made under the act – The biosafety aspects of gene technology – The need for policy principles, policy guidelines, in relation to GMOs and GMO products and the content of such principles guidelines and codes.

Role of the CCC • To provide advice on: – Matters of general concern in relation to applications made under this act. – Matters of general concern in relation to GMOs – The need for policy principles, guidelines, codes of practice and technical and procedural guidelines in relation to GMOs and GM products. – The content of such principles guidelines and codes.

Function of GTEC • To provide advice on: – Ethical issues relating to Gene Technology – The need for, and content of Codes of Practice in relation to ethics in respect of dealings with GMOs – The need for and content of policy principles in relation to dealings with GMOs that should not be conducted for ethical reasons.

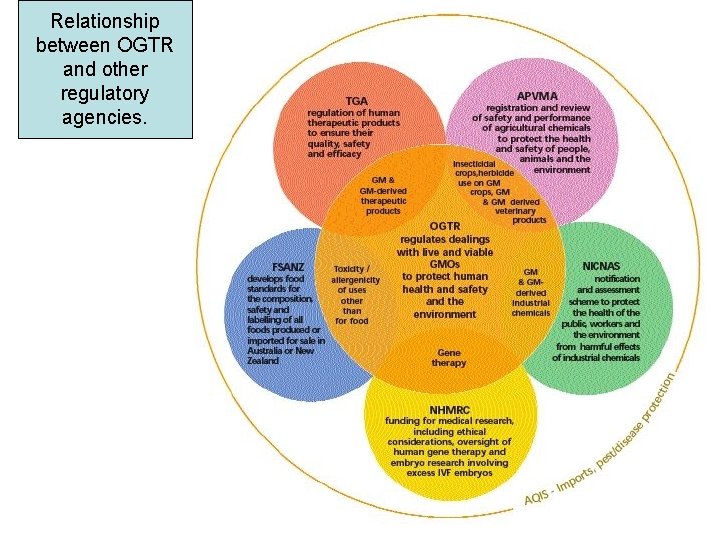
Relationship between OGTR and other regulatory agencies.

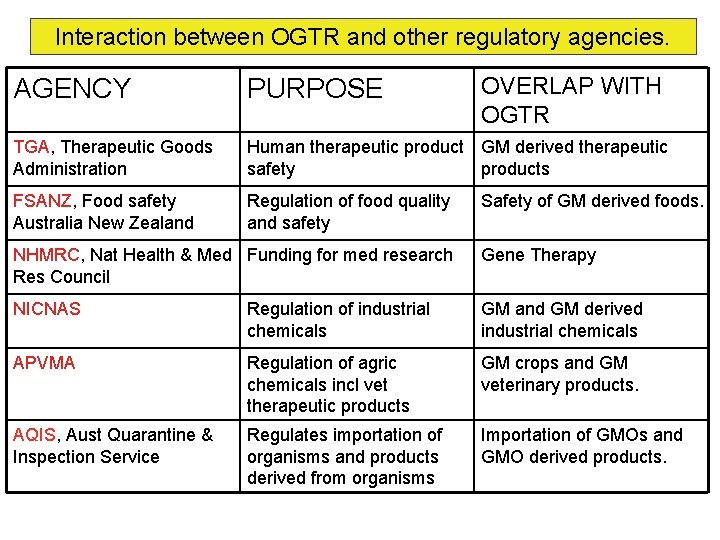
Interaction between OGTR and other regulatory agencies. OVERLAP WITH OGTR AGENCY PURPOSE TGA, Therapeutic Goods Administration Human therapeutic product GM derived therapeutic safety products FSANZ, Food safety Australia New Zealand Regulation of food quality and safety Safety of GM derived foods. NHMRC, Nat Health & Med Funding for med research Res Council Gene Therapy NICNAS Regulation of industrial chemicals GM and GM derived industrial chemicals APVMA Regulation of agric chemicals incl vet therapeutic products GM crops and GM veterinary products. AQIS, Aust Quarantine & Inspection Service Regulates importation of organisms and products derived from organisms Importation of GMOs and GMO derived products.

