Wood Chemistry PSE 406 Lecture 11 Lignin Structure













- Slides: 22

Wood Chemistry PSE 406 Lecture 11 Lignin Structure PSE 406 Lecture 11 1

Class Agenda l Functional groups » Methoxyl, phenolic hydroxyl, aliphatic hydroxyl, carbonyl l l Lignin structures Lignin – carbohydrate complexes Lignin analytical procedures Lignin trivial facts Appendix PSE 406 Lecture 11 2

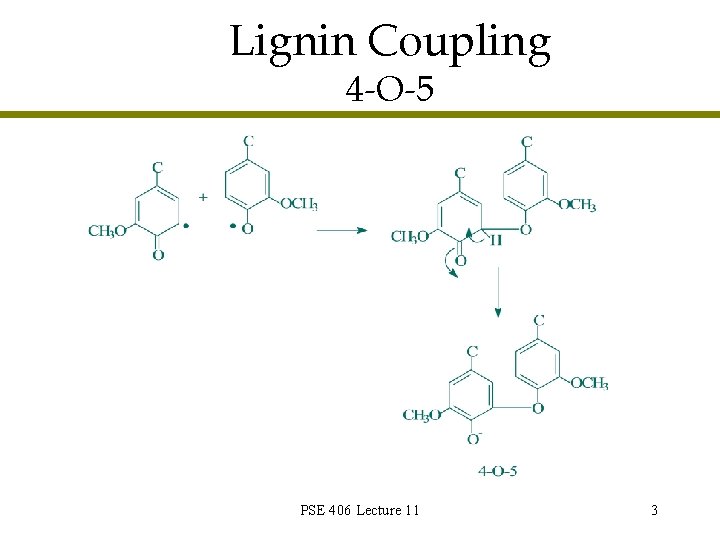
Lignin Coupling 4 -O-5 PSE 406 Lecture 11 3

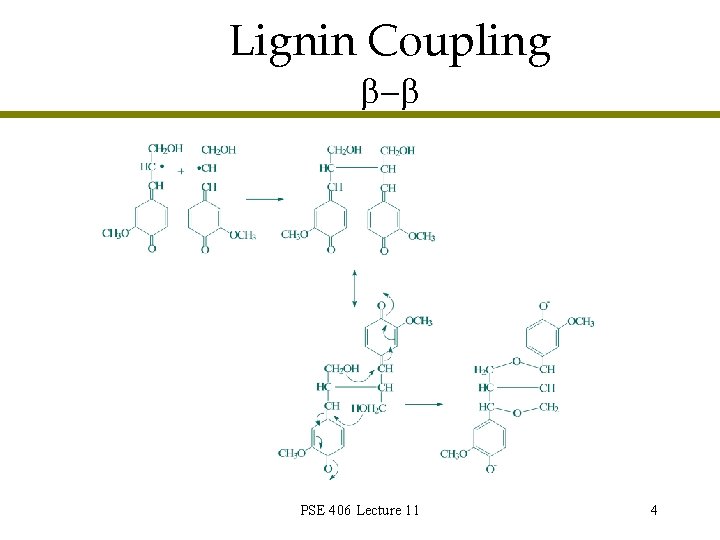
Lignin Coupling - PSE 406 Lecture 11 4

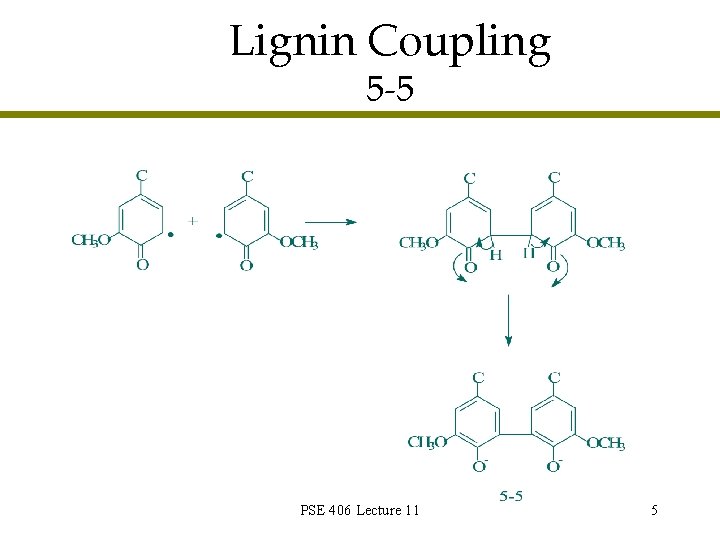
Lignin Coupling 5 -5 PSE 406 Lecture 11 5

