Word equations to balanced chemical equations drill 2

Word equations to balanced chemical equations drill 2 b Each of the following slides first gives you a word equation. You must write the balanced chemical equation, with phases, then click ahead to check your work. Each slide is numbered, so you can do some, come back and then do more. Practice makes perfect.

Aluminum and oxygen form aluminum oxide

SKELETON Aluminum and oxygen form aluminum oxide _Al(S) + _O 2(G) → _Al 2 O 3(S) Al+3 cation and O-2 anion, criss cross to get the right formula.

Aluminum and oxygen form aluminum oxide _Al(S) + _O 2(G) → _Al 2 O 3(S) SAY: one aluminum up front in reactants, but two aluminums in the product, put a 2 on the reactant side to start.

Aluminum and oxygen form aluminum oxide 2 Al(S) + _O 2(G) → _Al 2 O 3(S) SAY: two aluminums up front in reactants, and two aluminums in the product, good. SAY: there are 2 oxygens in the reactants, but three in the products. Even 2 and Odd 3, bad. To fix this, we double the odd one, by doubling up Al 2 O 3

Aluminum and oxygen form aluminum oxide 2 Al(S) + _O 2(G) → 2 Al 2 O 3(S) SAY: two aluminums up front in reactants, and four aluminums in the product, bad. To fix this we must change the 2 Al in the reactants to a four.

Aluminum and oxygen form aluminum oxide 4 2 Al (S) + _O 2(G) → 2 Al 2 O 3(S) SAY: now we’ve got 2 Al in reactants, but 4 in products, so change the first “ 2” into a four, then start again.

Aluminum and oxygen form aluminum oxide 4 Al(S) + _O 2(G) → 2 Al 2 O 3(S) 2 Al 2 O 3 Means, Al 2 O 3 and another Al 2 O 3 That’s 6 oxygens! SAY: 4 Al in reactants, and 4 Al in products (good). Only 2 oxygen in reactants, but SIX in product. HOW DO WE GET 6 OXGYEN in reactants?

Aluminum and oxygen form aluminum oxide 4 Al(S) + 3 O 2(G) → 2 Al 2 O 3(S) Put a 3 in front of O 2, and now it’s balanced. It’s a 4: 3: 2 ratio, simple whole numbers, just like our friend John Dalton, and your teacher like. All good.

Pentane gas (C 5 H 10) combusts

SKELETON Pentane combusts _C 5 H 10(G) + _O 2(G) → _CO 2(G) + _H 2 O(G) Combustion is ALWAYS a hydrocarbon – in this case the pentane – combining with pure oxygen, and then ALWAYS forming carbon dioxide and water.
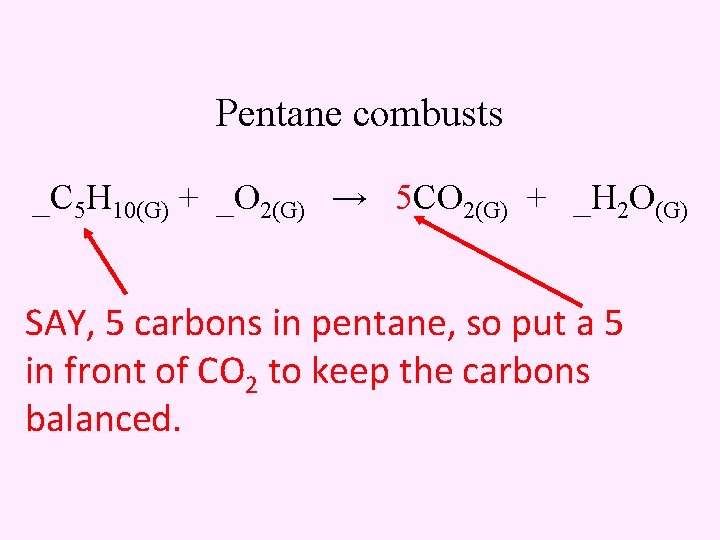
Pentane combusts _C 5 H 10(G) + _O 2(G) → 5 CO 2(G) + _H 2 O(G) SAY, 5 carbons in pentane, so put a 5 in front of CO 2 to keep the carbons balanced.

