Worked Example 9 1 Formation of Solutions Which























- Slides: 24

Worked Example 9. 1 Formation of Solutions Which of the following pairs of substances would you expect to form solutions? (a) Carbon tetrachloride (CCl 4) and hexane (C 6 H 14). (b) Octane (C 8 H 18) and methyl alcohol (CH 3 OH). Analysis Identify the kinds of intermolecular forces in each substance (Section 8. 2). Substances with similar intermolecular forces tend to form solutions. Solution (a) Hexane contains only C — H and C — C bonds, which are nonpolar. Carbon tetrachloride contains polar C — Cl bonds, but they are distributed symmetrically in the tetrahedral molecule so that it too is nonpolar. The major intermolecular force for both compounds is London dispersion forces, so they will form a solution. (b) Octane contains only C — H and C — C bonds and so is nonpolar; the major intermolecular force is dispersion. Methyl alcohol contains polar C — O and O — H bonds; it is polar and forms hydrogen bonds. The intermolecular forces for the two substances are so dissimilar that they do not form a solution. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

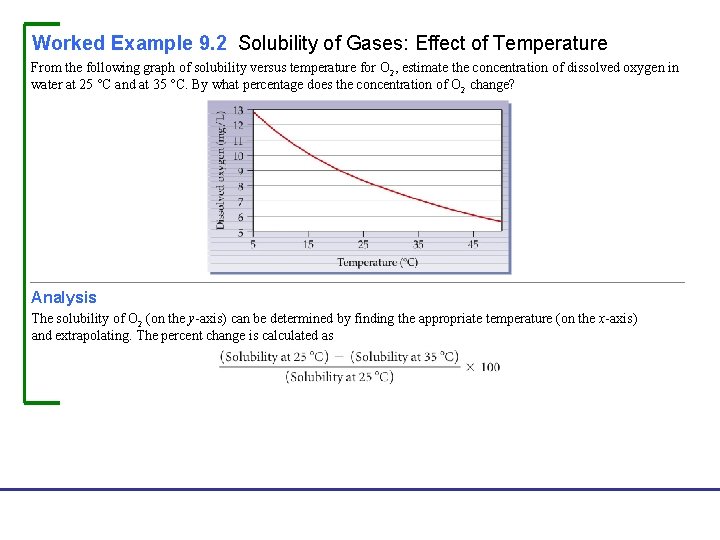
Worked Example 9. 2 Solubility of Gases: Effect of Temperature From the following graph of solubility versus temperature for O 2, estimate the concentration of dissolved oxygen in water at 25 °C and at 35 °C. By what percentage does the concentration of O 2 change? Analysis The solubility of O 2 (on the y-axis) can be determined by finding the appropriate temperature (on the x-axis) and extrapolating. The percent change is calculated as Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Continued Solution From the graph we estimate that the solubility of O 2 at 25 °C is approximately 8. 3 mg/L and at 35 °C is 7. 0 mg/L. The percent change in solubility is Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 3 Solubility of Gases: Henry’s Law At a partial pressure of oxygen in the atmosphere of 159 mm. Hg, the solubility of oxygen in blood is 0. 44 g/100 m. L. What is the solubility of oxygen in blood at 11, 000 ft, where the partial pressure of O 2 is 56 mm. Hg? Analysis According to Henry’s law, the solubility of the gas divided by its pressure is constant: Of the four variables in this equation, we know P 1, C 1, and P 2, and we need to find C 2. Ballpark Estimate The pressure drops by a factor of about 3 (from 159 mm. Hg to 56 mm. Hg). Since the ratio of solubility to pressure is constant, the solubility must also drop by a factor of 3 (from 0. 44 g/100 m. L to about 0. 15 g/100 m. L). Solution STEP 1: Identify known information. We have values for P 1, C 1, and P 2. P 1 = 159 mm. Hg C 1 = 0. 44 g/100 m. L P 2 = 56 mm. Hg STEP 2: Identify answer and units. We are looking for the solubility of O 2 (C 2) at a partial pressure P 2. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson Solubility of O 2, C 2 = ? ? g/100 m. L © 2013 Pearson Education, Inc.

