Why is Water Important to Living Things Water

















- Slides: 23


Why is Water Important to Living Things? • Water is the substance that makes possible life as we know it here on Earth • Covers ¾ of the Earth’s Surface • Makes up 70 -95% of cells; most cells are surrounded by water • Only common substance to exist in all three physical states of matter – Solid, Liquid, and Gas – in the natural environment.

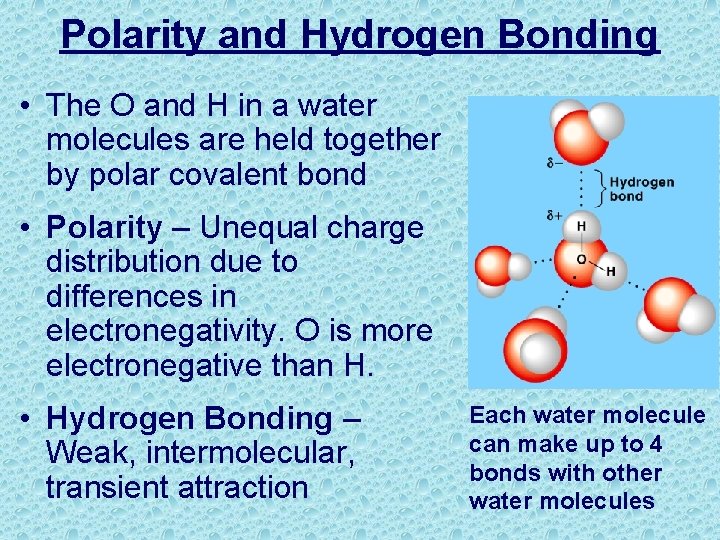
Polarity and Hydrogen Bonding • The O and H in a water molecules are held together by polar covalent bond • Polarity – Unequal charge distribution due to differences in electronegativity. O is more electronegative than H. • Hydrogen Bonding – Weak, intermolecular, transient attraction Each water molecule can make up to 4 bonds with other water molecules

Cohesion, Adhesion • Cohesion – Hydrogen bonds hold water molecules together. One water molecule can bond with up to 4 other water molecules at one time. • Adhesion – Allow water molecules to adhere to hydrophilic surfaces. Benefit to Life • Transport of water through the xylem of plants against gravity.

Water Cohesion/Adhesion • Allows water to move as continuous column upward through stems of plants

Surface Tension • Surface Tension – a measure of how difficult it is to stretch or break the surface of a liquid • Water has a greater surface tension than most other liquids • At the interface between water and air is an ordered arrangement of water molecules, hydrogen -bonded to one another and to the water below Benefit to Life • Some animals can stand, walk, or run on water without breaking the surface

Heat and Temperature • Anything that moves has kinetic energy, the energy of motion • The faster a molecule moves, the greater its kinetic energy. • Heat is a measure of the total quantity of kinetic energy due to molecular motion • Temperature measures the intensity of heat due to the average kinetic energy of the molecules – when the average speed of the molecules increases, a thermometer records this as a rise in temperature

High Specific Heat of Water • Amount of heat that must be absorbed or lost for 1 g of substance to change its temp. by 1 o. C • For water, the specific heat is 1 cal/g/o. C • Water resists changes in its temp. • Heat is absorbed to break H-bonds; released when bonds form.

High Specific Heat Benefit to Life • Large bodies of water (i. e. oceans) can absorb and store a huge amount of heat. In winter, heat lost to the air can warm it and make coastal areas milder in climate than inland regions. • Stabilizes temperatures – favorable environment for life – keeps land/water temperatures within limits that permit life. • Living things are mostly water – resists change in temperature and maintains somewhat constant temp.

High Heat of Vaporization • Energy required to change 1 g of substance from a liquid to a gas. • Water has a high heat of vap. – it takes a lot of energy to vaporize it. (580 cal at 25 o. C for 1 g of water) • An increase in heat breaks H-bonds releasing molecules to a gaseous state. Benefit to Life • The solar heat absorbed by tropical seas causes evaporation of water. As the moist air travels towards the poles it condenses to form rain

Evaporative Cooling • Water molecules with high kinetic energy (the “hottest” ones) evaporate (change from a liquid to a gas); remaining molecules are cooler. Benefit to Life • Stabilizes temperatures in lake and ponds. • Prevent terrestrial organisms from overheating. • High humidity prevents evaporation and sweat and makes us more uncomfortable.

