Why do Covalent bonds form When nonmetals react

Why do Covalent bonds form? When non-metals react with one another, their atoms share valence electrons. The bond formed between atoms that share electrons is known as covalent bonds After bonding, each atom attains the electronic configuration of a noble gas.

What are molecules? When atoms combined by sharing electrons, a molecule is formed. A molecule is a group of 2 or more atoms held together by covalent bonds.

Molecules of Elements Many non-metallic Elements exist as molecules made up of two or more identical atoms. Let’s look at how electrons are shared and arranged within molecules of some elements: hydrogen, chlorine and oxygen
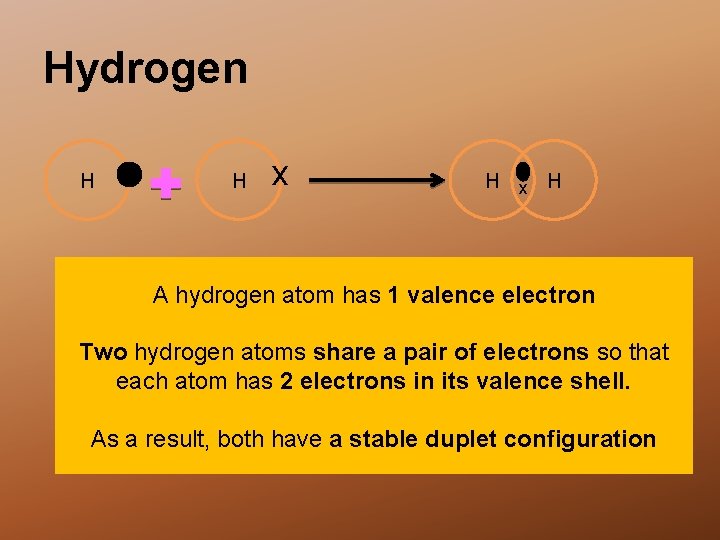
Hydrogen H H x H A hydrogen atom has 1 valence electron Two hydrogen atoms share a pair of electrons so that each atom has 2 electrons in its valence shell. As a result, both have a stable duplet configuration
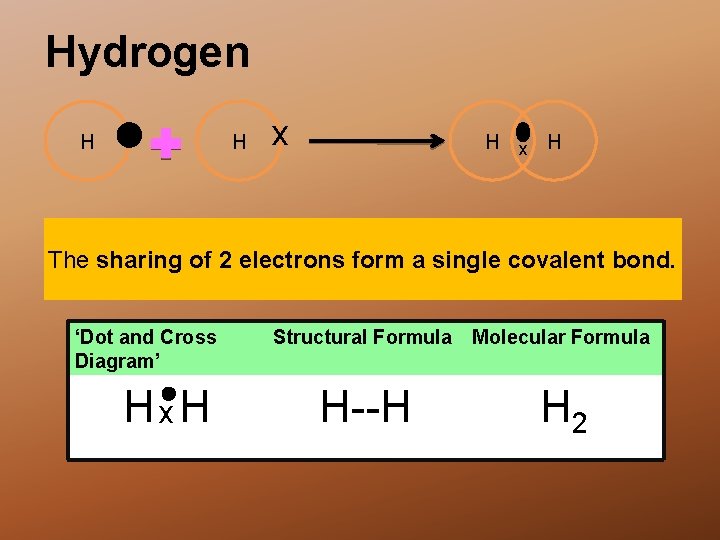
