Why do atoms form bonds To attain a


Why do atoms form bonds? To attain a noble gas configuration How do atoms form bonds? By gaining, losing, or sharing electrons Gain or loss of electrons ionic bonding Sharing of electrons covalent bonding

Covalent Bonding • Results from: electrostatic attraction between nucleus (protons) one atom & electrons of neighbor’s atom
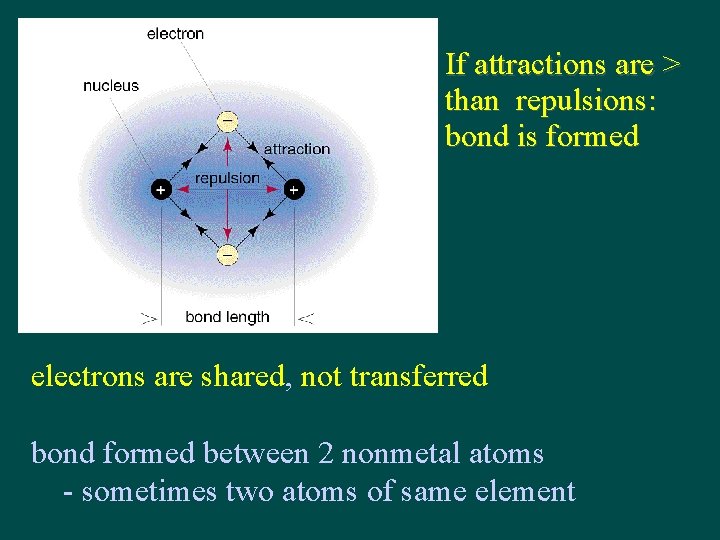
If attractions are > than repulsions: bond is formed electrons are shared, not transferred bond formed between 2 nonmetal atoms - sometimes two atoms of same element
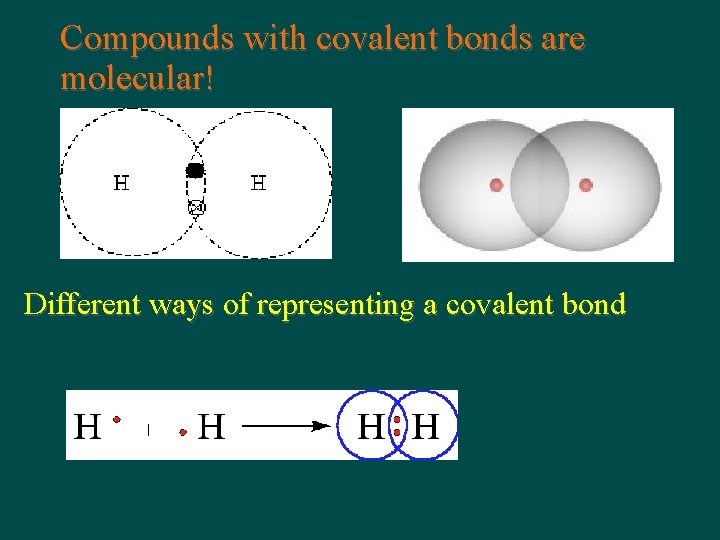
Compounds with covalent bonds are molecular! Different ways of representing a covalent bond

Recognizing covalent formulas • Covalent bonding occurs between nonmetal atoms so… – formulas contain only nonmetals

Identify Bond Type from Formula CH 4 Covalent Na. Cl Mg Ionic Cu Metallic SO 2 Covalent H 2 O Covalent Cs. F Ionic Metallic Na 2 S Ionic CO 2 Covalent Ag Metallic Ca. Br 2 Ionic Al 2 O 3 Ionic Al Metallic N 2 O 5 Covalent Li 3 N Au Ionic Metallic CH 3 OHCovalent Zn. O Ionic

