Why study Organic Synthesis o Heart of Chemistry










- Slides: 25

Why study Organic Synthesis? o Heart of Chemistry “Chemists make new things and we study reactions. That’s the core of this profession” - D. G. Nocera (MIT) in C&EN (Jan. 1998) OCFC-Intro-Chap 2 o Broad applications • • • • Petrochemicals Pharmaceuticals Agrochemicals (Bio)Polymers Dyes & Pigments Cosmetics Food (Additives) Household products Electrochemicals Display materials Semiconductors Organic electronics and so on…. . 1

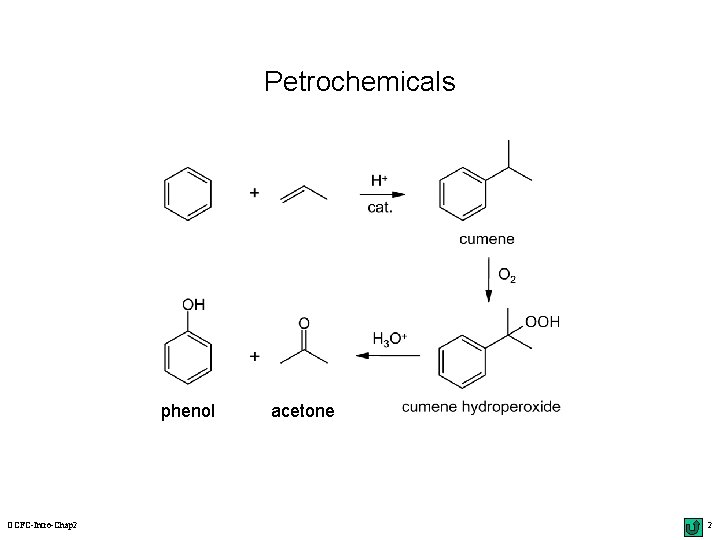
Petrochemicals phenol OCFC-Intro-Chap 2 acetone 2

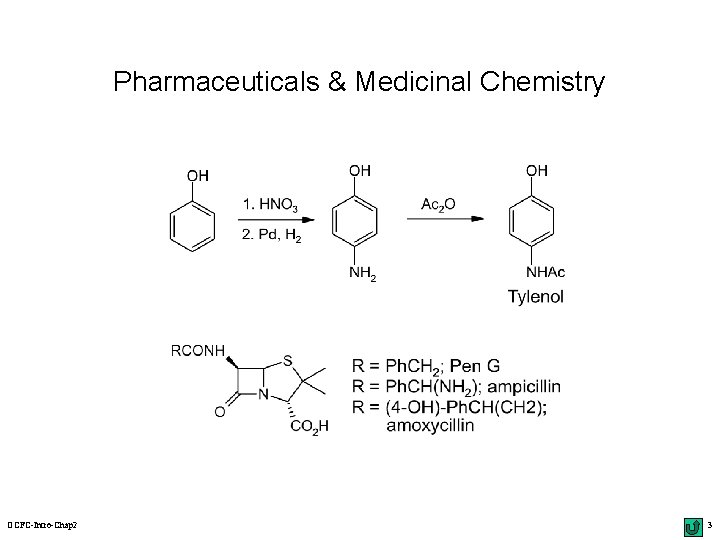
Pharmaceuticals & Medicinal Chemistry OCFC-Intro-Chap 2 3

