WI 04 OSU ISMS 2013 LOWERING OF KETOENOL



- Slides: 23

WI 04, OSU ISMS 2013 LOWERING OF KETO-ENOL TAUTOMERIZATION BARRIER OF CYCLIC DIKETONES VIA CH···O INTERACTION -Tapas Chakraborty Indian Association for the Cultivation of Science, Calcutta, India

CH···O hydrogen bond LOWERING OF KETO-ENOL TAUTOMERIZATION BARRIER OF CYCLIC DIKETONES VIA CH···O INTERACTION • CH···O interactions are categorized as weak hydrogen bonds • Bond energies< 4 kcal/mol (OH···O energy ~ 5 -15 kcal/mol) • Important for stability of the structures of biological macromolecules and crystal structures of organic molecules • Direct role in chemical reactions are rarely cited

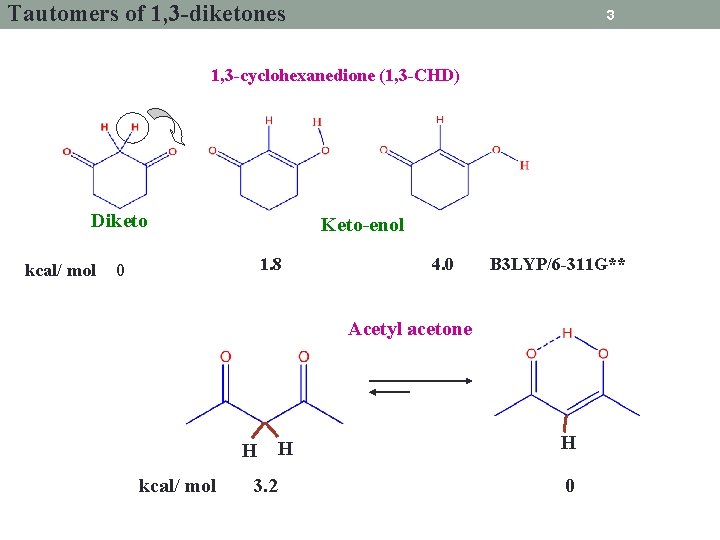
Tautomers of 1, 3 -diketones 3 1, 3 -cyclohexanedione (1, 3 -CHD) Diketo kcal/ mol Keto-enol 1. 8 0 4. 0 B 3 LYP/6 -311 G** Acetyl acetone H kcal/ mol H 3. 2 H 0

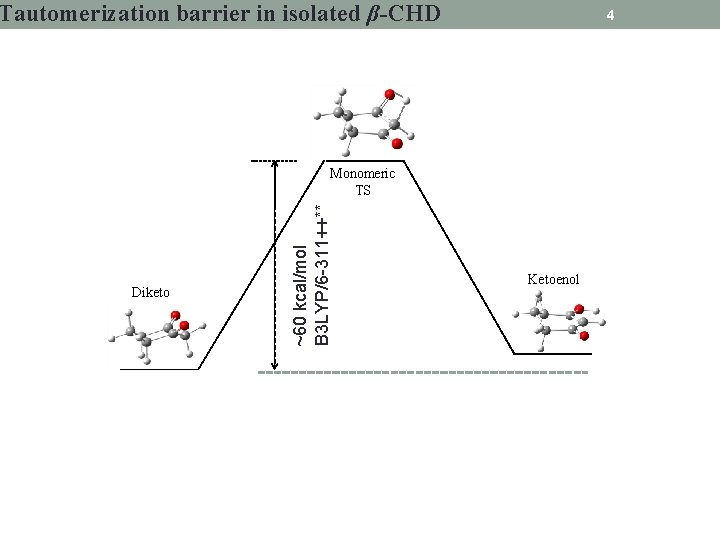
Tautomerization barrier in isolated β-CHD 4 Diketo ~60 kcal/mol B 3 LYP/6 -311++** Monomeric TS Ketoenol