Structure of Covalent Compounds • form molecules • more reactants have, the more molecules can make
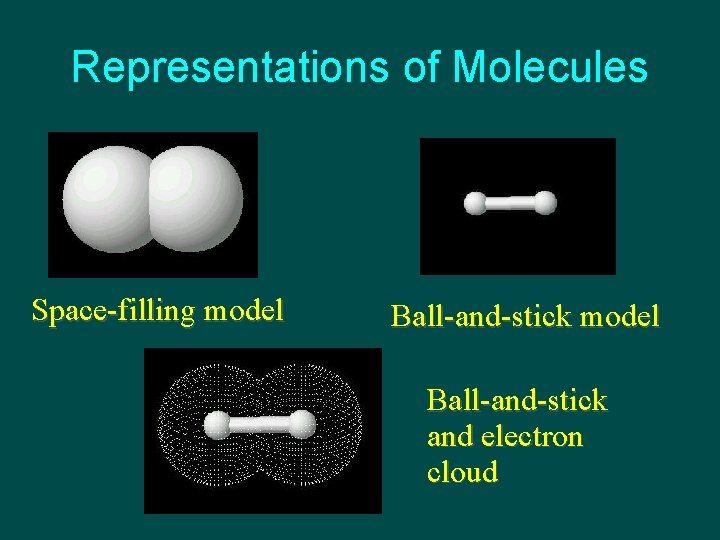
Representations of Molecules Space-filling model Ball-and-stick and electron cloud
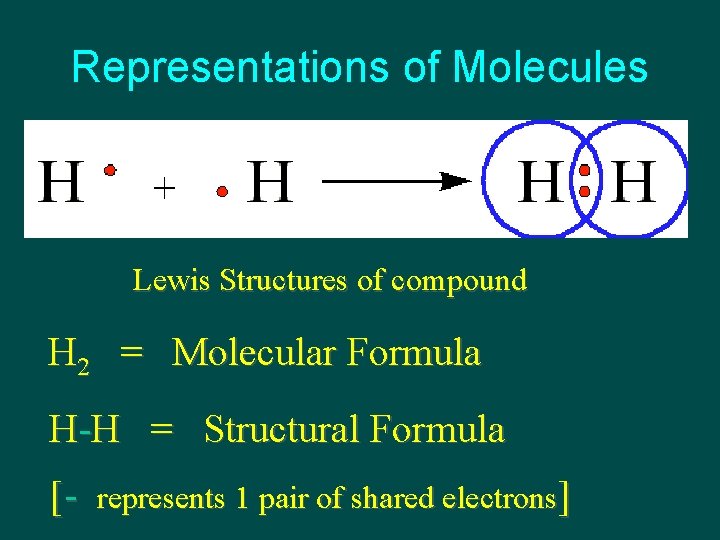
Representations of Molecules Lewis Structures of compound H 2 = Molecular Formula H-H = Structural Formula [- represents 1 pair of shared electrons]
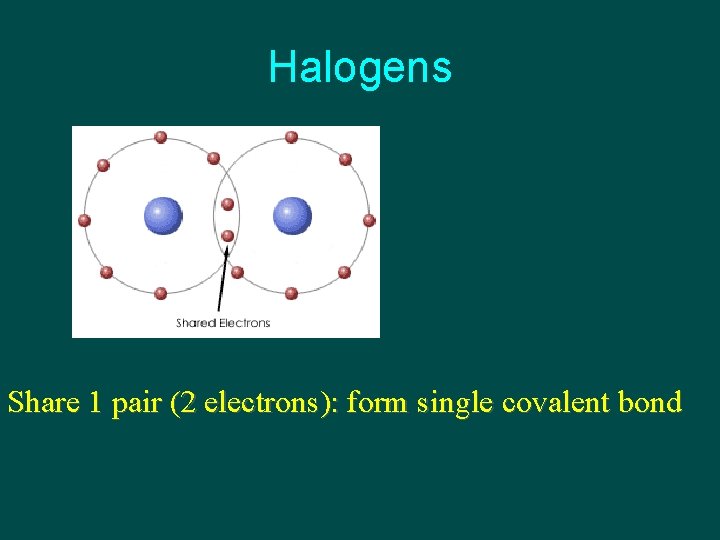
Halogens Share 1 pair (2 electrons): form single covalent bond

Rules for drawing Lewis Diagrams 1. Arrange symbols on paper the way think atoms are arranged - Diatomics easy – next to each other - Hydrogens always terminal (on outside) - Atom with least attraction (lowest electronegativity) for shared electrons placed in center 2. Add up # valence electrons all atoms 3. Distribute electrons, start with single bonds (electron pairs) between all atoms 4. Test validity (2 tests)

Tests for Lewis Structures Must Pass Both! 1. # dots = # valence electrons found in rule 2 2. Every atom has octet of electrons around it (except H: needs 2) Bonding electrons get counted 2 times – once for each atom sharing them

Assessing Lewis Diagrams • If dot structure passes both tests, you’re finished • If fails one or both tests, try again • If single bonds don’t work - try multiple bonds – Single bond = 2 electrons shared – Double bond = 4 electrons shared – Triple bond = 6 electrons shared

Try some examples • H 2, F 2, HF, O 2, N 2 • Step 1: Draw symbols the way think atoms are arranged – Diatomics easy – atoms next to each other!