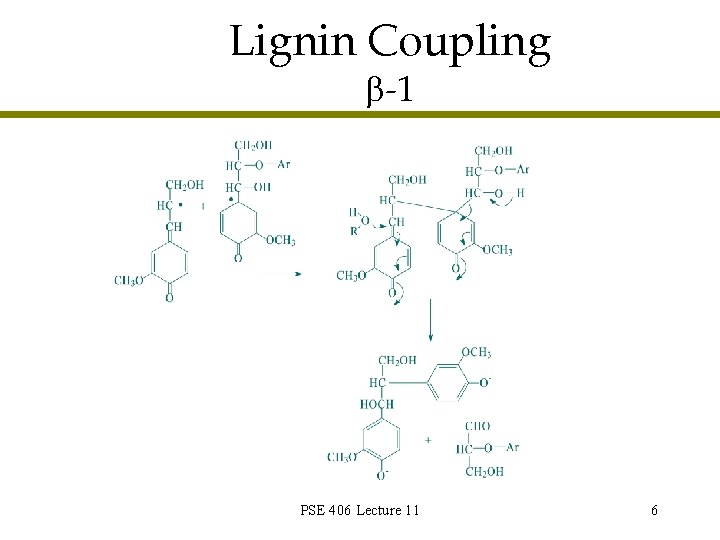
Lignin Coupling -1 PSE 406 Lecture 11 6

Lignin Coupling -5 PSE 406 Lecture 11 7

Lignin Functional Groups Phenolic Hydroxyl l 15 -30 free phenolic hydroxyl/100 C 9: Softwood 10 -15 free phenolic hydroxyl/100 C 9: Hardwood Reactivity » Units containing free phenolic hydroxyl groups much more susceptible to cleavage reactions hydrolysis of a and aryl ether linkages » Structures much more reactive towards modification reactions PSE 406 Lecture 11 8

Lignin Functional Groups Methoxyl l l ~0. 95/C 9 in softwoods ~1. 5/C 9 in hardwoods Generally resistant to acid and alkali H 2 S cleaves to form thiols, mercaptans (Kraft mill odor) PSE 406 Lecture 11 9

Lignin Functional Groups Aliphatic Hydroxyl l Majority of aliphatic hydroxyl groups are primary: on g carbon » Site relatively non-reactive » In some species, g carbon oxygen linked through ester linkage to r-coumaric acid, etc l Benzyl alcohols » Debated amount: 16 -40/100 C 9 in spruce » Play dominant role in delignification reactions PSE 406 Lecture 11 10

Lignin Functional Groups Carbonyl Groups l Total carbonyl groups 20/100 C 9 in spruce » 1/2 Conjugated Structures – Coniferaldehyde and a-keto structures – Play important role in delignifcation reactions » 1/2 Non-conjugated Structures – Glyceraldehyde from -1 coupling l Larger amount in certain hardwoods and grasses due to esters. PSE 406 Lecture 11 11

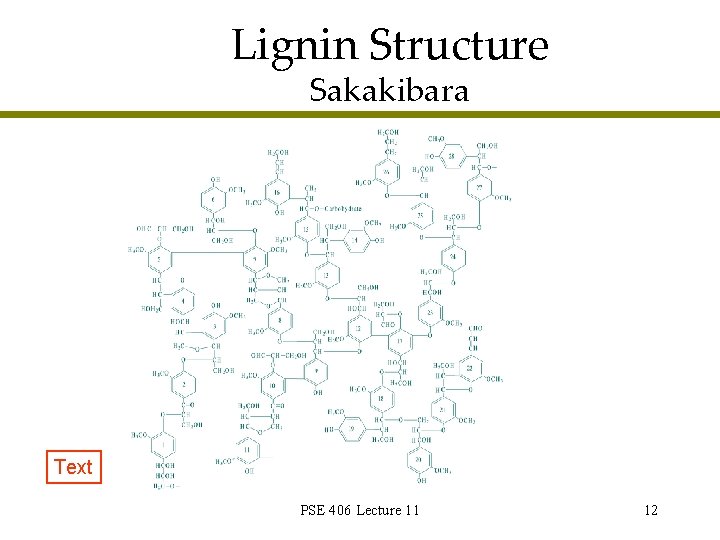
Lignin Structure Sakakibara Text PSE 406 Lecture 11 12