Continued STEP 3: Identify conversion factors or equations. In this case, we restate Henry’s law to solve for C 2. STEP 4: Solve. Substitute the known values into the equation and calculate C 2. Ballpark Check The calculated answer matches our estimate. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 4 Mass Percent as Conversion Factor: Mass of Solution to Mass of Solute The percentage of gold in jewelry is typically reported in carats, with 24 carats representing 100% gold. A sample of 18 -carat gold would contain 18 grams of gold in 24 grams of metal, which would equal a (m/m)% of 75%. Calculate the mass of gold in a 5. 05 g ring that is 18 -carat gold. Analysis We are given a concentration and the total mass of the sample solution (the gold alloy in the ring), and we need to find the mass of gold by rearranging the equation for (m/m)% concentration. Ballpark Estimate A 75% (m/m) solution contains 75 g for every 100 g of solution, so 10 g contains 7. 5 g. The mass of the ring is a little more than 5 g (or half of 10 g) so the amount of gold in the ring will be slightly more than half of 7. 5 g, or ~ 3. 8 g gold. Solution Ballpark Check The calculated answer is consistent with our estimate of 3. 8 g gold. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 5 Volume Percent as Conversion Factor: Volume of Solution to Volume of Solute How many milliliters of methyl alcohol are needed to prepare 75 m. L of a 5. 0% (v/v) solution? Analysis We are given a solution volume (75 m. L) and a concentration [5. 0% (v/v), meaning 5. 0 m. L solute/100 m. L solution]. The concentration acts as a conversion factor finding the amount of methyl alcohol needed. Ballpark Estimate A 5% (v/v) solution contains 5 m. L of solute in 100 m. L of solution, so the amount of solute in 75 m. L of solution must be about three-fourths of 5 m. L, which means between 3 and 4 m. L. Solution Ballpark Check The calculated answer is consistent with our estimate of between 3 and 4 m. L. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 6 Solution Concentration: Mass/Volume Percent A solution of heparin sodium, an anticoagulant for blood, contains 1. 8 g of heparin sodium dissolved to make a final volume of 15 m. L of solution. What is the mass/volume percent concentration of this solution? Analysis Mass/volume percent concentration is defined as the mass of the solute in grams divided by the volume of solution in milliliters and multiplied by 100%. Ballpark Estimate The mass of solute (1. 8 g) is smaller than the volume of solvent (15 m. L) by a little less than a factor of 10. The weight/volume percent should thus be a little greater than 10%. Solution Ballpark Check The calculated (m/v)% is reasonably close to our original estimate of 10%. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 7 Mass/Volume Percent as Conversion Factor: Volume to Mass How many grams of Na. Cl are needed to prepare 250 m. L of a 1. 5% (m/v) saline solution? Analysis We are given a concentration and a volume, and we need to find the mass of solute by rearranging the equation for (m/v)% concentration. Ballpark Estimate The desired (m/v)% value, 1. 5%, is between 1 and 2%. For a volume of 250 m. L, we would need 2. 5 g of solute for a 1% (m/v) solution and 5. 0 g of solute for a 2% solution. Thus, for our 1. 5% solution, we need a mass midway between 2. 5 and 5. 0 g, or about 3. 8 g. Solution Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 7 Mass/Volume Percent as Conversion Factor: Volume to Mass Continued Ballpark Check The calculated answer matches our estimate. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 8 ppm as Conversion Factor: Mass of Solution to Mass of Solute The maximum allowable concentration of chloroform, CHCl 3, in drinking water is 100 ppb. What is the maximum amount (in grams) of chloroform allowed in a glass containing 400 g (400 m. L) of water? Analysis We are given a solution amount (400 g) and a concentration (100 ppb). This concentration of 100 ppb means This equation can be rearranged to find the mass of solute. Ballpark Estimate A concentration of 100 ppb means there are 100 10– 9 g (1 10– 7 g) of solute in 1 g of solution. In 400 g of solution, we should have 400 times this amount, or 400 10– 7 = 4 10– 5 g. Solution Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 8 ppm as Conversion Factor: Mass of Solution to Mass of Solute Continued Ballpark Check The calculated answer matches our estimate. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 9 Solution Concentration: Molarity What is the molarity of a solution made by dissolving 2. 355 g of sulfuric acid (H 2 SO 4) in water and diluting to a final volume of 50. 0 m. L? The molar mass of H 2 SO 4 is 98. 1 g/mol. Analysis Molarity is defined as moles of solute per liter of solution: M = mol/L. Thus, we must first find the number of moles of sulfuric acid by doing a mass to mole conversion, and then divide the number of moles by the volume of the solution. Ballpark Estimate The molar mass of sulfuric acid is about 100 g/mol, so 2. 