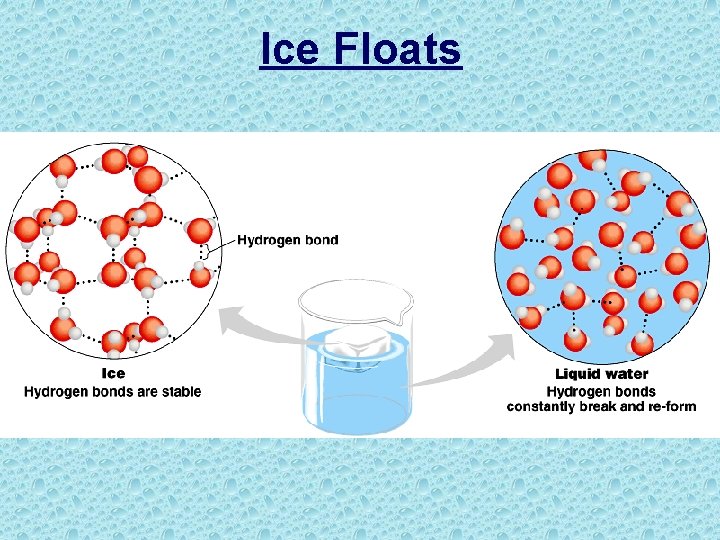
Ice Floats • Water expands as it freezes; less dense as a solid (fewer molecules for same volume) • Hydrogen bonds in ice keep the molecules far enough apart to make ice less dense than liquid water at 4 o. C or above. Benefit to Life • Floating ice insulates bodies of water so they don’t freeze solid – keeps the water below the ice from the colder air

Ice Floats

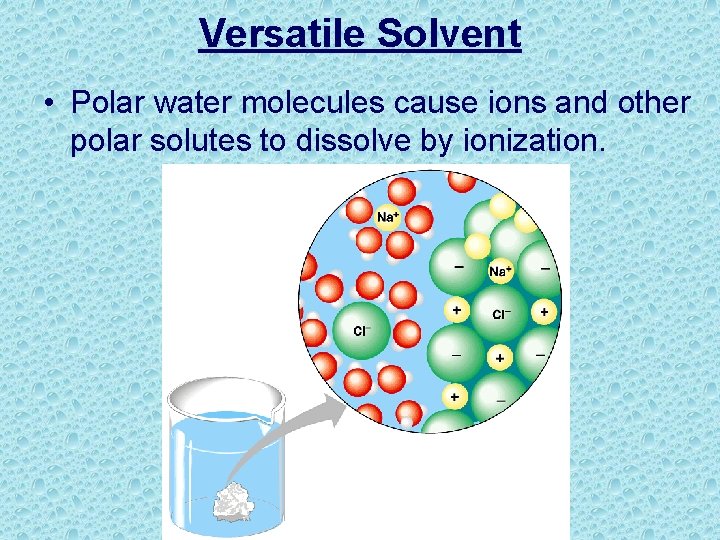
Versatile Solvent • Polar water molecules cause ions and other polar solutes to dissolve by ionization.

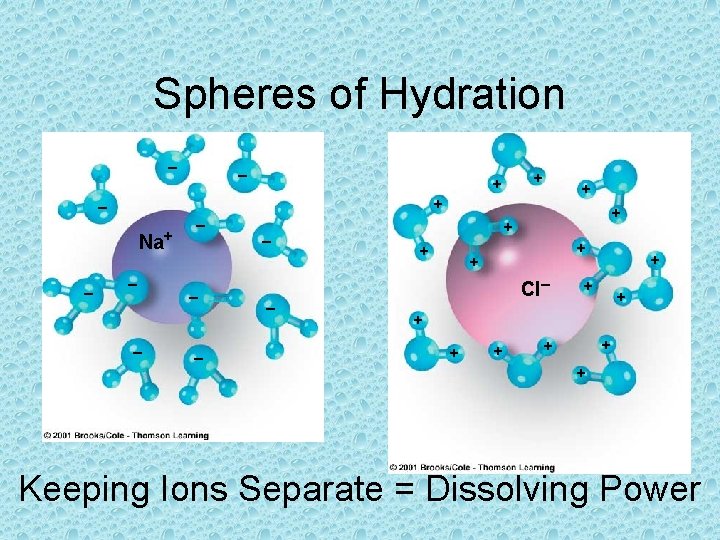
Spheres of Hydration – + + – Na+ – – – – – + + + Cl– + + + + + Keeping Ions Separate = Dissolving Power

Versatile Solvent • Solution – a liquid that is a completely homogeneous mixture of two or more substances • Solvent – the dissolving agent of a solution • Solute – the substance that is dissolved • Aqueous solution- solution where water is the solvent Benefit to Life • Most chemical reactions in living things involve solutes dissolved in water

To Dissolve or Not to Dissolve? • Hydrophilic: A substance with an affinity/able to dissolve in water. – What kind of substances? • Hydrophobic: A substance that repels water/don’t dissolve. – What kind of substances?