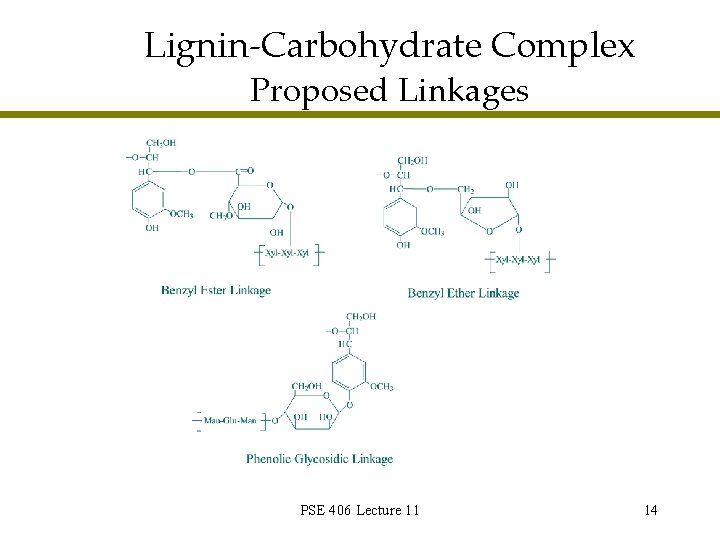
Lignin-Carbohydrate Complex l l All purified holocellulose materials contain a certain amount of lignin All purified lignin fractions contain a certain amount of monosaccharides LCCs have been enzymatically prepared from lignin and monosaccharide model compounds Significant work studying isolated LCCs » No definitive information on exact covalent bonding patterns » Generally accepted bonding patterns PSE 406 Lecture 11 13

Lignin-Carbohydrate Complex Proposed Linkages PSE 406 Lecture 11 14

Lignin-Carbohydrate Complex General Information l l Mw of isolated LCCs 600® 15, 000 LCC linkage stability » Esters: alkali labile, acid labile » Ethers: selectively alkali labile, mildly acid labile » Glycosides: mildly alkali labile, acid labile l l Formation during pulping processes possible LCCs: residual lignin and bleaching » Removal of that last little bit of lignin » Enzyme assisted bleaching PSE 406 Lecture 11 15

Lignin Structure Analytical Procedures l l All analysis require model compound studies! Linkages » Enzymatic dehydrogenation (test tube studies) » Degradation studies (see appendix) » NMR l Functional groups » Wet Chemistry techniques » Spectroscopy PSE 406 Lecture 11 16

Lignin Trivial Facts I l Solubility » Native lignins: limited/no solubility in all solvents without modification l Molecular Weight » Average Mw for softwood ~20, 000, lower for hardwoods » Polydispersity ~ 2. 5 -3. 0 » Mw measured for lignosulfonates as high as 1, 000 PSE 406 Lecture 11 17

Lignin Trivial Facts II l Elemental Composition MWL » Spruce C 9 H 7. 92 O 2. 40(OCH 3)0. 92 C 9 H 8. 83 O 2. 37(OCH 3)0. 96 » Beech C 9 H 8. 50 O 2. 86(OCH 3)1. 43 C 9 H 7. 93 O 2. 95(OCH 3)1. 46 l UV Absorption » Strong adsorption at 205 and 280 nm » Carbohydrates do not adsorb at 280 nm l Compression Wood (Softwoods) » High % lignin (~40%), high % of r-hydroxy units (to 70%) l Tension Wood (Hardwoods) » Reduced lignin content PSE 406 Lecture 11 18