355 g is roughly 0. 025 mol. The volume of the solution is 50. 0 m. L, or 0. 05 L, so we have about 0. 025 mol of acid in 0. 05 L of solution, which is a concentration of about 0. 5 M. Solution STEP 1: Identify known information. We know the mass of sulfuric acid and the final volume of solution. STEP 2: Identify answer including units. We need to find the molarity (M) in units of moles per liter. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 9 Solution Concentration: Molarity Continued STEP 3: Identify conversion factors and equations. We know both the amount of solute and the volume of solution, but first we must make two conversions: convert mass of H 2 SO 4 to moles of H 2 SO 4, using molar mass as a conversion factor, and convert volume from milliliters to liters. STEP 4: Solve. Substitute the moles of solute and volume of solution into the molarity expression. Ballpark Check The calculated answer is close to our estimate, which was 0. 5 M. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 10 Molarity as Conversion Factor: Molarity to Mass A blood concentration of 0. 065 M ethyl alcohol (Et. OH) is sufficient to induce a coma. At this concentration, what is the total mass of alcohol (in grams) in an adult male whose total blood volume is 5. 6 L? The molar mass of ethyl alcohol is 46. 0 g/mol. (Refer to the flow diagram in Figure 9. 7 to identify which conversions are needed. ) Analysis We are given a molarity (0. 065 M) and a volume (5. 6 L), which allows us to calculate the number of moles of alcohol in the blood. A mole to mass conversion then gives the mass of alcohol. Solution Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 11 Molarity as Conversion Factor: Molarity to Volume In our stomachs, gastric juice that is about 0. 1 M in HCl aids in digestion. How many milliliters of gastric juice will react completely with an antacid tablet that contains 500 mg of magnesium hydroxide? The molar mass of Mg(OH)2 is 58. 3 g/mol, and the balanced equation is Analysis We are given the molarity of HCl and need to find the volume. We first convert the mass of Mg(OH) 2 to moles and then use the coefficients in the balanced equation to find the moles of HCl that will react. Once we have the moles of HCl and the molarity in moles per liter, we can find the volume. Solution Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 12 Dilution of Solutions: Concentration What is the final concentration if 75 m. L of a 3. 5 M glucose solution is diluted to a volume of 450 m. L? Analysis The number of moles of solute is constant, so Mc. Vc = Md. Vd Of the four variables in this equation, we know the initial concentration Mc (3. 5 M), the initial volume Vc (75 m. L), and the final volume Vd (450 m. L), and we need to find the final concentration Md. Ballpark Estimate The volume increases by a factor of 6, from 75 m. L to 450 m. L, so the concentration must decrease by a factor of 6, from 3. 5 M to about 0. 6 M. Solution Solving the above equation for Md and substituting in the known values gives Ballpark Check The calculated answer is close to our estimate of 0. 6 M. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 13 Dilution of Solutions: Volume Aqueous Na. OH can be purchased at a concentration of 1. 0 M. How would you use this concentrated solution to prepare 750 m. L of 0. 32 M Na. OH? Analysis The number of moles of solute is constant, so Mc. Vc = Md. Vd Of the four variables in this equation, we know the initial concentration Mc (1. 0 M), the final volume Vd (750 m. L), and the final concentration Md (0. 32 M), and we need to find the initial volume Vc. Ballpark Estimate We want the solution concentration to decrease by a factor of about 3, from 1. 0 M to 0. 32 M, which means we need to dilute the 1. 0 M solution by a factor of 3. This means the final volume must be about three times greater than the initial volume. Because our final volume is to be 750 m. L, we must start with an initial volume of about 250 m. L. Solution Solving the above equation for V 1 and substituting in the known values gives To prepare the desired solution, dilute 240 m. L of 1. 0 M Na. OH with water to make a final volume of 750 m. L. Ballpark Check The calculated answer (240 m. L) is reasonably close to our estimate of 250 m. L. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 14 Equivalents as Conversion Factors: Volume to Mass The normal concentration of Ca 2+ in blood is 5. 0 m. Eq/L. How many milligrams of Ca 2+ are in 1. 00 L of blood? Analysis We are given a volume and a concentration in milliequivalents per liter, and we need to find an amount in milligrams. Thus, we need to calculate the gramequivalent for Ca 2+ and then use concentration as a conversion factor between volume and mass, as indicated in the following flow diagram: Ballpark Estimate The molar mass of calcium is 40. 08 g/mol, and the calcium ion carries a charge of 2+. Thus, 1 g-Eq of Ca 2+ equals about 20 g/Eq or 20 mg/m. Eq. This means that the 5. 0 m. Eq of Ca 2+ ions in 1. 00 L of blood corresponds to a mass of 5. 