Hydrogen H H x H The sharing of 2 electrons form a single covalent bond. ‘Dot and Cross Diagram’ Hx. H Structural Formula Molecular Formula H--H H 2
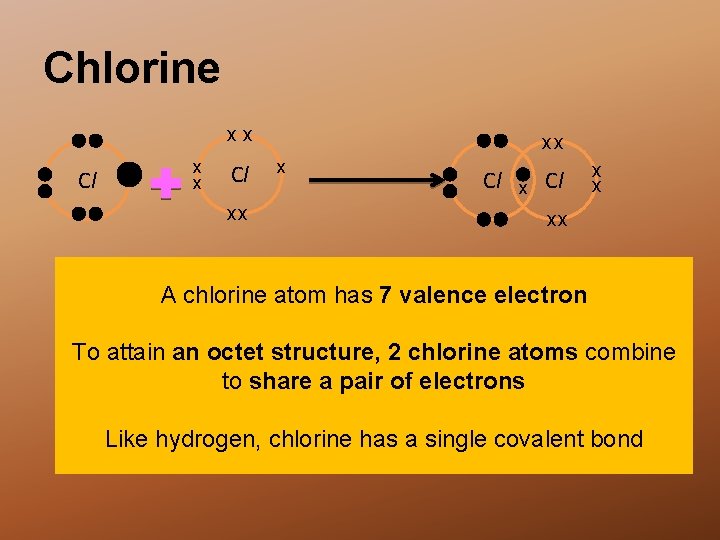
Chlorine xx Cl xx xx x Cl x x xx A chlorine atom has 7 valence electron To attain an octet structure, 2 chlorine atoms combine to share a pair of electrons Like hydrogen, chlorine has a single covalent bond
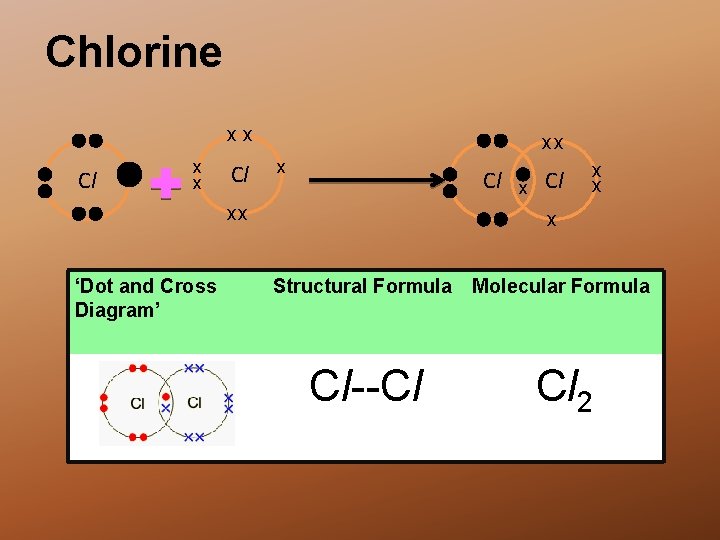
Chlorine xx Cl xx ‘Dot and Cross Diagram’ x Cl x x x Structural Formula Molecular Formula Cl--Cl Cl 2
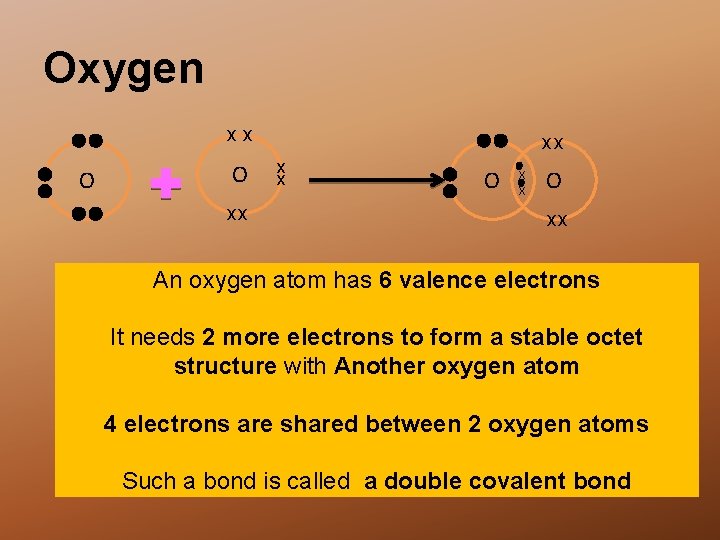
Oxygen xx O O xx xx x x O xx An oxygen atom has 6 valence electrons It needs 2 more electrons to form a stable octet structure with Another oxygen atom 4 electrons are shared between 2 oxygen atoms Such a bond is called a double covalent bond
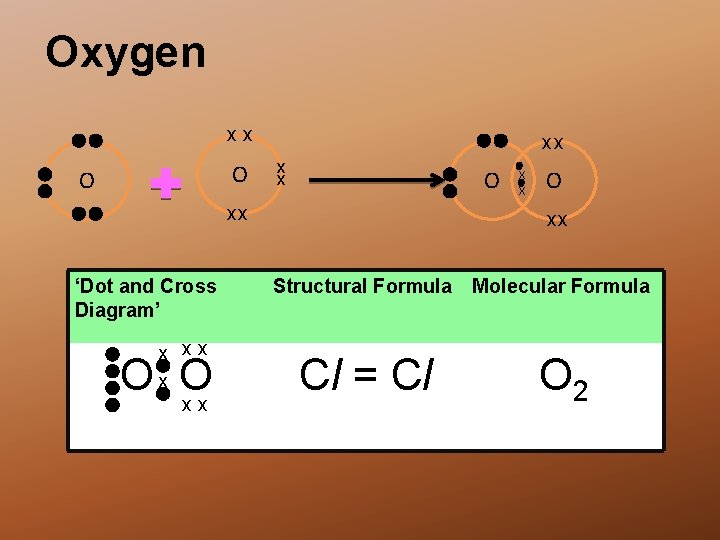
Oxygen xx O O xx x x O xx ‘Dot and Cross Diagram’ x xx Ox O xx x x O xx Structural Formula Molecular Formula Cl = Cl O 2