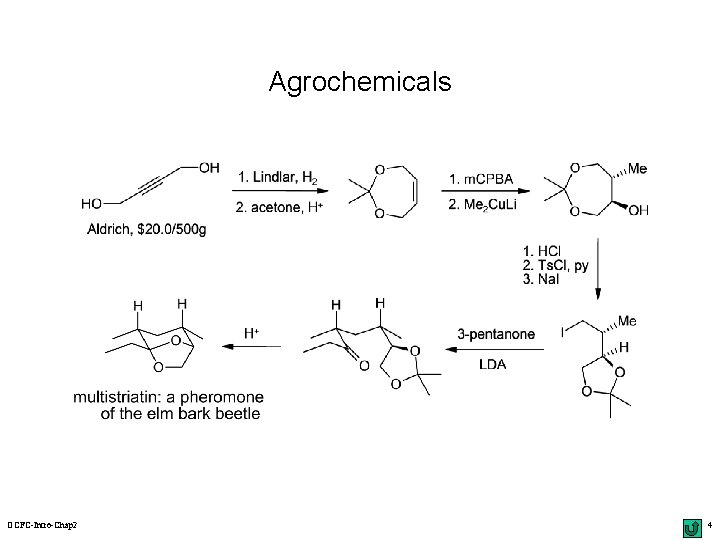
Agrochemicals OCFC-Intro-Chap 2 4

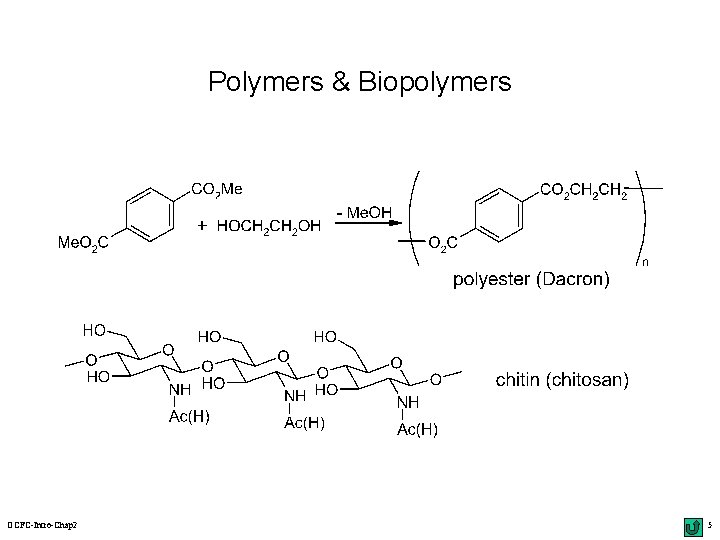
Polymers & Biopolymers OCFC-Intro-Chap 2 5

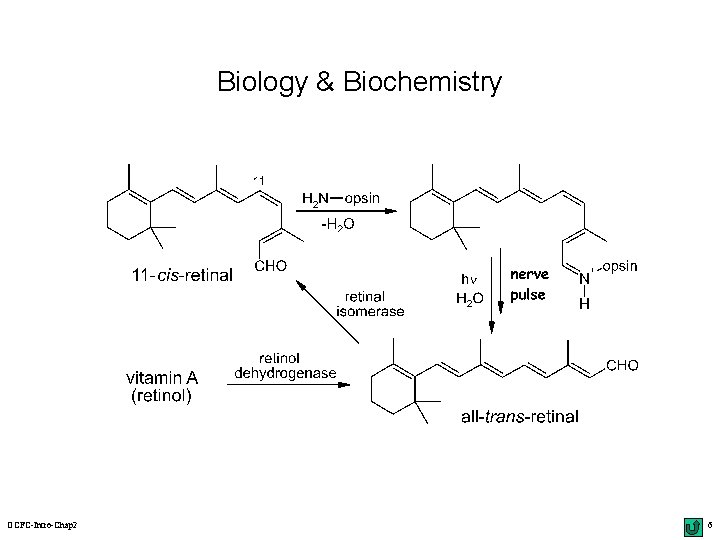
Biology & Biochemistry OCFC-Intro-Chap 2 6

