William H Brown Christopher S Foote Brent L

William H. Brown Christopher S. Foote Brent L. Iverson Eric Anslyn http: //academic. cengage. com/chemistry/brown Chapter 12 Infrared Spectroscopy William H. Brown • Beloit College 12 -1

Molecular Spectroscopy u Molecular spectroscopy The study of which frequencies of electromagnetic radiation are absorbed or emitted by a particular substance and the correlation of these frequencies with details of molecular structure. • we study three types of molecular spectroscopy 12 -2
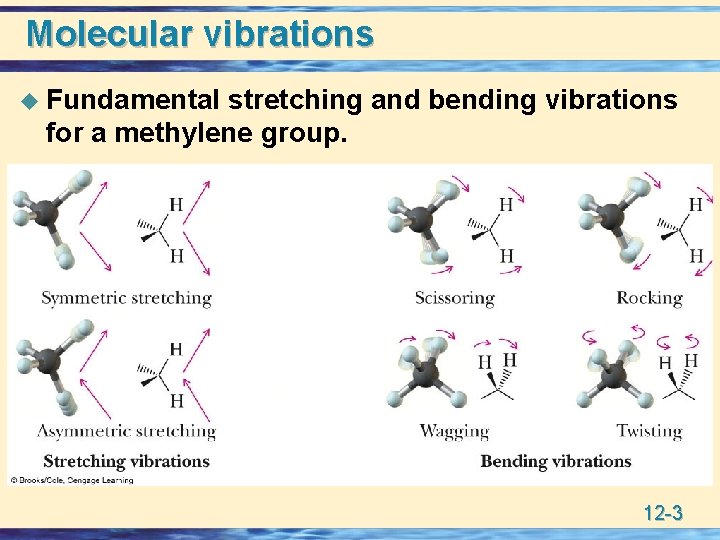
Molecular vibrations u Fundamental stretching and bending vibrations for a methylene group. 12 -3

Molecular Vibrations u For a molecule to absorb IR radiation • the bond undergoing vibration must be polar and its vibration must cause a periodic change in the bond dipole moment. u Covalent bonds which do not meet these criteria are said to be IR inactive. • The C-C double and triple bonds of symmetrically substituted alkenes and alkynes, for example, are IR inactive because they are not polar bonds. 12 -4
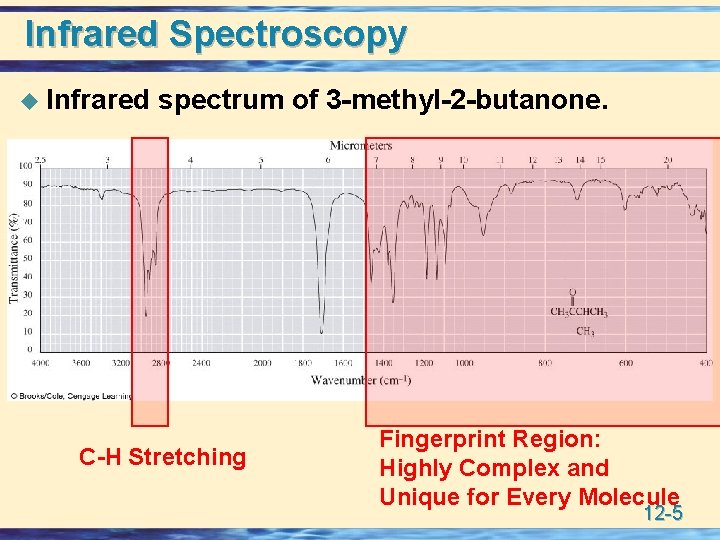
Infrared Spectroscopy u Infrared spectrum of 3 -methyl-2 -butanone. C-H Stretching Fingerprint Region: Highly Complex and Unique for Every Molecule 12 -5

