Why is the Periodic Table important to me

Why is the Periodic Table important to me? • It organizes lots of information about all the known elements. What are the “known elements”? I’m glad you asked! http: //www. youtube. com/watch? v=Eg. Jv 2 ARBck. A

The Current Periodic Table • The elements are put in rows by ATOMIC NUMBER!! • The horizontal rows are called periods and are labeled from 1 to 7. – Identify atomic # 1 – Identify atomic # 8 – If these two elements combine in a chemical compound, what is the new substance called? • The vertical columns are called groups/families are labeled from 1 to 18. – Identify atomic #11 – Identify atomic #19 – Why are these elements in the same group/family?
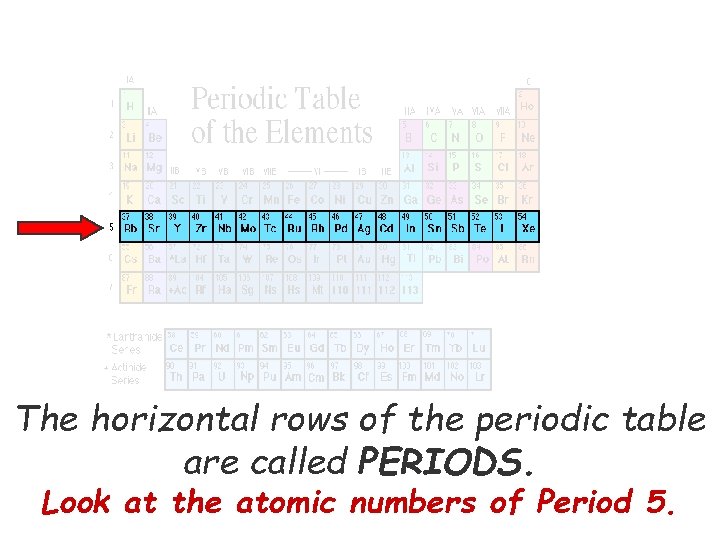
The horizontal rows of the periodic table are called PERIODS. Look at the atomic numbers of Period 5.

Groups…Here’s Where the Periodic Table Gets Useful!! • Elements in the same group have similar chemical and physical properties!!
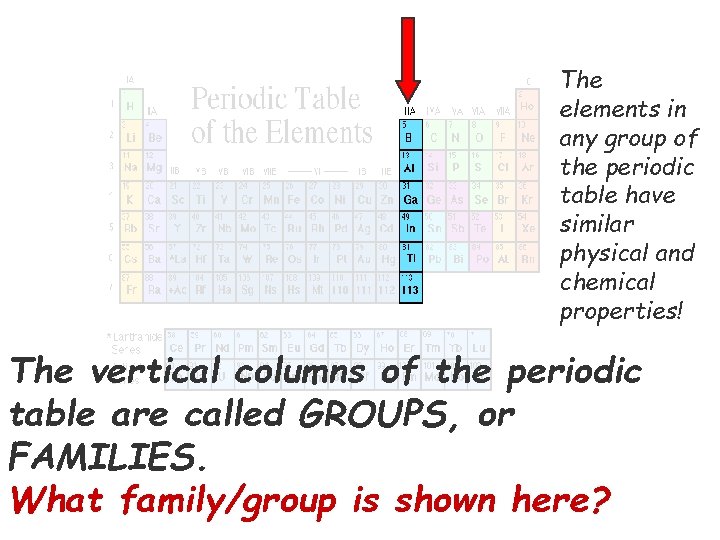
The elements in any group of the periodic table have similar physical and chemical properties! The vertical columns of the periodic table are called GROUPS, or FAMILIES. What family/group is shown here?

Families on the Periodic Table • Columns are also grouped into families. – We have observed Hydrogen, 1, 2, and 8 • Families may be one column, or several columns put together. – We have observed the Transition Elements • Families have names (Just like your family has a common last name. )

Hydrogen – in a family all by itself • Hydrogen belongs to a family of its own. . • Hydrogen was involved in the explosion of the Hindenberg. • Hydrogen is promising as an alternative fuel source for automobiles aka.

HYDROGEN – The Hindenburg • http: //www. youtu be. com/watch? v =xi. AT 9 xv. TVKI&f eature=related • http: //www. youtu be. com/watch? v =q 7 Fc 8 k. Iy. MCo& feature=related

Alkali Metals – this is Family 1’s NAME • 1 st column on the periodic table (Group 1) not including hydrogen. • Soft enough to cut with a butter knife • Very reactive or volatile when exposed to moisture (aka. Water) • Combined with something else in nature (like in salt, Na + Cl, Na. Cl).
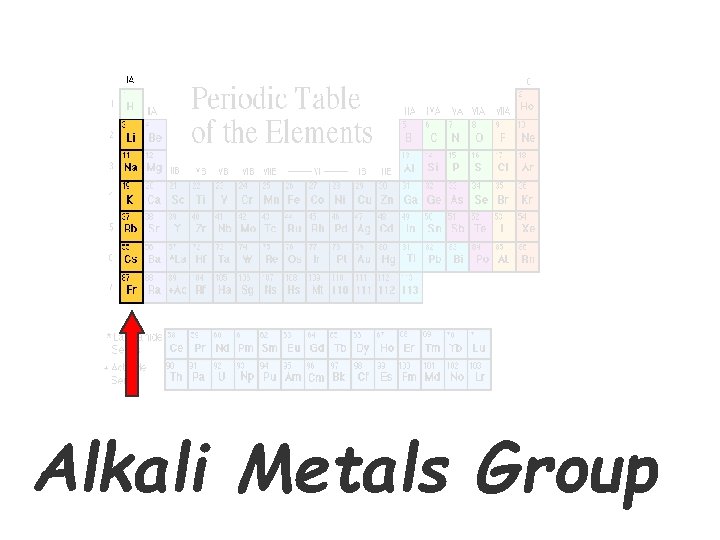
Alkali Metals Group