Electrochemicals OCFC-Intro-Chap 2 7

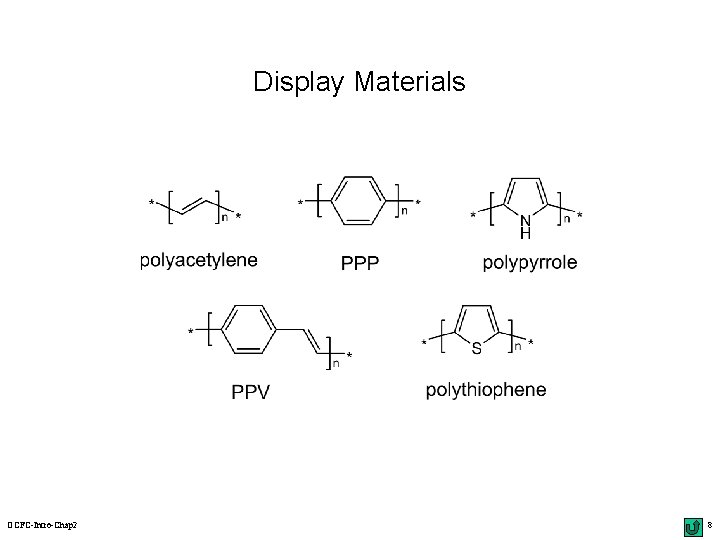
Display Materials OCFC-Intro-Chap 2 8

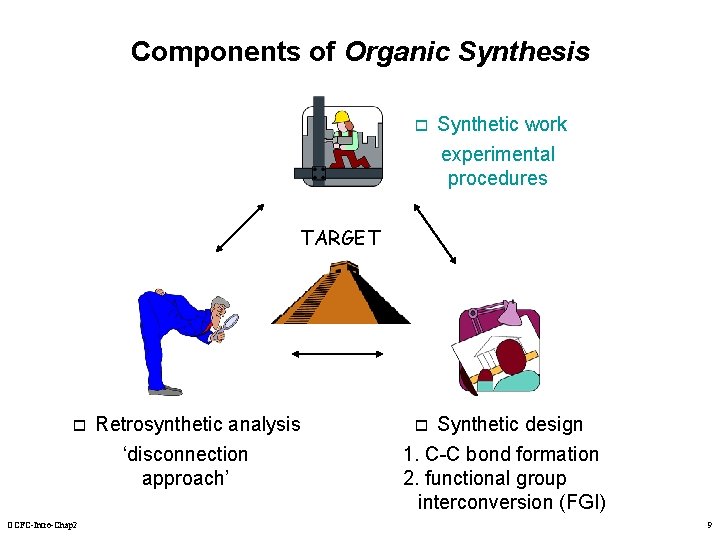
Components of Organic Synthesis o Synthetic work experimental procedures TARGET o OCFC-Intro-Chap 2 Retrosynthetic analysis ‘disconnection approach’ Synthetic design 1. C-C bond formation 2. functional group interconversion (FGI) o 9

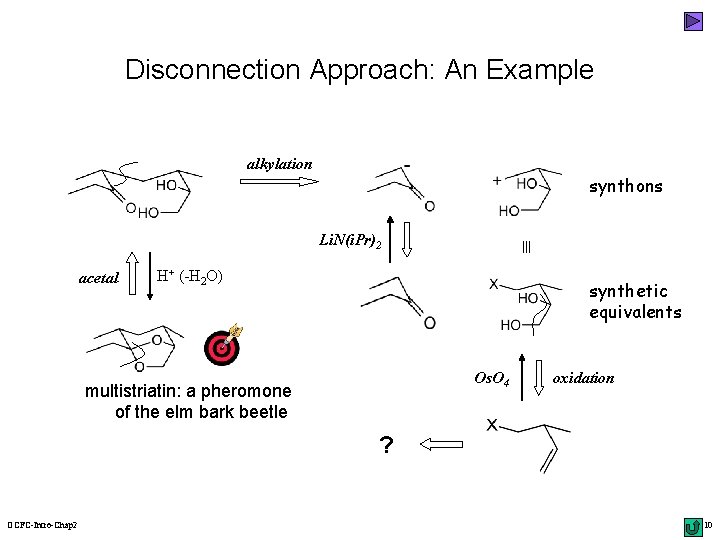
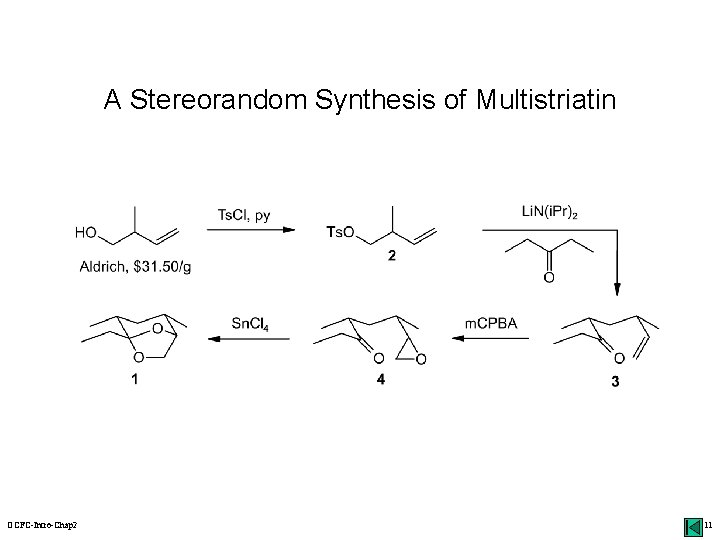
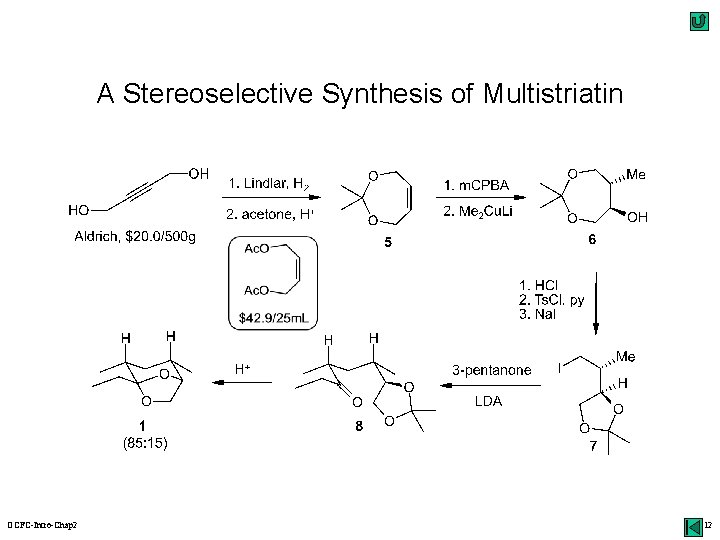
Disconnection Approach: An Example alkylation synthons acetal Li. N(i. Pr)2 H+ (-H 2 O) synthetic equivalents Os. O 4 multistriatin: a pheromone of the elm bark beetle oxidation ? OCFC-Intro-Chap 2 10