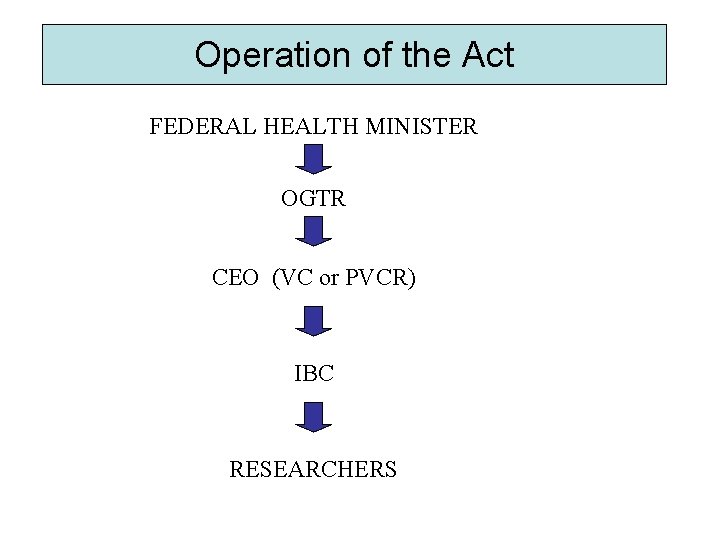
Operation of the Act FEDERAL HEALTH MINISTER OGTR CEO (VC or PVCR) IBC RESEARCHERS

Organisations. • All organizations involved in dealings are accredited by the OGTR. • Every accredited organisation has access to an Inst. Biosafety Committee (IBC) which monitors compliance with the regulations within that organisation.

Role of the IBC • • Maintains a register of all researchers and projects. Processes applications to work with GMO’s. Notifies the OGTR of all such work. Ensures compliance with regulations. Carries out inspection of facilities. Provides training to researchers. Provides an annual report to the OGTR. Advises the CEO on matters relating to dealings with GMO’s within that organisation.

Statutory Requirements • All dealings with GMO’s no matter how trivial must be notified to the OGTR through the organisation’s IBC. • All personnel dealing with GMO’s must attend a training course. • Dealings must only be carried out in certified premises unless permission is given for other facilities (eg. , DIR’s) • Failure to notify will incur penalties.

Penalties • For dealing without a licence. – – fines up to $55, 000 imprisonment for 2 years • Aggravated offence. – – fine of $1. 1 m imprisonment up to 5 years.

More Likely Penalties • Loss of accreditation by organisation. – corporate culture of non-compliance. • Rejection of subsequent applications. – breaches must be indicated on each proposal. • Adverse publicity. – breaches of the regulations are reported in parliament.

RESPONSIBILITIES I: The Organisation • CEO (or delegate) is responsible to ensure: • Adequate staffing and resources are available to maintain quality assurance systems for compliance. • A competent indemnified IBC is part of the quality assurance system. • That facilities are appropriate for the conduct of work with GMOs.

RESPONSIBILITIES II: Heads of Schools/Sections. • Ensure safe working procedures are understood and enforced. • Involves provision of training to new researchers and • Requires compliance by researchers.

RESPONSIBILITIES III: Principal Researchers • Ensure safe working and GM procedures are understood and enforced. • Provide an SOP manual. • Ensure that all dealings are licenced/ notified. • Ensure that all staff are aware of specific licence conditions. • Advise the IBC immediately of breaches of conditions/regulations, unexplained illnesses or absences or changes to dealings (names of new staff). • Maintain a register of all GM projects. • Inform IBC when projects terminate.

RESPONSIBILITIES IV: Researchers. • Undertake GTR training. • Adhere to SOP’s for GM facilities. • Notify your supervisor or the IBC of any hazards, incidents or injuries. • Notify your supervisor and the IBC of any changes to the project. • Check that your project has been notified to the IBC.