The SOFT BUT REACTIVE family Soft metals Very reactive • http: //www. youtube. com/watc h? v=uixx. Jt. JPVXk&feature=rel ated

Alkaline Earth Metals • Second column on the periodic table. (Group 2) • Fairly Reactive metals that are always combined with nonmetals in nature. (like in milk Ca + P + O, Ca. PO 4 • Several of these elements are important mineral nutrients for our bodies (such as Mg, K,
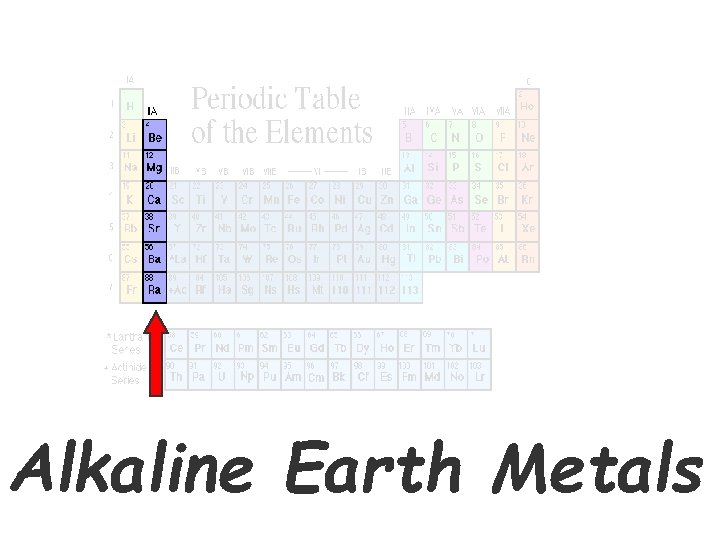
Alkaline Earth Metals
Transition Metals • Elements in groups 3 -12 • Less reactive harder metals • Includes metals used in jewelry and construction. • Many have very LOW REACTIVITY which is why they are good for coins, nails, bolts, jewelry, bridges. • Good thing bridges are not made from Sodium #11, huh?
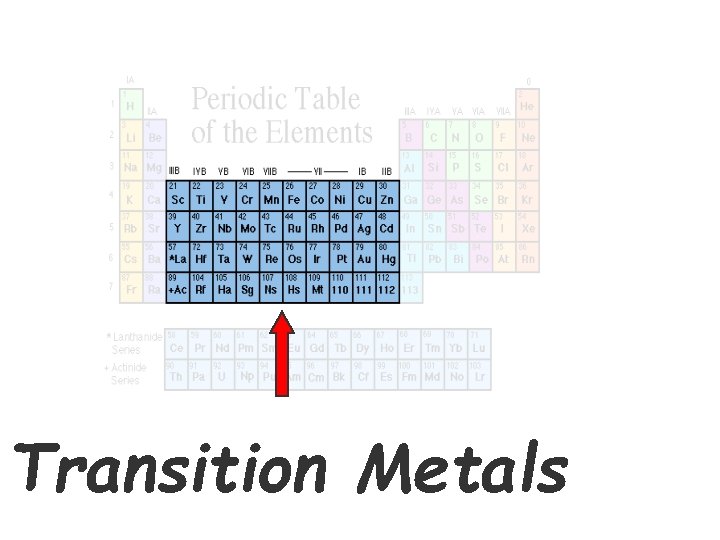
Transition Metals

The Noble Gases

The Noble Gases • Elements in group 18 • VERY unreactive gases • Used in lighted “neon” signs (He, Ne, Ar, Xe) • Used in blimps to fix the Hindenberg problem (He).
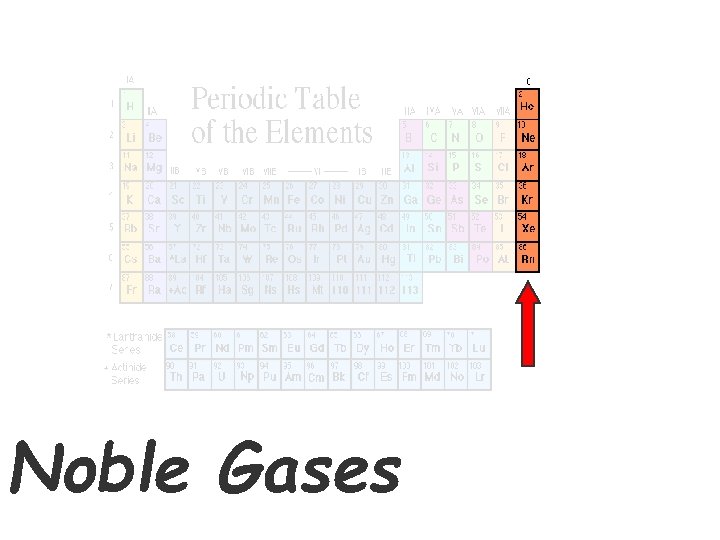
Noble Gases

Atoms of elements combine to make EVERYTHING …to make MATTER …to make all things that have MASS and VOLUME …aka. The AMOUNT OF SPACE something occupies …they are grouped by how they are alike • http: //www. youtube. com/watch? v=jq 37 UQ-
- Slides: 19