A Stereorandom Synthesis of Multistriatin OCFC-Intro-Chap 2 11

A Stereoselective Synthesis of Multistriatin OCFC-Intro-Chap 2 12

Design and Synthesis r Retrosynthetic Analysis 1. recognise the functional groups in the target molecule 2. disconnect by known methods and reliable reactions 3. repeat 1 and 2 until the readily available starting materials are obtained 4. design as many alternative retrosynthetic routes as possible r Synthesis 1. write down the synthetic schemes containing the detailed reaction conditions according to the analyses 2. compare the pros and cons between the syntheses designed; the number of steps, availability of reagents/starting materials, selectivity (chemo-/regio-/stereo-), economy, process, etc 3. modify the selected synthetic plan whenever unexpected problems are encountered OCFC-Intro-Chap 2 13

What to Know for Organic Synthesis molecular structure r reaction mechanisms r stereochemistry r dependable reactions r availability of compounds r selectivity r analytical methods r lab technique r process r creativity r economy r OCFC-Intro-Chap 2 14


Chapter 2 Functional Group Transformations r selectivity/specificity: chemo-, regio- & stereo- r functionalization of alkanes: unreactive uradical r reaction: selectivity; 5 bottom functionalization of alkenes: addition & substitution uaddition to double bonds: 6 Scheme 2. 1 m ‘(anti)Markovnikov’/oxymercuration/hydroboration/HBr: m stereoselectivity: uallylic OCFC-Intro-Chap 2 7 top 7 bottom substitution: radical; 8 top & 188~189 (oxidation) 16

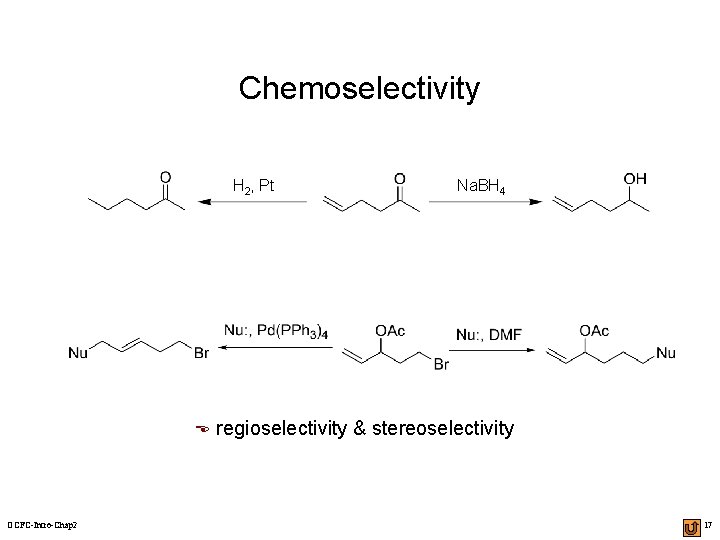
Chemoselectivity H 2, Pt E OCFC-Intro-Chap 2 Na. BH 4 regioselectivity & stereoselectivity 17