Pentane combusts _C 5 H 10(G) + _O 2(G) → 5 CO 2(G) + 5 H 2 O(G) Next, there are 10 hydrogens in pentane, we need to put a 5 in front of water, to make the hydrogens balance.
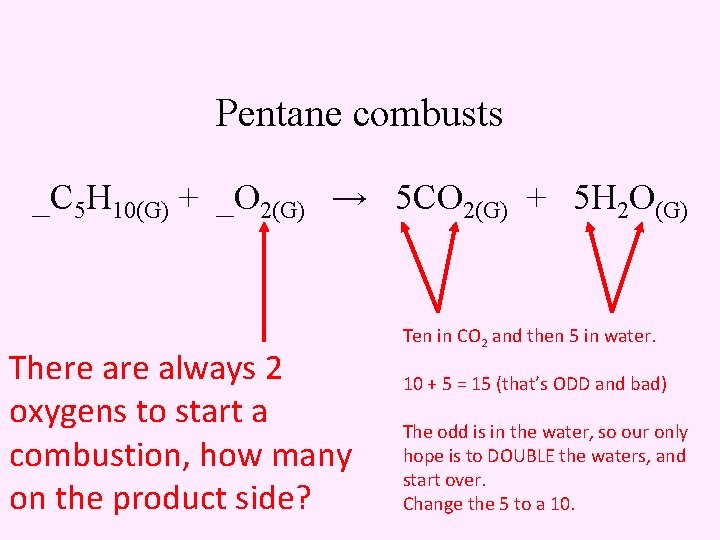
Pentane combusts _C 5 H 10(G) + _O 2(G) → 5 CO 2(G) + 5 H 2 O(G) There always 2 oxygens to start a combustion, how many on the product side? Ten in CO 2 and then 5 in water. 10 + 5 = 15 (that’s ODD and bad) The odd is in the water, so our only hope is to DOUBLE the waters, and start over. Change the 5 to a 10.

Pentane combusts _C 5 H 10(G) + _O 2(G) → 5 CO 2(G) + 10 H 2 O(G) There always 2 oxygens to start a combustion, how many on the product side? Ten in CO 2 and then 5 in water. 10 + 5 = 15 (that’s ODD and bad) The odd is in the water, so our only hope is to DOUBLE the waters, and start over. Change the 5 to a 10.

Pentane combusts 2 C 5 H 10(G) + _O 2(G) → 5 CO 2(G) + 10 H 2 O(G) Still 5 carbon in pentane and 5 CO 2 (good so far). 10 H in pentane, but now 20 H in the water. Put a 2 in front of pentane to balance the “H”, start again.

Pentane combusts 2 C 5 H 10(G) + _O 2(G) → 10 CO 2(G) + 10 H 2 O(G) SAY: 10 C in pentane, only 5 in CO 2, fix that five into a 10, then start over again. (yeohhh! Crazy huh? )
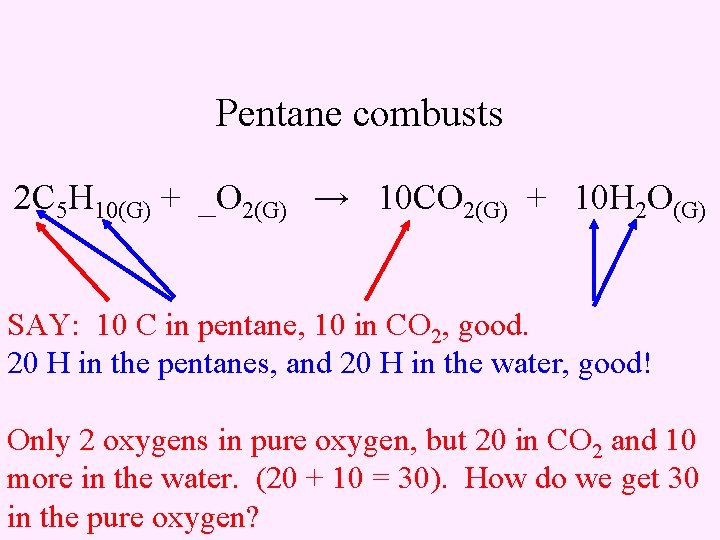
Pentane combusts 2 C 5 H 10(G) + _O 2(G) → 10 CO 2(G) + 10 H 2 O(G) SAY: 10 C in pentane, 10 in CO 2, good. 20 H in the pentanes, and 20 H in the water, good! Only 2 oxygens in pure oxygen, but 20 in CO 2 and 10 more in the water. (20 + 10 = 30). How do we get 30 in the pure oxygen?

