Why do atoms form bonds To attain a


Why do atoms form bonds? To attain a noble gas configuration.

How do atoms form bonds? • By gaining, losing, or sharing electrons. • Gain or loss of electrons ionic bonding • Sharing of electrons covalent bonding • Type of bonding is determined by the electronegativity difference between the atoms.

R U L E O F T H U M B Electronegativity Difference, EN EN 0 – 0. 3 Type of Bonding Nonpolar Covalent 0. 4 – 1. 7 Polar Covalent > 1. 7 Ionic For ΔEN between 1. 6 and 2. 0: If a metal is involved, the bond is considered ionic. If only nonmetals are involved, the bond is considered polar covalent.
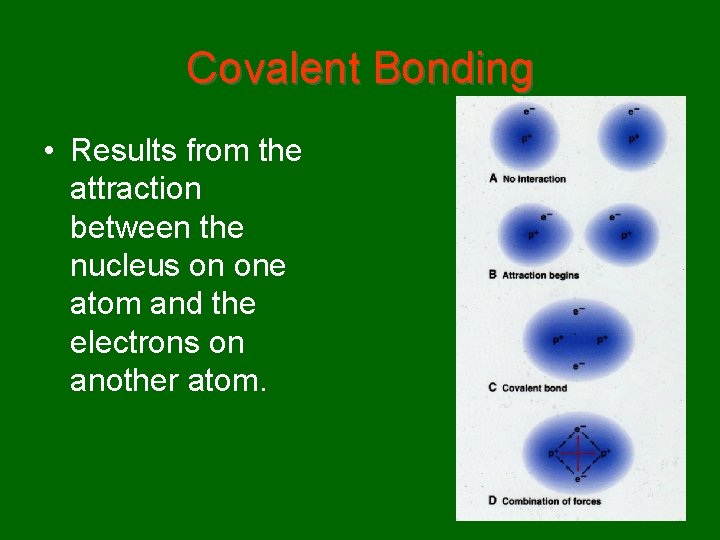
Covalent Bonding • Results from the attraction between the nucleus on one atom and the electrons on another atom.

If Attractions are greater than Repulsions, a bond is formed. The electrons are shared, but not transferred. Often formed between 2 nonmetal atoms of the same element.
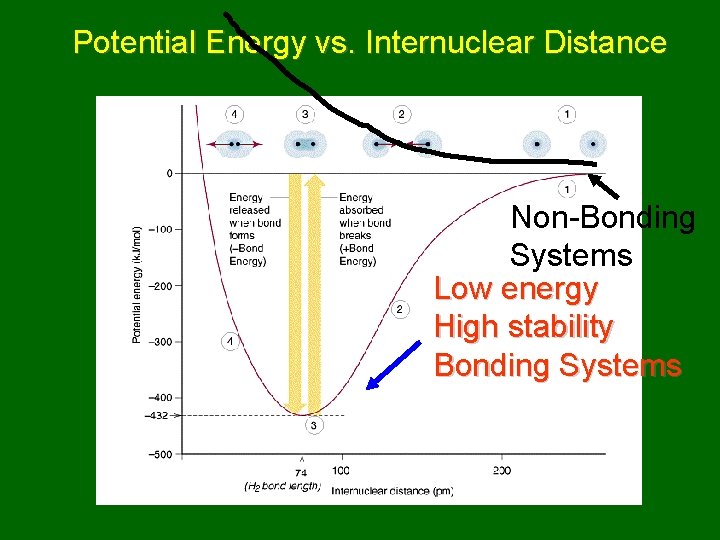
Potential Energy vs. Internuclear Distance Non-Bonding Systems Low energy High stability Bonding Systems
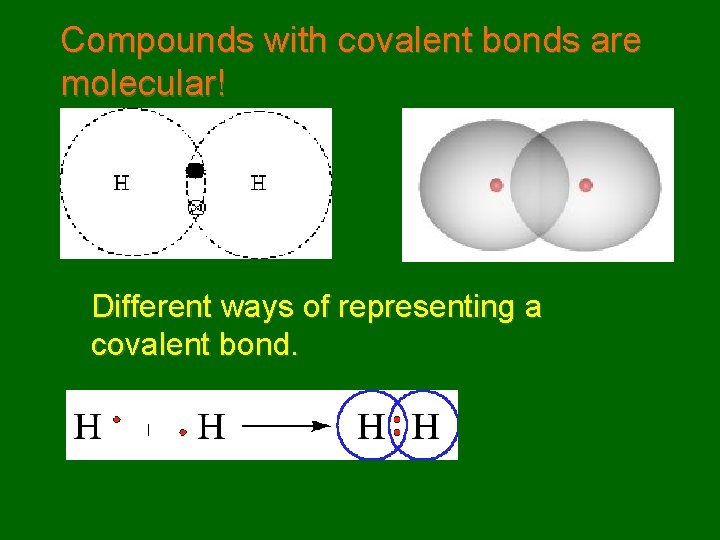
Compounds with covalent bonds are molecular! Different ways of representing a covalent bond.

Recognizing covalent formulas • Covalent bonding occurs between nonmetal atoms. • Formulas contain nonmetals.