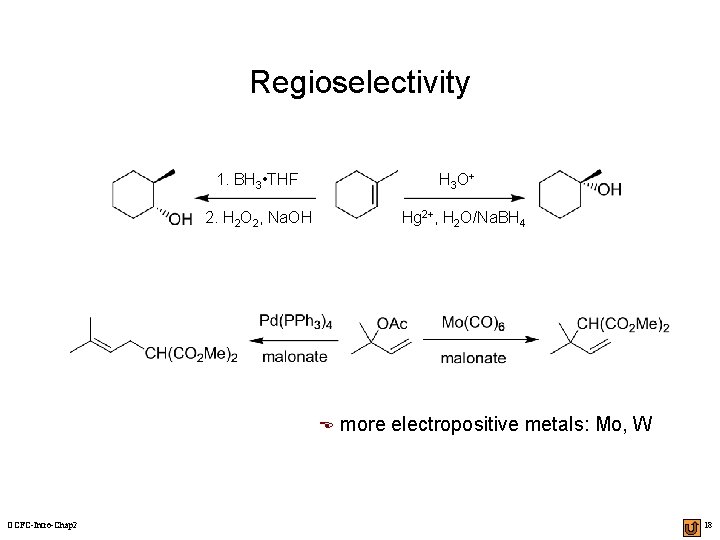
Regioselectivity 1. BH 3 • THF H 3 O + 2. H 2 O 2, Na. OH Hg 2+, H 2 O/Na. BH 4 E OCFC-Intro-Chap 2 more electropositive metals: Mo, W 18

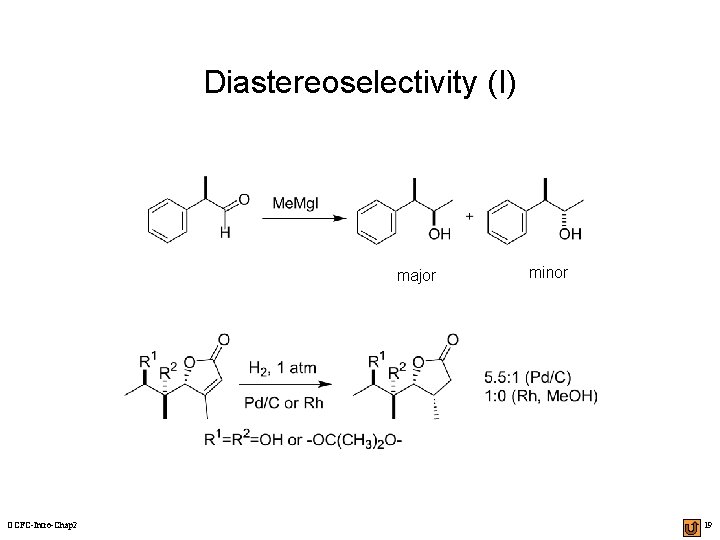
Diastereoselectivity (I) major OCFC-Intro-Chap 2 minor 19