Pentane combusts 2 C 5 H 10(G) + 15 O 2(G) → 10 CO 2(G) + 10 H 2 O(G) Change the pure oxygen to a 15 O 2 (15 x 2 = 30) now balanced.

Rubidium metal is put into calcium nitrate solution
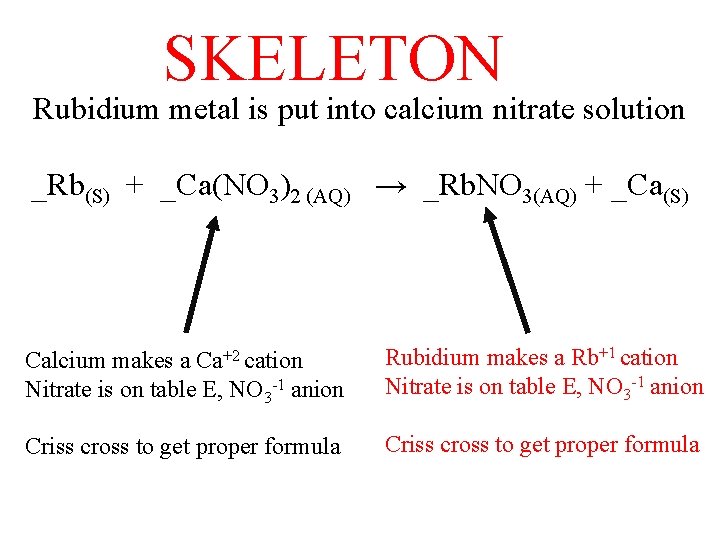
SKELETON Rubidium metal is put into calcium nitrate solution _Rb(S) + _Ca(NO 3)2 (AQ) → _Rb. NO 3(AQ) + _Ca(S) Calcium makes a Ca+2 cation Nitrate is on table E, NO 3 -1 anion Rubidium makes a Rb+1 cation Nitrate is on table E, NO 3 -1 anion Criss cross to get proper formula

Rubidium metal is put into calcium nitrate solution _Rb(S) + _Ca(NO 3)2 (AQ) → 2 Rb. NO 3(AQ) +_ Ca(S) SAY: 1 Rb in reactants, and 1 Rb in products, so far so good. Then… 1 Ca in reactants, and 1 a in products, also good. But… two nitrates in the reactant side, so DOUBLE the Rb. NO 3 to double nitrates in the products (then start over).
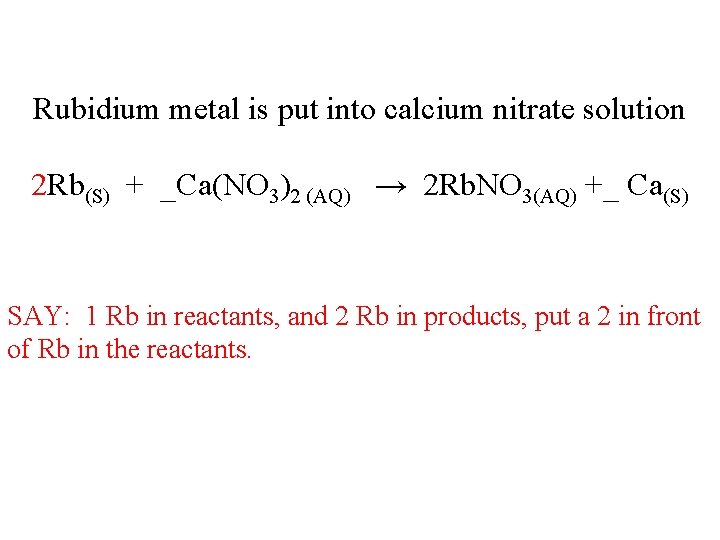
Rubidium metal is put into calcium nitrate solution 2 Rb(S) + _Ca(NO 3)2 (AQ) → 2 Rb. NO 3(AQ) +_ Ca(S) SAY: 1 Rb in reactants, and 2 Rb in products, put a 2 in front of Rb in the reactants.

Rubidium metal is put into calcium nitrate solution 2 Rb(S) + _Ca(NO 3)2 (AQ) → 2 Rb. NO 3(AQ) +_ Ca(S) SAY: Rb good 2: 2 Ca good at 1: 1 nitrates good at 2: 2 Balanced! In a 2: 1: 2: 1 ratio – John Dalton would be happy, I’m happy. You be happy too.