Matrix-isolated 1, 3 -CHD 5

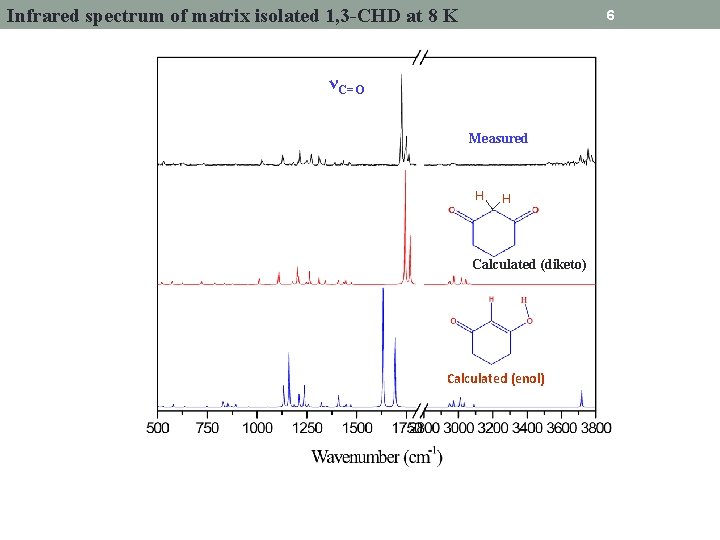
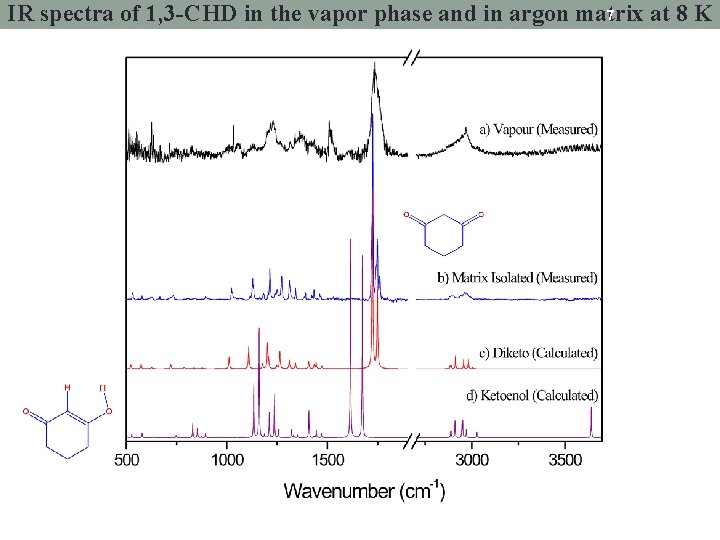
Infrared spectrum of matrix isolated 1, 3 -CHD at 8 K 6 νC=O Measured H H Calculated (diketo) Calculated (enol)

7 IR spectra of 1, 3 -CHD in the vapor phase and in argon matrix at 8 K

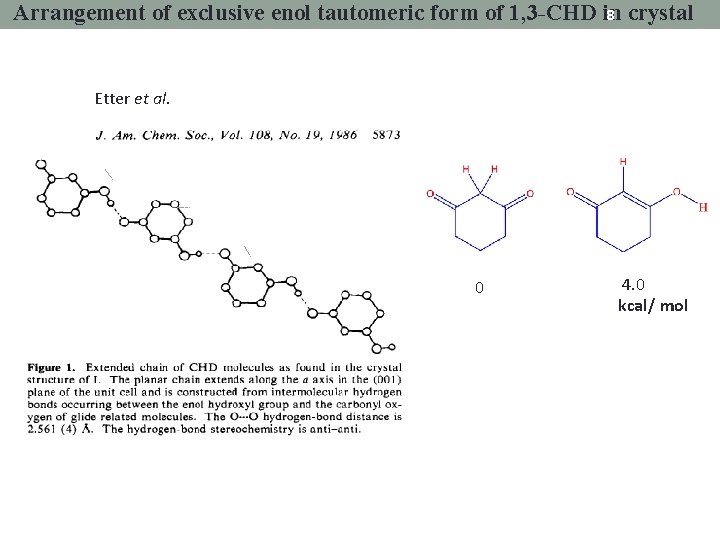
8 crystal Arrangement of exclusive enol tautomeric form of 1, 3 -CHD in Etter et al. 0 4. 0 kcal/ mol