Correlation Tables u Infrared stretching frequencies of selected functional groups. Less Valuable Almost all organic molecules have C-H bonds C=C often too weak or encroaching on fingerprint region Buried in fingerprint region More Valuable Potentially difficult to distinguish between oneanother, but good indication of heteroatomhydrogen bond Signature Stretch. Very strong, very identifiable 12 -6
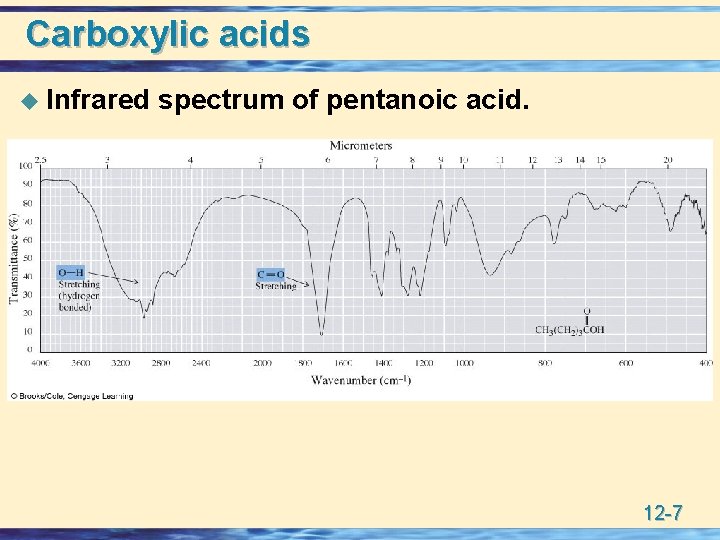
Carboxylic acids u Infrared spectrum of pentanoic acid. 12 -7
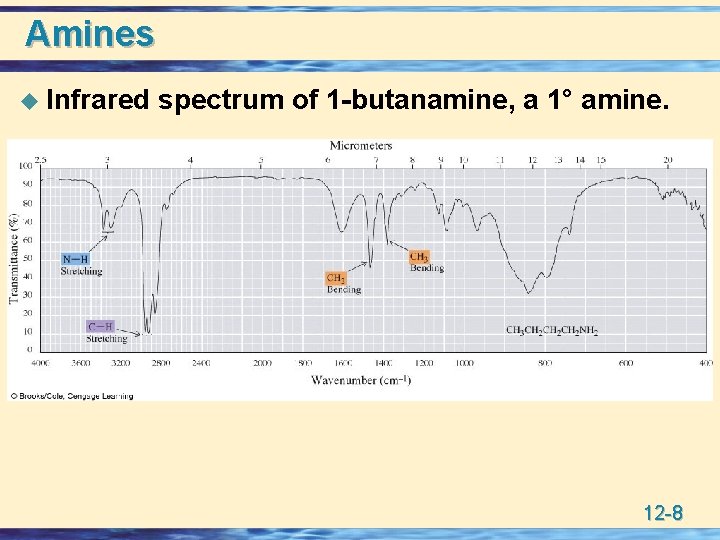
Amines u Infrared spectrum of 1 -butanamine, a 1° amine. 12 -8
Hydrocarbons-Table 12. 5 Difficult to distinguish between oneanother 12 -9
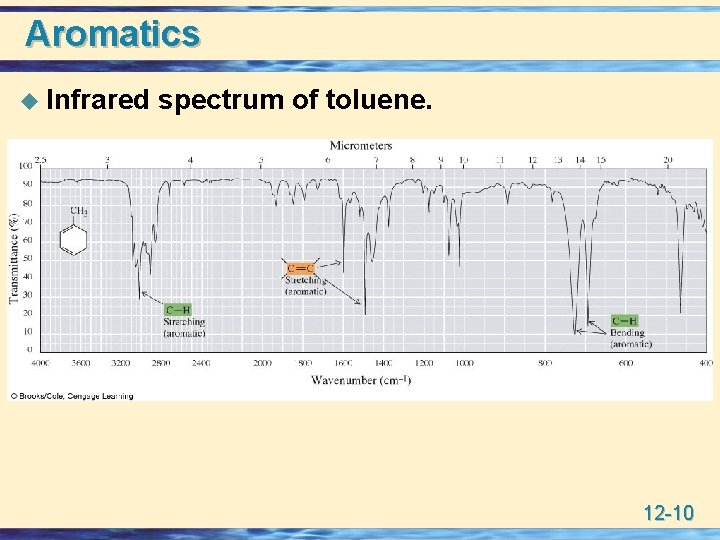
Aromatics u Infrared spectrum of toluene. 12 -10

Hydrocarbons-Table 12. 5 Can be useful. In fairly distinct regions 12 -11
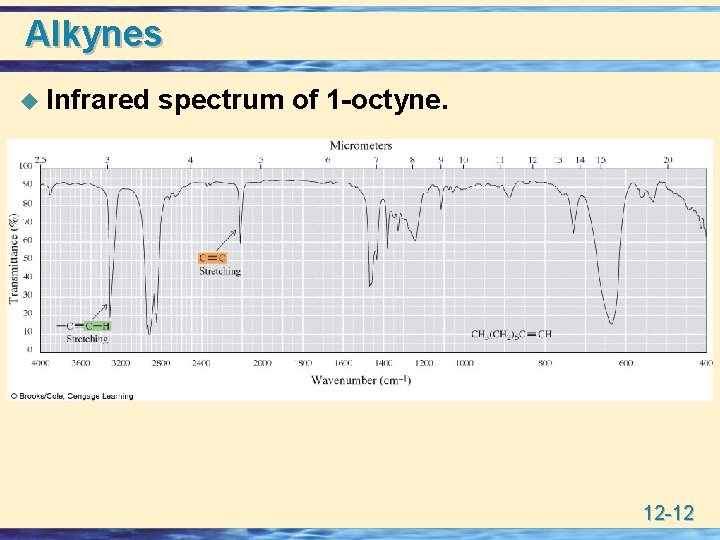
Alkynes u Infrared spectrum of 1 -octyne. 12 -12
- Slides: 12