Rubidium metal is put into calcium nitrate solution 2 Rb(S) + Ca(NO 3)2 (AQ) → 2 Rb. NO 3(AQ) + Ca(S) Why you might have got this one wrong: Rubidium makes only a +1 cation so forming rubidium nitrate can only be in a 1: 1 ratio. The “ 2” in the calcium nitrate needs to be BALANCED away, not by you sticking a 2 in as in: Rb(NO 3)2 because it’s easier to do. This: Rb(NO 3)2 is just not possible.

Cobalt (III) chlorate solution and magnesium chromate solutions are mixed together

SKELETON Cobalt (III) chlorate solution and magnesium chromate solutions are mixed together _Co(Cl. O 3)3 (AQ) + _Mg. Cr. O 4(AQ) → This is the set up for a double replacement reaction, we’re starting with 2 AQ solutions. Don’t just do this in your head, in our class we will make the switch, and balance the equation. If by chance both products are AQ also, that means it was NO REACTION. For practice balancing, we always go through the motions. You can’t decide no reaction until it’s balanced and THEN you go to table F to find the solid precipitate, if it’s even there.

Cobalt (III) chlorate solution and magnesium chromate solutions are mixed together _Co(Cl. O 3)3 (AQ) + _Mg. Cr. O 4(AQ) → _Co Cr. O 4(? ) + _Mg. Cl. O 3(? ) Switch the anions around, here the chromate anion starts with the Mg, but ends up with the Co. The chlorate anion starts with Co, but ends up with the Mg. It’s a double switch (like a square dance, switch your partners, do si do). FIX IN NEXT STEP! Note, we have NO IDEA YET about the solubility of the products, nor would we look before balancing the equation. Switch, fix, balance, and then table “F” them.
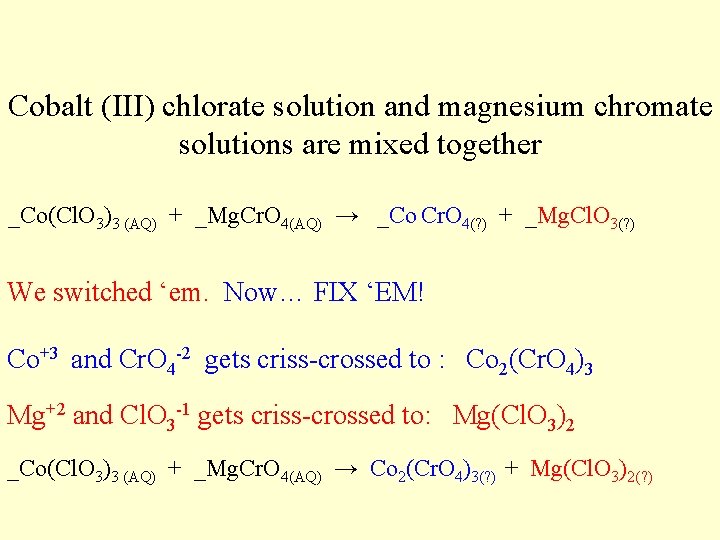
Cobalt (III) chlorate solution and magnesium chromate solutions are mixed together _Co(Cl. O 3)3 (AQ) + _Mg. Cr. O 4(AQ) → _Co Cr. O 4(? ) + _Mg. Cl. O 3(? ) We switched ‘em. Now… FIX ‘EM! Co+3 and Cr. O 4 -2 gets criss-crossed to : Co 2(Cr. O 4)3 Mg+2 and Cl. O 3 -1 gets criss-crossed to: Mg(Cl. O 3)2 _Co(Cl. O 3)3 (AQ) + _Mg. Cr. O 4(AQ) → Co 2(Cr. O 4)3(? ) + Mg(Cl. O 3)2(? )

Cobalt (III) chlorate solution and magnesium chromate solutions are mixed together 2 Co(Cl. O 3)3 (AQ) + _Mg. Cr. O 4(AQ) → _Co 2(Cr. O 4)3(? ) + _Mg(Cl. O 3)2(? ) We switched ‘em, we fixed ‘em… now balance ‘em! SAY: one Co in reactants, but two in products… Put a 2 in front of Co(Cl. O 3)3

Cobalt (III) chlorate solution and magnesium chromate solutions are mixed together 2 Co(Cl. O 3)3 (AQ) + _Mg. Cr. O 4(AQ) → _Co 2(Cr. O 4)3(? ) + 3 Mg(Cl. O 3)2(? ) SAY: 2 Co in reactants, and 2 in products… good! SIX chlorates, in reactants, two in products, Put a 3 in front of Mg(Cl. O 3)2 to get six chlorates in products, start over.