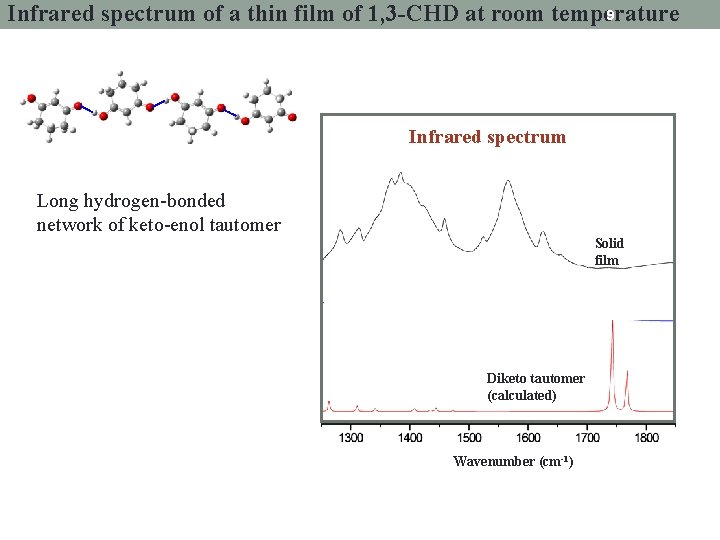
9 Infrared spectrum of a thin film of 1, 3 -CHD at room temperature Infrared spectrum Long hydrogen-bonded network of keto-enol tautomer Solid film CHCl 3 solution Diketo tautomer (calculated) Wavenumber (cm-1)

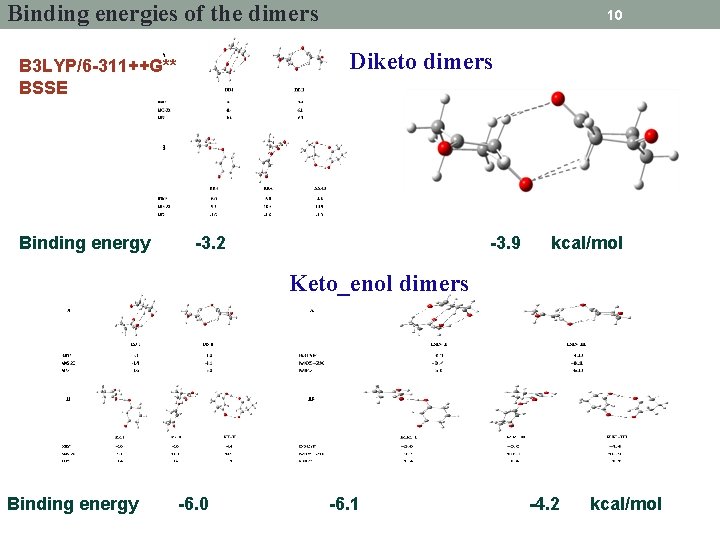
Binding energies of the dimers Diketo dimers B 3 LYP/6 -311++G** BSSE Binding energy 10 -3. 2 -3. 9 kcal/mol Keto_enol dimers Binding energy -6. 0 -6. 1 -4. 2 kcal/mol