RESPONSIBILITIES V: IBC • • Maintain a register of all dealings and staff. Maintaining a list of facilities. Inspecting facilities. Provide training. Assess proposals and advise CEO. Liase with OGTR. Provide an annual report to OGTR. Develop procedures to ensure compliance

Facilities • All work with GMOs (except emempt dealings) must be carried out in certified premises • Apply to OGTR to have facilities approved • Facilities must conform to the standards specified in ASNZ standards 2243. 3 2002 • Facilities must be inspected every year by the IBC. • Facilities will also be spot inspected by OGTR • Work practices must conform to ASNZ std 2243. 3 2002

Required work practices for certified labs! Access to the lab is restricted to authorised personnel. Must always wear a lab coat and remove it before leaving the lab. Must wash hands before leaving the lab. If storing cultures outside the facility they must be stored in an unbreakable container in a lockable fridge/freezer/room. If transporting GM material between facilities or to the autoclave they must be contained in an unbreakable container. Any breaches of containment must be reported to the IBC immediately.

If I fail to notify how would they find out? • • Published papers. Theses Annual Report from University. Information from funding agencies. Conference presentations. Press releases. Informants

Types of Dealings • • Exempt Notifiable Low Risk Dealing (NLRD). Dealings Not Involving Release (DNIR). Dealings Involving Release (DIR). Increasing risk

Exempt Dealings • Notify the IBC of the dealing using the exempt form on the IBC website (Edusafe). • Start work. • You do not need permission of the IBC or the OGTR. The only requirement is to notify them. • Can apply to have dealings added to the exempt list.

Exempt Dealing Form

Notifiable Low Risk Dealings (NLRD) • • Obtain form from OGTR website Fill in form and submit to IBC. Proposal is evaluated by the IBC. Cannot start work until permission is granted by the IBC. • The proposal is approved by the IBC and OGTR notified in annual report.

Dealings Not Involving Release (DNIR) • • Submit proposal to IBC. Proposal is evaluated and submitted to OGTR. Proposal is sent out for public consultation. OGTR prepares a Risk Assessment Risk Management Plan (RAMP). This is sent out for consultation. The conditions of the proposal are finalised. A licence is issued within 90 working days. Work cannot start until the licence is issued.

Dealings Involving Release (DIR) • • Submit proposal to IBC. Proposal is evaluated and submitted to OGTR. Proposal is sent out for public consultation. OGTR prepares a Risk Assessment Risk Management Plan (RAMP). This is sent out for consultation. The conditions of the proposal are finalised. A licence is issued within 180 working days of receipt of proposal. Work cannot start until the licence is issued.

DIR Considerations • Potential for spread through pollen and seeds. • Presence of pollinator species. • Proximity of compatible species. • Persistence in seed bed. • Weediness. • Proximity to waterways.

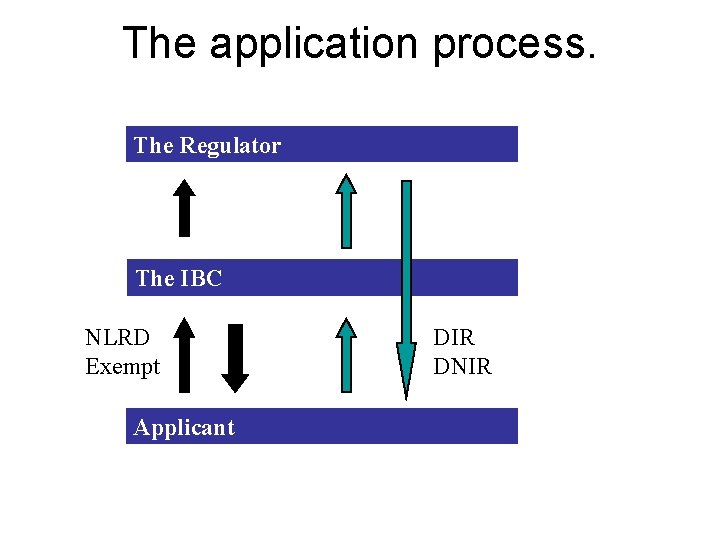
The application process. The Regulator The IBC NLRD Exempt Applicant DIR DNIR

If your project involves working with, or creation of GMO’s then: • Check with your supervisor that appropriate permission has been obtained. – Projects (unless exempt) will have an OGTR reference number. Ask your supervisor for that number. This number should be listed alongside all organisms used in the project. • If permission has not been obtained, then you should apply for permission. • To do this visit the IBC website • Even if your project is exempt you need to notify the IBC of the project. • Changes to the project should be notified as a variation to the licence.