Cobalt (III) chlorate solution and magnesium chromate solutions are mixed together 2 Co(Cl. O 3)3 (AQ) + 3 Mg. Cr. O 4(AQ) → _Co 2(Cr. O 4)3(? ) + 3 Mg(Cl. O 3)2(? ) SAY: 2 Co in reactants, and 2 in products… good! SIX chlorates, in reactants, SIX in products…. good! Only 1 Mg in reactants, but 3 are in the products, so put a 3 in front of Mg. Cr. O 4 , start over again.

Cobalt (III) chlorate solution and magnesium chromate solutions are mixed together 2 Co(Cl. O 3)3 (AQ) + 3 Mg. Cr. O 4(AQ) → _Co 2(Cr. O 4)3(? ) + 3 Mg(Cl. O 3)2(? ) SAY: 2 Co in reactants, and 2 in products… good! SIX chlorates, in reactants, SIX in products…. good! 3 Mg in reactants, and 3 are in the products… good! 3 chromates in reactants, and LOOK, 3 chromates in products, balanced! This is a 2: 3: 1: 3 ratio, it can’t be reduced so - John Dalton is happy!

Cobalt (III) chlorate solution and magnesium chromate solutions are mixed together 2 Co(Cl. O 3)3 (AQ) + 3 Mg. Cr. O 4(AQ) → _Co 2(Cr. O 4)3(S) + 3 Mg(Cl. O 3)2(AQ) LAST STEP: check products on table F, which is AQ or S? ? ? If your last name is chromate (right side) you are insoluble but there are exceptions – like Mg, but not Co, so the first product is SOLID! A precipitate is the proof that a DR happened! If your last name is chlorate, you are soluable (AQ) without exception! DONE finally!

Hydrogen mono-iodide gas decomposes

SKELETON Hydrogen monoiodide gas decomposes _HI(G) → _H 2(G) + _I 2(S) Hydrogen mono-ioidide is a 1: 1 ratio, and the both products are HONCl. Br. IF twins, diatomic. The reactant is a gas, we’re told that. Hydrogen is a gas normally, and iodine is a solid at normal temperatures.

SKELETON Hydrogen monoiodide gas decomposes 2 HI(G) → _H 2(G) + _I 2(S) SAY: One H in reactant, two in products. Put a 2 in front of HI, and start over.
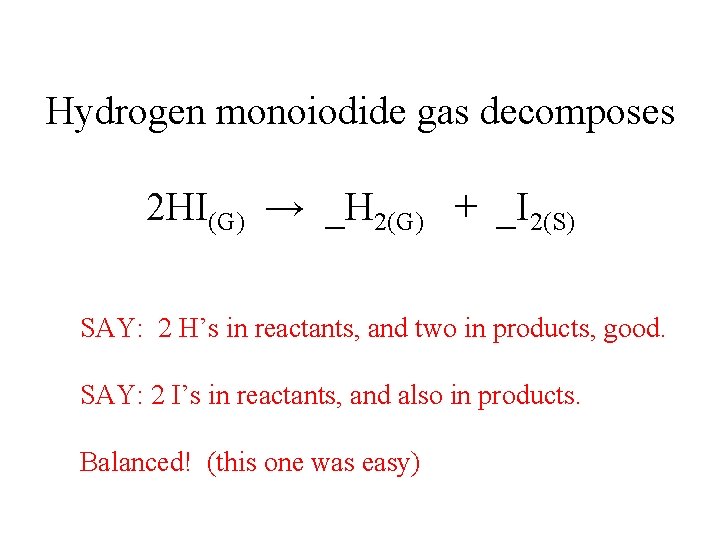
Hydrogen monoiodide gas decomposes 2 HI(G) → _H 2(G) + _I 2(S) SAY: 2 H’s in reactants, and two in products, good. SAY: 2 I’s in reactants, and also in products. Balanced! (this one was easy)

Carbon and hydrogen form ethane (C 2 H 6) gas

SKELETON Carbon and hydrogen form ethane (C 2 H 6) gas _C(S) + _H 2(G) → _C 2 H 6(G) Carbon is a solid and it is atomic, just “C” Hydrogen is a gas, and is diatomic HONCl. Br. IF Twin You’re given the formula for ethane and it’s a gas.