Dimer is synthesized in the matrix by controlled annealing 11 at 15 -30 K

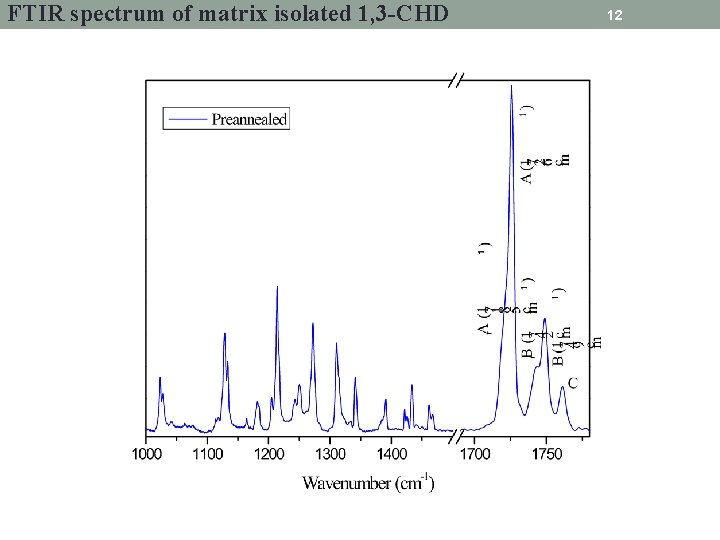
FTIR spectrum of matrix isolated 1, 3 -CHD 12

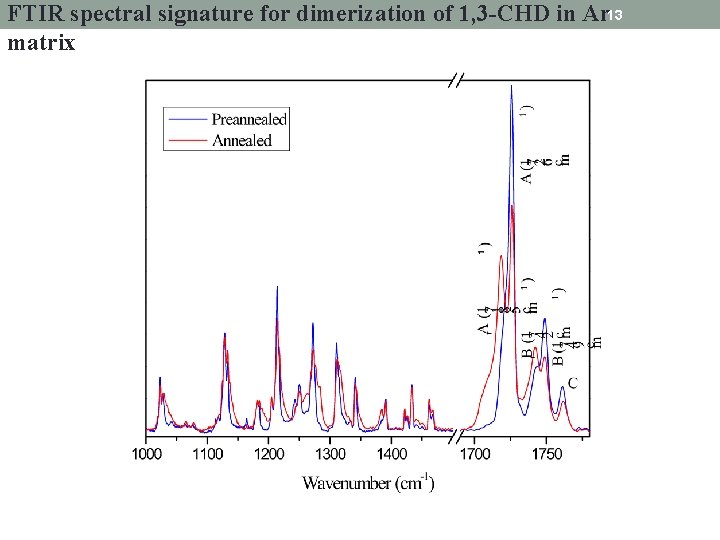
FTIR spectral signature for dimerization of 1, 3 -CHD in Ar 13 matrix

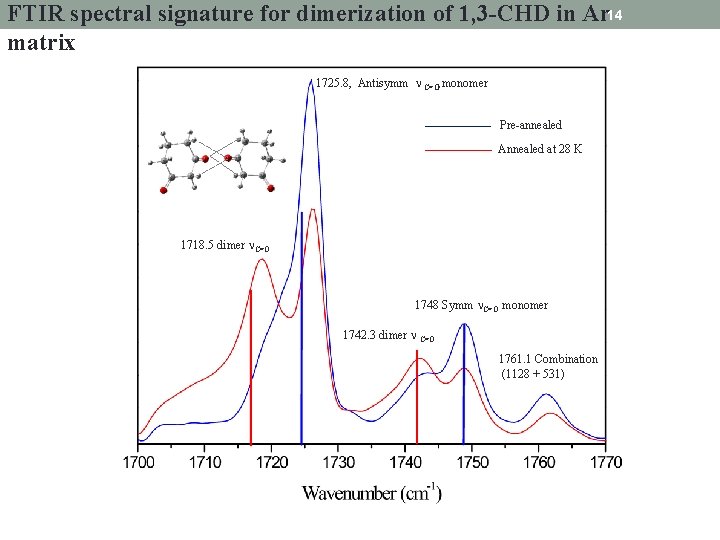
FTIR spectral signature for dimerization of 1, 3 -CHD in Ar 14 matrix 1725. 8, Antisymm C=O monomer Pre-annealed Annealed at 28 K 1718. 5 dimer C=O 1748 Symm C=O monomer 1742. 3 dimer C=O 1761. 1 Combination (1128 + 531)