Molecules of Compounds When 2 or more different types of atoms form covalent bonds, a molecular compounds or covalent compound is formed. Water, methane and carbon dioxide are examples of covalent compounds

Water A water molecule is formed by the reaction of 2 hydrogen atoms with one oxygen atoms. All 3 atoms in the molecule attain noble gas configuration. ‘Dot and Cross Diagram’ x x xx Ox x H H A Structural Formula Molecular Formula O–H O A H 2 O Each water molecule contains 2 single covalent bonds.
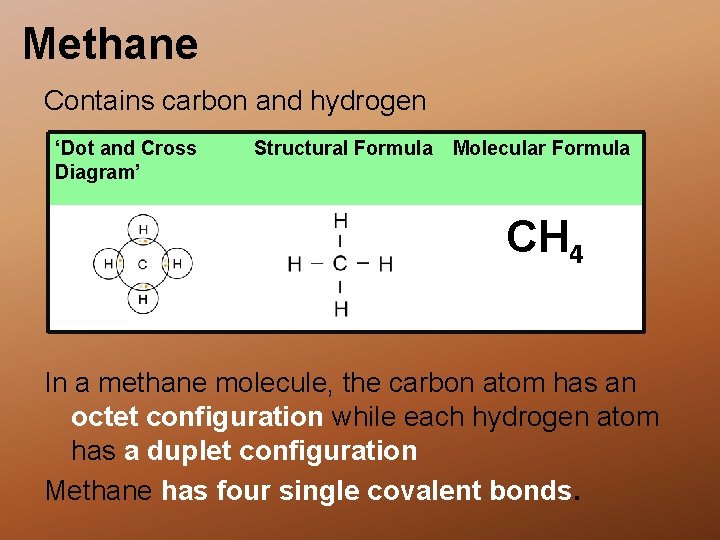
Methane Contains carbon and hydrogen ‘Dot and Cross Diagram’ A Structural Formula Molecular Formula CH 4 In a methane molecule, the carbon atom has an octet configuration while each hydrogen atom has a duplet configuration Methane has four single covalent bonds.
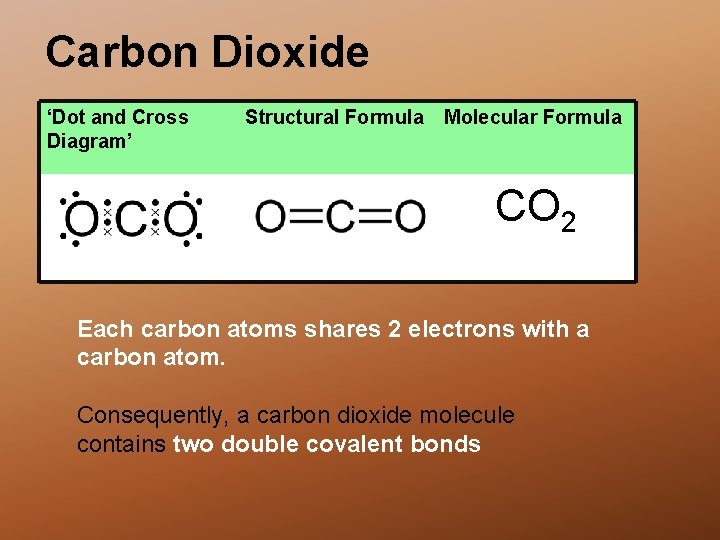
Carbon Dioxide ‘Dot and Cross Diagram’ A Structural Formula Molecular Formula CO 2 Each carbon atoms shares 2 electrons with a carbon atom. Consequently, a carbon dioxide molecule contains two double covalent bonds
- Slides: 13