0 m. Eq Ca 2+ 20 mg/m. Eq = 100 mg Ca 2+. Solution Ballpark Check The calculated answer (100 mg of Ca 2+ in 1. 00 L of blood) matches our estimate. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 15 Properties of Solutions: Boiling Point Elevation What is the boiling point of a solution of 0. 75 mol of KBr in 1. 0 kg of water? Analysis The boiling point increases 0. 51 °C for each mole of solute per kilogram of water. Since KBr is a strong electrolyte, there are 2 moles of ions (K+ and Br–) for every 1 mole of KBr that dissolves. Ballpark Estimate The boiling point will increase about 0. 5 °C for every 1 mol of ions in 1 kg of water. Since 0. 75 mol of KBr produce 1. 5 mol of ions, the boiling point should increase by (1. 5 mol ions) (0. 5 °C/mol ions) = 0. 75 °C. Solution The normal boiling point of pure water is 100 °C, so the boiling point of the solution increases to 100. 77 °C. Ballpark Check The 0. 77 °C increase is consistent with our estimate of 0. 75 °C. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 16 Properties of Solutions: Freezing Point Depression The cells of a tomato contain mostly an aqueous solution of sugar and other substances. If a typical tomato freezes at – 2. 5 °C, what is the concentration of dissolved particles in the tomato cells (in moles of particles per kg of water)? Analysis The freezing point decreases by 1. 86 °C for each mole of solute dissolved in 1 kg of water. We can use the decrease in freezing point (2. 5 °C) to find the amount of solute per kg of water. Ballpark Estimate The freezing point will decrease by about 1. 9 °C for every 1 mol of solute particles in 1 kg of water. To lower the freezing point by 2. 5 °C (about 30% more) will require about 30% more solute, or 1. 3 mol. Solution We can rearrange this expression to Ballpark Check The calculated answer agrees with our estimate of 1. 3 mol/kg. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 17 Properties of Solutions: Osmolarity The solution of glucose commonly used intravenously has a concentration of 5. 0% (m/v) glucose. What is the osmolarity of this solution? The molar mass of glucose is 180 g/mol. Analysis Since glucose is a molecular substance that does not give ions in solution, the osmolarity of the solution is the same as the molarity. Recall from Section 9. 7 that a solution of 5. 0% (m/v) glucose has a concentration of 5. 0 g glucose per 100 m. L of solution, which is equivalent to 50 g per liter of solution. Thus, finding the molar concentration of glucose requires a mass to mole conversion. Ballpark Estimate One liter of solution contains 50 g of glucose (MW = 180 g/mol). Thus, 50 g of glucose is equal to a little more than 0. 25 mol, so a solution concentration of 50 g/L is equal to about 0. 25 osmol, or 0. 25 M. Solution STEP 1: Identify known information. We know the (m/v)% concentration of the glucose solution. STEP 2: Identify answer and units. We are looking for osmolarity, which in this case is equal to the molarity of the solution because glucose is a molecular substance and does not dissociate into ions. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson Osmolarity = Molarity = ? ? mol/liter © 2013 Pearson Education, Inc.

Worked Example 9. 17 Properties of Solutions: Osmolarity Continued STEP 3: Identify conversion factors. The (m/v)% concentration is defined as grams of solute per 100 m. L of solution, and molarity is defined as moles of solute per liter of solution. We will need to convert from milliliters to liters and then use molar mass to convert grams of glucose to moles of glucose. STEP 4: Solve. Starting with the (m/v)% glucose concentration, we first find the number of grams of glucose in 1 L of solution and then convert to moles of glucose per liter. Ballpark Check The calculated osmolarity is reasonably close to our estimate of 0. 25 osmol. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.

Worked Example 9. 18 Properties of Solutions: Osmolarity What mass of Na. Cl is needed to make 1. 50 L of a 0. 300 osmol solution? The molar mass of Na. Cl is 58. 44 g/mol. Analysis Since Na. Cl is an ionic substance that produces 2 mol of ions (Na+, Cl–) when it dissociates, the osmolarity of the solution is twice the molarity. From the volume and the osmolarity we can determine the moles of Na. Cl needed and then perform a mole to mass conversion. Solution STEP 1: Identify known information. We know the volume and the osmolarity of the final Na. Cl solution. STEP 2: Identify answer and units. We are looking for the mass of Na. Cl. STEP 3: Identify conversion factors. Starting with osmolarity in the form (moles Na. Cl/L), we can use volume to determine the number of moles of solute. We can then use molar mass for the mole to mass conversion. STEP 4: Solve. Use the appropriate conversions, remembering that Na. Cl produces two ions per formula unit, to find the mass of Na. Cl. Fundamentals of General, Organic, and Biological Chemistry, 7 e John Mc. Murry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson © 2013 Pearson Education, Inc.