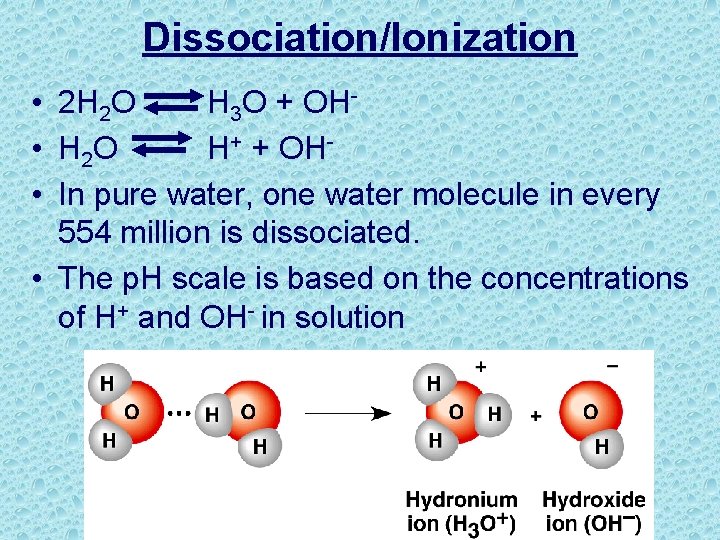
Dissociation/Ionization • 2 H 2 O H 3 O + OH • H 2 O H+ + OH • In pure water, one water molecule in every 554 million is dissociated. • The p. H scale is based on the concentrations of H+ and OH- in solution

Acids and Bases • Acid – when dissolved in water, add H+ ions to the solution. HCl H+ + Clp. H< 7 • Base – reduces the hydrogen ion concentration of a solution; work by either accepting hydrogen ions or dissociating to form hydroxide ions. Na. OH Na+ + OHp. H>7

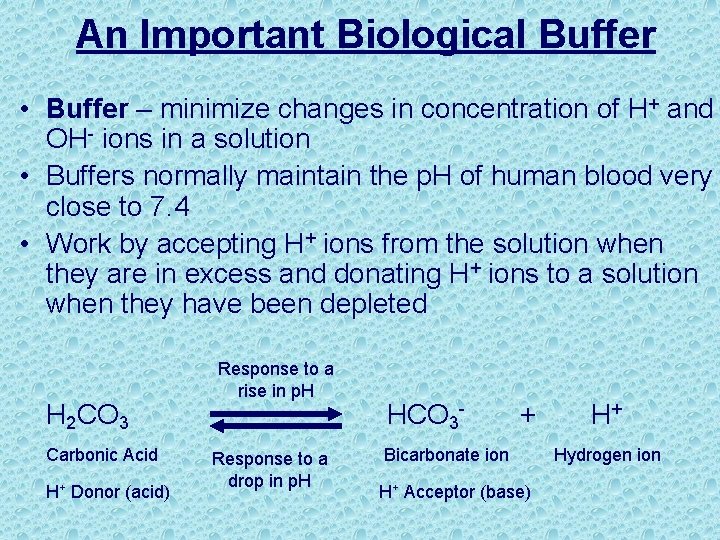
An Important Biological Buffer • Buffer – minimize changes in concentration of H+ and OH- ions in a solution • Buffers normally maintain the p. H of human blood very close to 7. 4 • Work by accepting H+ ions from the solution when they are in excess and donating H+ ions to a solution when they have been depleted H 2 CO 3 Carbonic Acid H+ Donor (acid) Response to a rise in p. H Response to a drop in p. H HCO 3 - + Bicarbonate ion H+ Acceptor (base) H+ Hydrogen ion

Carbonic Acid. Bicarbonate Buffer System • When blood p. H rises, carbonic acid dissociates to form bicarbonate and H+ H 2 C 03 -----> HC 03 - + H+ • When blood p. H drops, bicarbonate binds H+ to form carbonic acid HC 03 - + H+ -----> H 2 C 03

Acid Precipitation • Mostly sulfur oxides and nitrogen oxides dissolved in rainwater. • Come from fossil fuels burned in factories and automobiles • Aquatic animal’s egg and young are vulnerable to low p. H levels – can alter the structure of biochemical molecules and prevent them from carrying out essential chemical processes of life.

Acid Precipitation