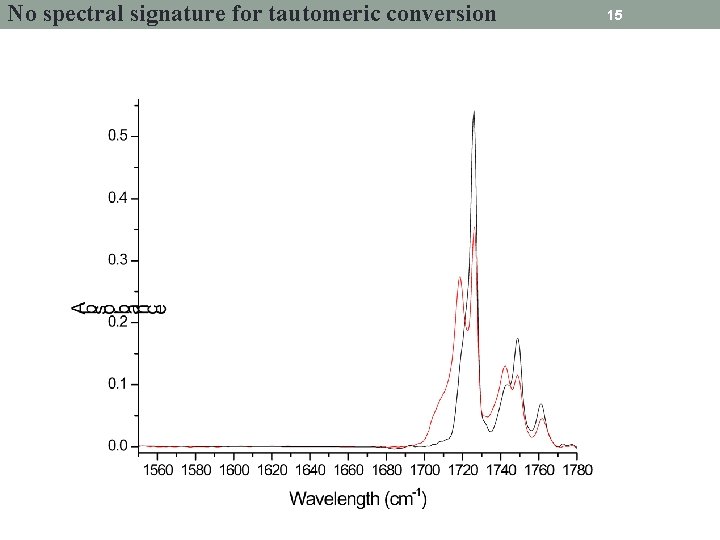
No spectral signature for tautomeric conversion 15

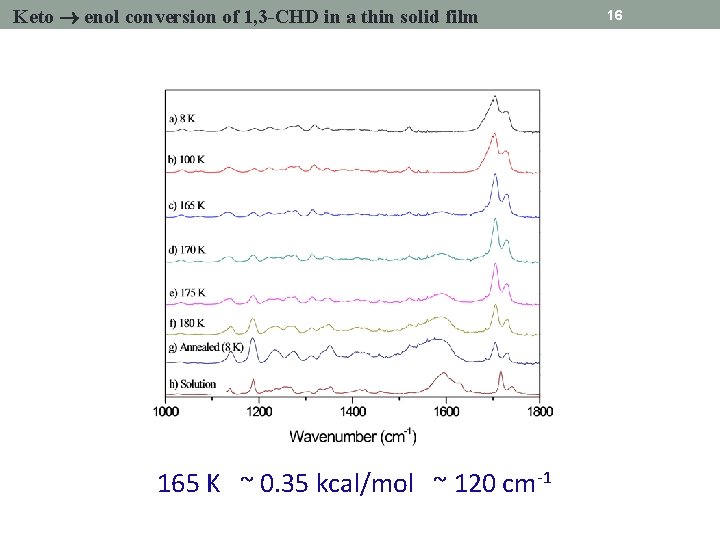
Keto enol conversion of 1, 3 -CHD in a thin solid film 165 K ~ 0. 35 kcal/mol ~ 120 cm-1 16

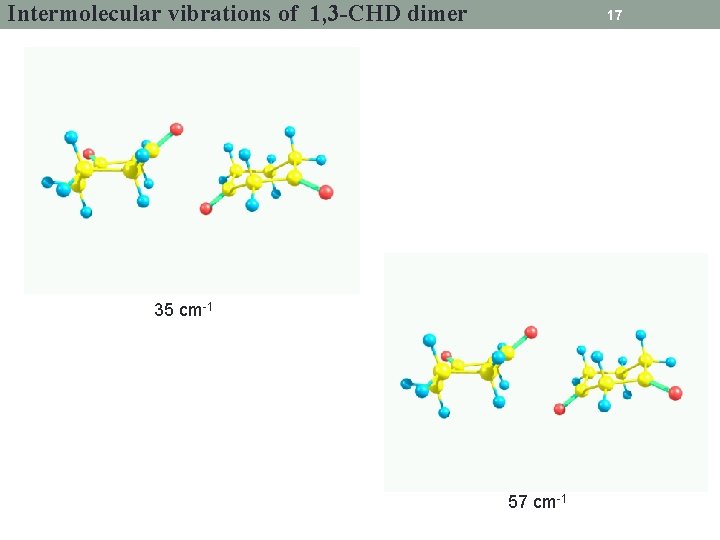
Intermolecular vibrations of 1, 3 -CHD dimer 17 35 cm-1 57 cm-1