Carbon and hydrogen form ethane (C 2 H 6) gas 2 C(S) + _H 2(G) → _C 2 H 6(G) SAY: one C in reactant, but two in products, put a 2 in front of carbon, start over.

Carbon and hydrogen form ethane (C 2 H 6) gas 2 C(S) + 3 H 2(G) → _C 2 H 6(G) SAY: two C in reactants, and two in products, good. SAY: 2 H’s in reactants, but 6 H’s in products. How to get six H’s in front? Add a three in front of the pure hydrogen. Start over again!

Carbon and hydrogen form ethane (C 2 H 6) gas 2 C(S) + 3 H 2(G) → _C 2 H 6(G) SAY: two C in reactants, and two in products, good. SAY: 6 H’s in reactants, but 6 H’s in products. Good! Balanced, this was easier also. In a 2: 3: 1 ratio (JD says yay!

Propanol combusts (C 3 H 7 OH) Propanol is a type of alcohol, it’s an oxygenated hydrocarbon. That means it is made up of hydrogen and carbon (like a hydrocarbon) PLUS some oxygen. This is fine, but it makes balancing a bit trickier, as now there is oxygen in both reactants as well as both products, but you’re smart, this is fun.

SKELETON Propanol combusts (C 3 H 7 OH) _C 3 H 7 OH(G) + _O 2(G) → _CO 2(G) + _H 2 O(G) In combustion, a hydrocarbon, or OXYGENTATED HYDROCARBON combines rapidly with oxygen and forms only carbon dioxide and water. Pure oxygen is diatomic, and the product formulas are well known already.
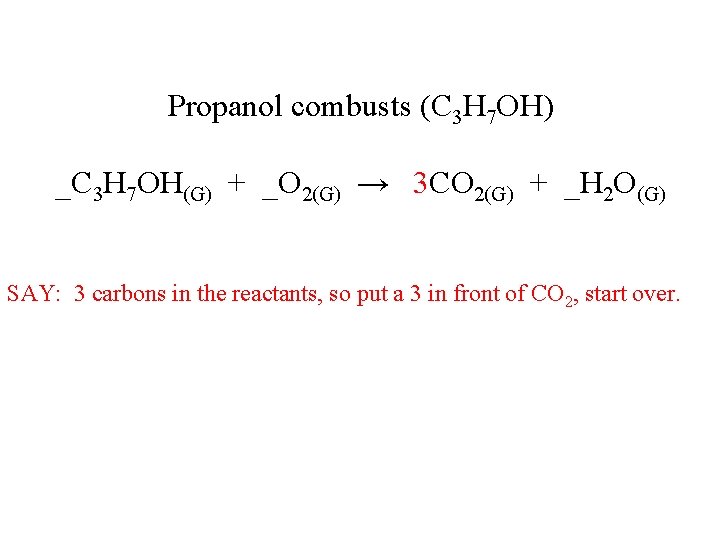
Propanol combusts (C 3 H 7 OH) _C 3 H 7 OH(G) + _O 2(G) → 3 CO 2(G) + _H 2 O(G) SAY: 3 carbons in the reactants, so put a 3 in front of CO 2, start over.
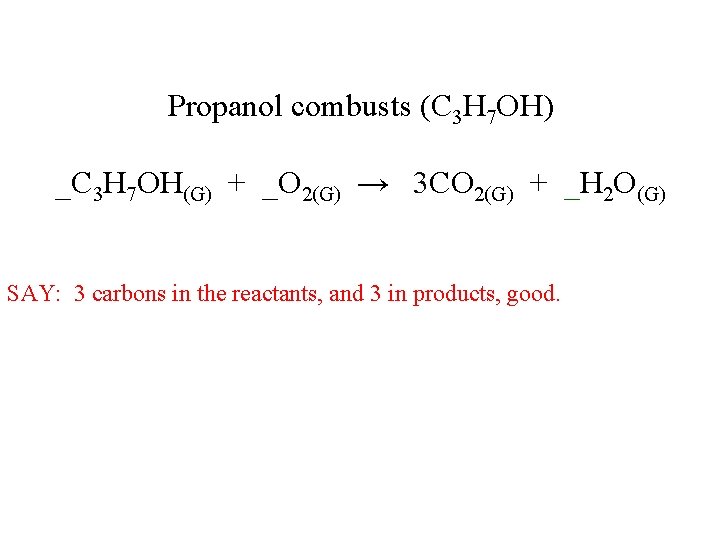
Propanol combusts (C 3 H 7 OH) _C 3 H 7 OH(G) + _O 2(G) → 3 CO 2(G) + _H 2 O(G) SAY: 3 carbons in the reactants, and 3 in products, good.