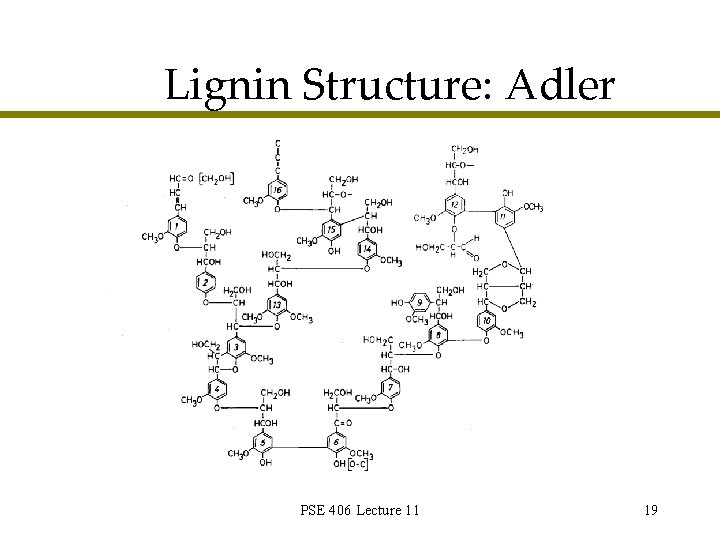
Lignin Structure: Adler PSE 406 Lecture 11 19

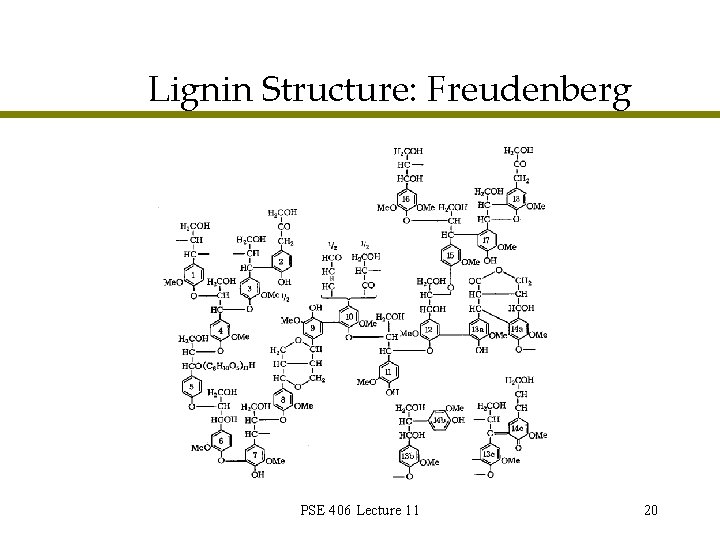
Lignin Structure: Freudenberg PSE 406 Lecture 11 20

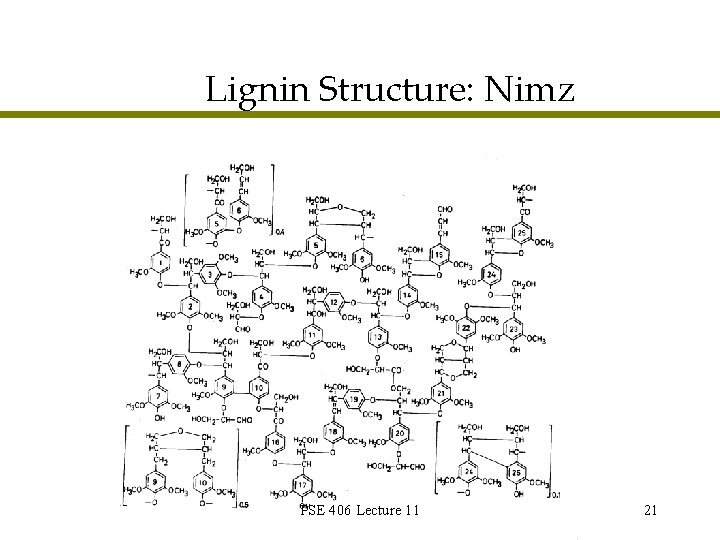
Lignin Structure: Nimz PSE 406 Lecture 11 21

Lignin Structure: Forss PSE 406 Lecture 11 22