Drawing Lewis Diagrams H : H Step 2: Count up the valence electrons Each H has 1 valence electron so the total = 2 Step 3: Distribute the valence electrons, starting with single bonds between all atoms

Testing Lewis Diagrams Test 1: 2 dots in diagram = 2 valence electrons H : H Test 2: Each H has 2 valence electrons (Remember H only wants 2)
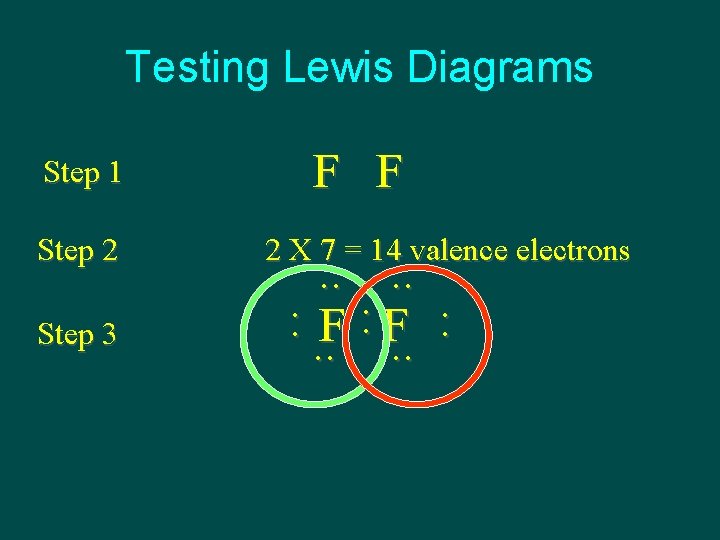
Testing Lewis Diagrams Step 1 Step 2 Step 3 F F 2 X 7 = 14 valence electrons . . : . . F : F. . :
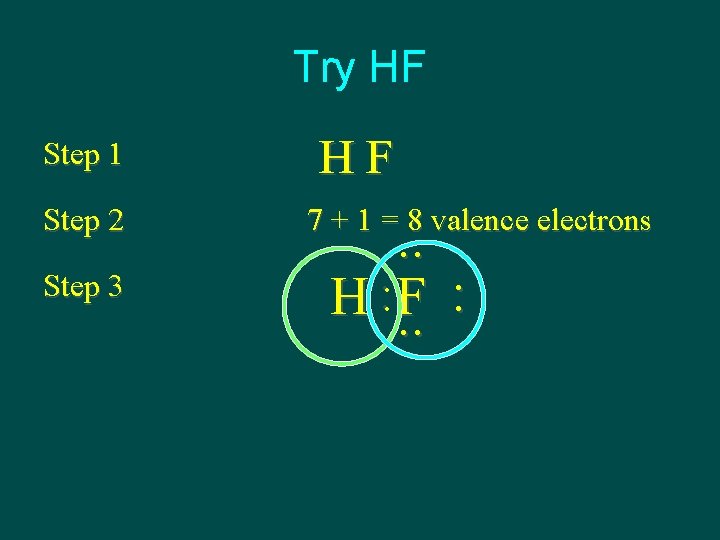
Try HF Step 2 Step 3 HF 7 + 1 = 8 valence electrons . . : H F. . : Step 1

Types of Covalent Bonds • Single: 2 atoms share 1 pair of electrons • Double: 2 atoms share 2 pairs of electrons • Triple: 2 atoms share 3 pairs of electrons

Try O 2 Step 1: OO Step 2: 2 X 6 = 12 valence electrons Step 3: . . : O : . . Step 4: Test failed No Good!

Try O 2 Again! Step 1: Step 2: 2 X 6 =12 . . : : O. . Step 3: Distribute electrons (single bond between atoms didn't work so try a double bond) Step 4: Test!
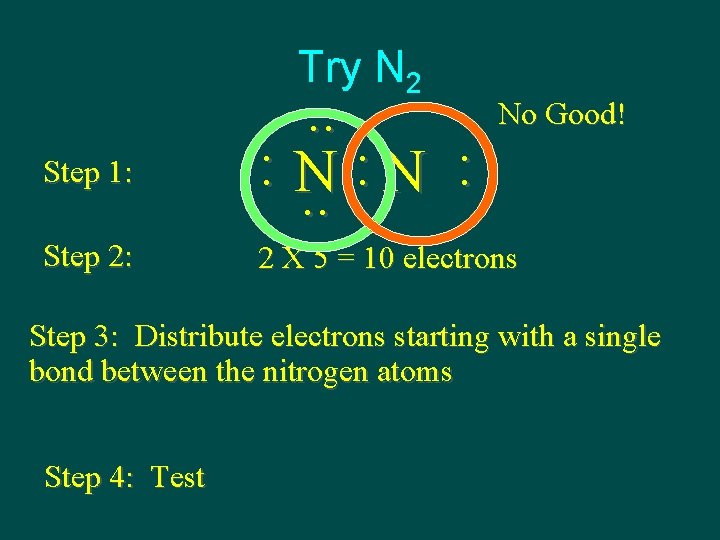
Try N 2 Step 1: Step 2: . . : N : : N. . No Good! 2 X 5 = 10 electrons Step 3: Distribute electrons starting with a single bond between the nitrogen atoms Step 4: Test

Try N 2 with a double bond : N : . . No Good!

Try N 2 with a triple bond : N : : : N : Step 4: Test
- Slides: 25