Propanol combusts (C 3 H 7 OH) _C 3 H 7 OH(G) + _O 2(G) → 3 CO 2(G) + 4 H 2 O(G) SAY: 3 carbons in the reactants, and 3 in products, good. SAY: 7 + 1 H = 8 H in reactants, we need to put a 4 in front of the water. Start over.

Propanol combusts (C 3 H 7 OH) _C 3 H 7 OH(G) + _O 2(G) → 3 CO 2(G) + 4 H 2 O(G) SAY: 3 carbons in the reactants, and 3 in products, good. SAY: 7 + 1 H = 8 H in reactants, and 8 H in products, good. SAY: two oxygens in reactants, but 6 + 4 = 10 oxygen in products (NOT GOOD). To fix this, think… we need even oxygen on reactant side, we need to double the propanol to do that, then start over.
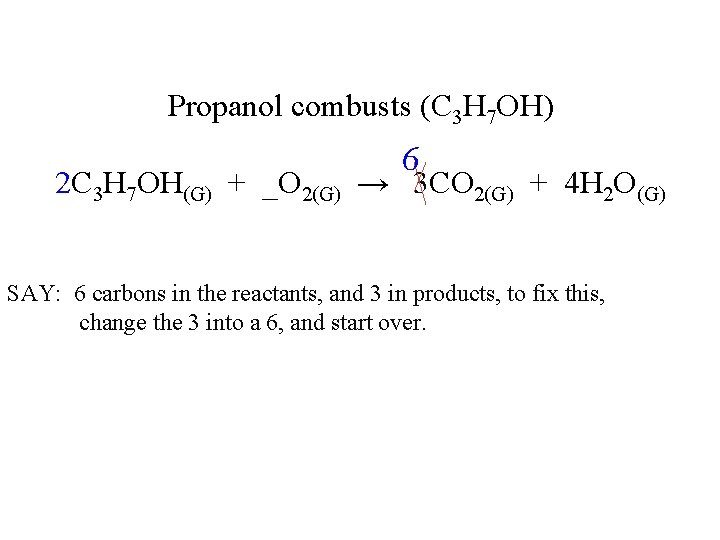
Propanol combusts (C 3 H 7 OH) 6 2 C 3 H 7 OH(G) + _O 2(G) → 3 CO 2(G) + 4 H 2 O(G) SAY: 6 carbons in the reactants, and 3 in products, to fix this, change the 3 into a 6, and start over.

Propanol combusts (C 3 H 7 OH) 8 2 C 3 H 7 OH(G) + _O 2(G) → 6 CO 2(G) + 4 H 2 O(G) SAY: 6 carbons in the reactants, and 3 in products, good. SAY 2 x (7+1) H’s in reactants = 16 H, and only 8 in products, to fix this change the four into an 8 and start over.

Propanol combusts (C 3 H 7 OH) 2 C 3 H 7 OH(G) + _O 2(G) → 6 CO 2(G) + 8 H 2 O(G) SAY: 6 carbons in the reactants, and 3 in products, good. SAY 2 x (7+1) H’s in reactants = 16 H, and now 16 H’s in products too. We have 2+2 oxygens in reactants, but 12 + 8 = 20 in products. To fix this we do not want to change propanol again, it wrecks everything. How can we get 20 oxygens but keep just 2 propanol? Make the pure oxygen 18 oxygens, but adding a 9!