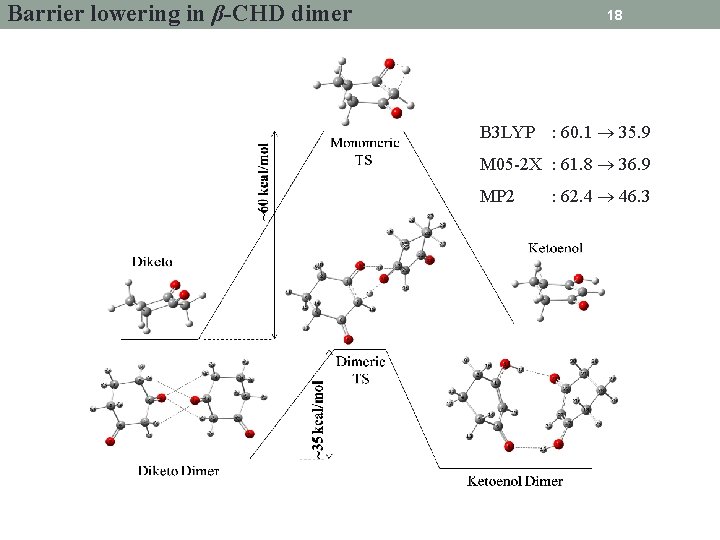
Barrier lowering in β-CHD dimer 18 B 3 LYP : 60. 1 35. 9 M 05 -2 X : 61. 8 36. 9 MP 2 : 62. 4 46. 3

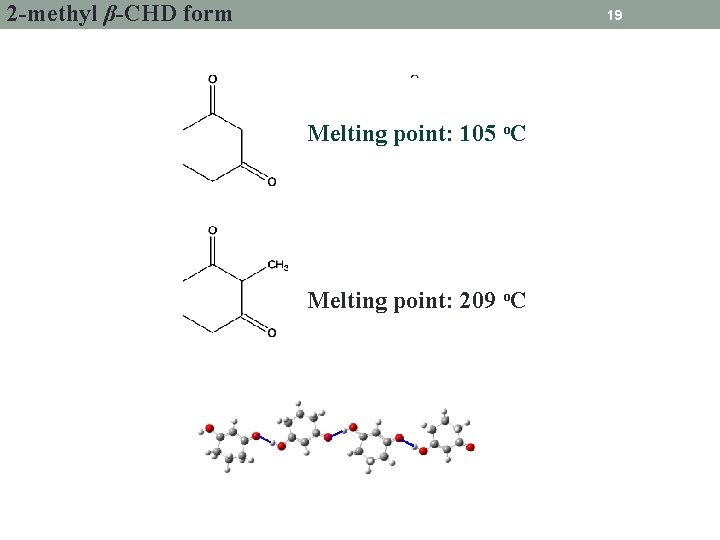
2 -methyl β-CHD form 19 Melting point: 105 o. C Melting point: 209 o. C