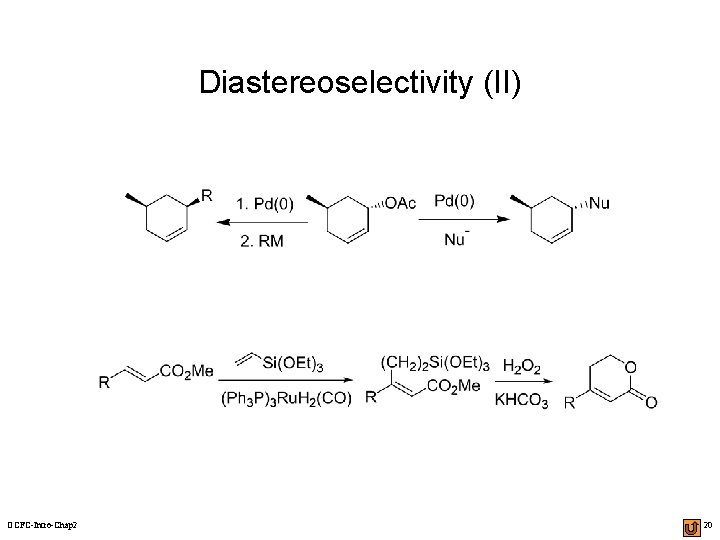
Diastereoselectivity (II) OCFC-Intro-Chap 2 20

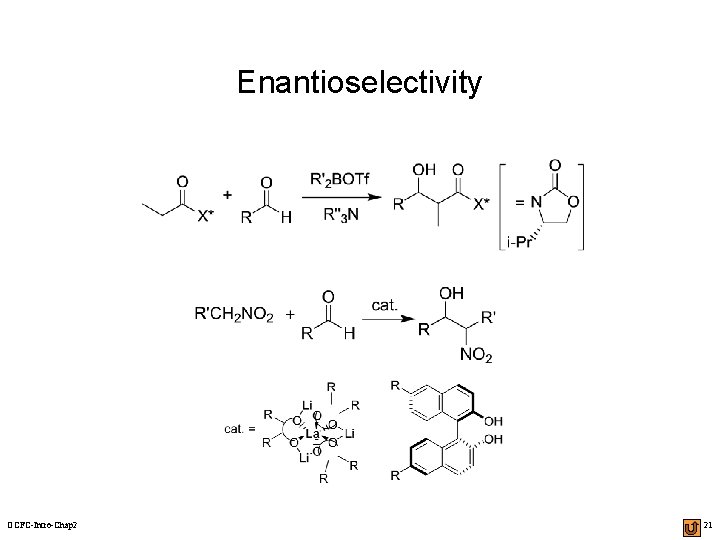
Enantioselectivity OCFC-Intro-Chap 2 21

Functionalization of Alkynes r addition / substitution reactions: 9 Scheme 2. 2 ureduction u. C-C to alkenes: 165 bottom – 166 bond formation: 51 – 52 top uelectrophilic addition: largely anti addition ma mixture of products: syn addition & participation of the reaction solvent, 8 bottom – 9 top m ketones OCFC-Intro-Chap 2 with oxymercuration: 9 middle 22

Functionalization of Aromatic Hydrocarbons r electrophilic substitution: the ring; 10 Scheme 2. 3 usubstituted m the rate & the orientation: 11 Table 2. 1 & 12 m lower r benzenes: depending on the substituents selectivity with a radical substitution: 13 top nucleophilic substitution: p-chloronitrobenzene u. Chichibabin r reaction ( 14 & 44) & benzyne intermediate benzylic oxidation: the side chain; 189~192 uautoxidation of cumene: acetone & phenol; 11 top uchlorination/bromination: OCFC-Intro-Chap 2 radical mechanism; 11 top 23

Functionalization of Aromatic Heterocycles r r pyridine: weak base & e--poor ring, 14 Scheme 2. 4 uelectrophilic substitution: difficult but higher e--density at C-3 unucleophilic substitution at C-2: ‘Chichibabin’; 14 Scheme 2. 5 usubstitution of pyridine-N-oxide at C-4: 15 Scheme 2. 6 pyrrole, furan, thiophene: e--rich; 16 Scheme 2. 7 usubstitution OCFC-Intro-Chap 2 at C-2 / C-5: resonance-stabilized 24

Interconversion of Functional Groups r alcohols & phenols: less basic; 17 Scheme 2. 8 r amines: basic & nucleophilic; 20 Scheme 2. 9 u amines as electrophiles: pyridinium ions; 18 bot & 19 top r halides: good leaving groups; 21 Scheme 2. 10 r aryl nitro compounds: 22 Scheme 2. 11 r aldehydes & ketones: addition, substitution, enolates r carboxylic acids: 23 Scheme 2. 12 OCFC-Intro-Chap 2 25