Propanol combusts (C 3 H 7 OH) 2 C 3 H 7 OH(G) + 9 O 2(G) → 6 CO 2(G) + 8 H 2 O(G) SAY: 6 carbons in the reactants, and 3 in products, good. SAY 2 x (7+1) H’s in reactants = 16 H, and now 16 H’s in products too. SAY: we have 2 + 18 Oxygens = 20 in reactants, and we have 12 + 8 oxygens = 20 oxygens in the products, balanced!

Bromine is poured into lithium iodide solution

SKELETON Bromine is poured into lithium iodide solution _Br 2(L) + _Li. I(AQ) → _Li. Br(AQ) + _I 2(S) Bromine is diatomic HONCl. Br. IF twin. Li makes the Li+1 cation. Iodine makes only the I-1 anion, 1: 1 ratio. Li makes the Li+1 cation. Bromine makes only the Br-1 anion, 1: 1 ratio. Iodine is diatomic HONCl. Br. IF twin.

Bromine is poured into lithium iodide solution _Br 2(L) + _Li. I(AQ) → 2 Li. Br(AQ) + _I 2(S) SAY: 2 bromines in reactants, so put a 2 in front of Li. Br, start over.

Bromine is poured into lithium iodide solution _Br 2(L) + 2 Li. I(AQ) → 2 Li. Br(AQ) + _I 2(S) SAY: 2 bromines in reactants, 2 in products, good. SAY: 1 lithium in reactant, but two in products, put a 2 in front of the Li. I and start over again.

Bromine is poured into lithium iodide solution _Br 2(L) + 2 Li. I(AQ) → 2 Li. Br(AQ) + _I 2(S) SAY: 2 bromines in reactants, 2 in products, good. SAY: 2 lithiums in reactant, but two in products, good. SAY: 2 iodines in reactants, and we have 2 already in products (great!) This is already balanced! It’s a 1: 2: 2: 1 ratio, me and JD say yay.

Barium nitrate solution is mixed with sodium sulfate solution Here we have two AQ solutions to start, the perfect set up for a double replacement reaction. We will write the reactants, then we will write the products.

SKELETON Barium nitrate solution is mixed with sodium sulfate solution _Ba(NO 3)2(AQ) + _Na 2 SO 4(AQ) → Ba+2 and NO 3 -1 get criss-crossed to… Ba(NO 3)2 Na+1 and SO 4 -2 gets criss-crossed to… Na 2 SO 4 Both are aqueous because they are solutions, but you could check table F to be sure… If your last name is nitrate, you’re always aqueous. If your first name is

Barium nitrate solution is mixed with sodium sulfate solution _Ba(NO 3)2(AQ) + _Na 2 SO 4(AQ) → _Na. NO 3(? ) + _Ba. SO 4(? ) Switch up the anions, to get barium with sulfate, and sodium with the nitrate. Just switch them now, we’ll fix them in a minute. As it turns out, the Na. NO 3 is in a 1: 1 ratio +1 cation with the -1 anion. The Ba. SO 4 is also a 1: 1 ratio, a +2 cation and a -2 anion.

Barium nitrate solution is mixed with sodium sulfate solution _Ba(NO 3)2(AQ) + _Na 2 SO 4(AQ) → 2 Na. NO 3(? ) + _Ba. SO 4(? ) SAY: one Ba in reactants, and one in products, good. SAY: two nitrates in reactants, put a 2 in front of the sodium nitrate to balance the nitrates, start over.

Barium nitrate solution is mixed with sodium sulfate solution _Ba(NO 3)2(AQ) + _Na 2 SO 4(AQ) → 2 Na. NO 3(? ) + _Ba. SO 4(? ) SAY: one Ba in reactants, and one in products, good. SAY: 2 nitrates in reactants, and 2 in products, good. SAY: 2 sodiums in reactants, and 2 in products, good. SAY: one sulfate in reactants and 1 in products, yay! Balanced with a 1: 1: 2: 1 ratio. Me and J. D. are okay now.

Barium nitrate solution is mixed with sodium sulfate solution _Ba(NO 3)2(AQ) + _Na 2 SO 4(AQ) → 2 Na. NO 3(AQ) + _Ba. SO 4(S) Both reactants are AQ, check the products… If your first name is sodium, you are AQ. If your last name is nitrate, you are AQ too. If your last name is sulfate, you are AQ (with exceptions, and Ba+2 is an exception, so this product is S, solid! (precipitate, and proof that a DR happened). DONE
- Slides: 64