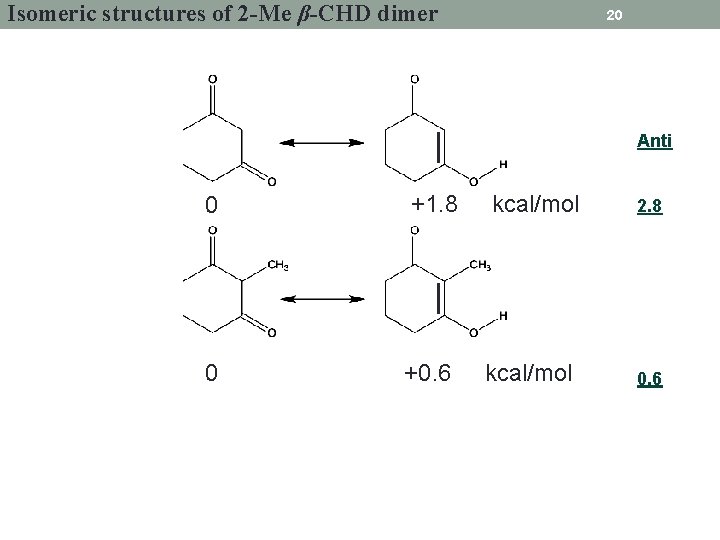
Isomeric structures of 2 -Me β-CHD dimer 20 Anti 0 +1. 8 kcal/mol 0 +0. 6 kcal/mol 2. 8 0. 6

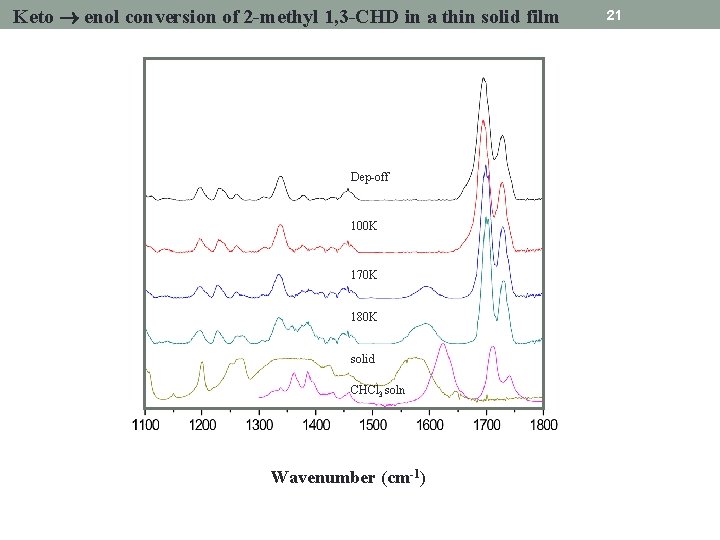
Keto enol conversion of 2 -methyl 1, 3 -CHD in a thin solid film Dep-off 100 K 170 K 180 K solid CHCl 3 soln Wavenumber (cm-1) 21

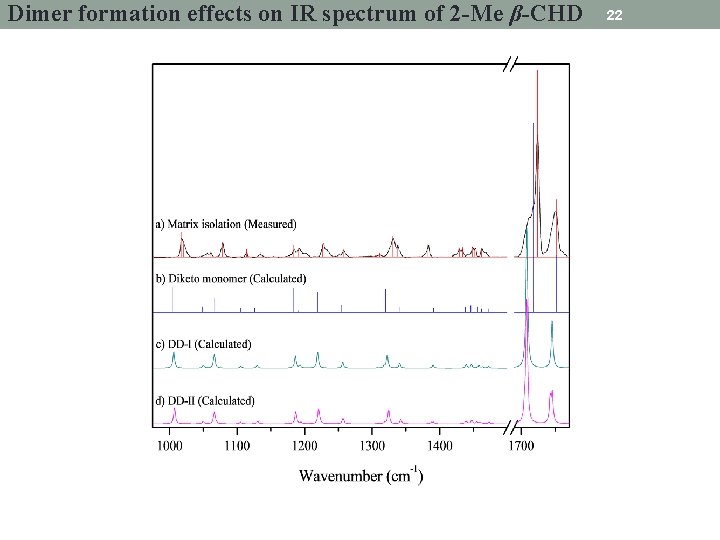
Dimer formation effects on IR spectrum of 2 -Me β-CHD 22

Acknowledgement: 23 Co-workers Dr. Biman Bandyopadhyay Ms Pujarini Banerjee Department of Science and Technology, Govt. of India