Identify Bond Type from Formula CH 4 Coval Na. Cl Mg Cu SO 2 Coval Ionic Metal H 2 O Coval Cs. F Ionic Metal Na 2 S Li 3 N Ionic CO 2 Coval Ag Ca. Br 2 Ionic Al 2 O 3 Ionic N 2 O 5 Coval Al Metal Au Ionic Metal CH 3 OH Coval Zn. O Ionic


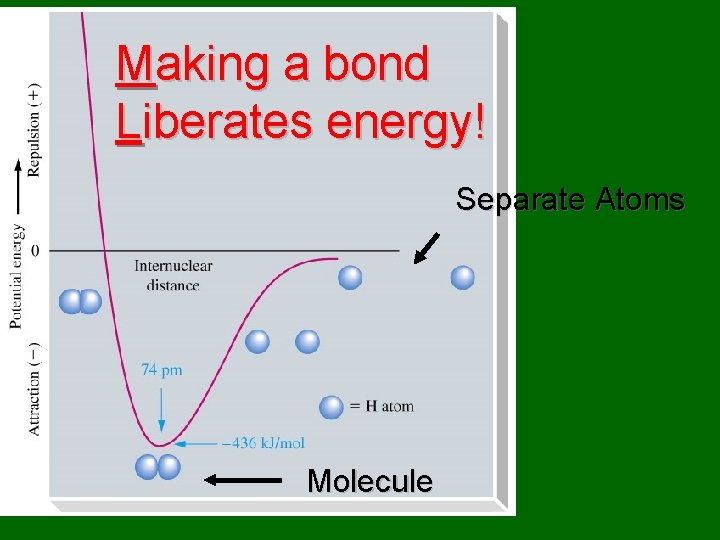
Making a bond Liberates energy! Separate Atoms Molecule

A + B AB + energy • A and B are both atoms • AB is a molecule • In this particular case, no bonds are broken and one bond is formed. • Energy is released. • The reaction is exothermic.

A + B AB + energy • A bond is formed & energy is released. • AB has less potential energy than A + B. • AB is more stable than A + B

Breaking a Bond Absorbs Energy!

CD + energy C + D • • CD is a molecule; C and D are atoms. One bond is broken; no bonds are formed. This reaction is endothermic. C and D have higher potential energy than CD.

Structure of Covalent Compounds • They form molecules. • The more reactants you have, the more molecules you can make.
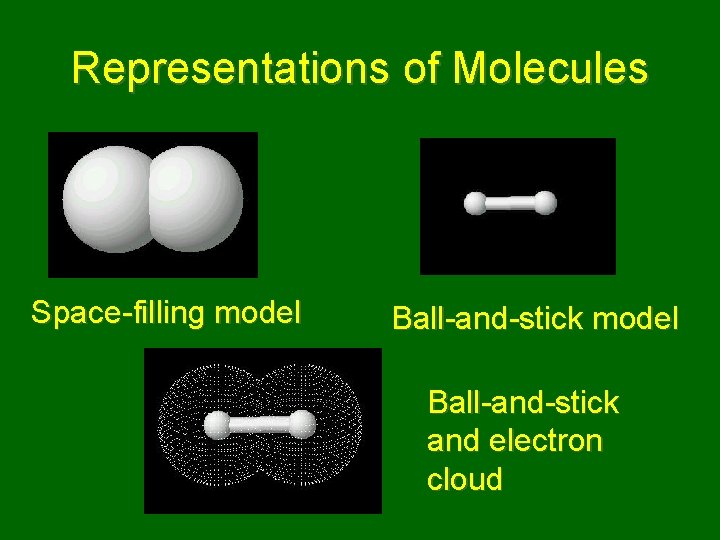
Representations of Molecules Space-filling model Ball-and-stick and electron cloud
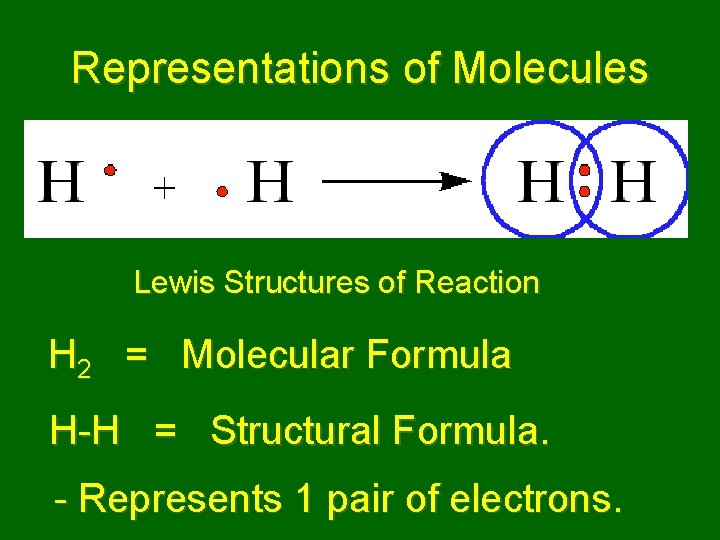
Representations of Molecules Lewis Structures of Reaction H 2 = Molecular Formula H-H = Structural Formula. - Represents 1 pair of electrons.
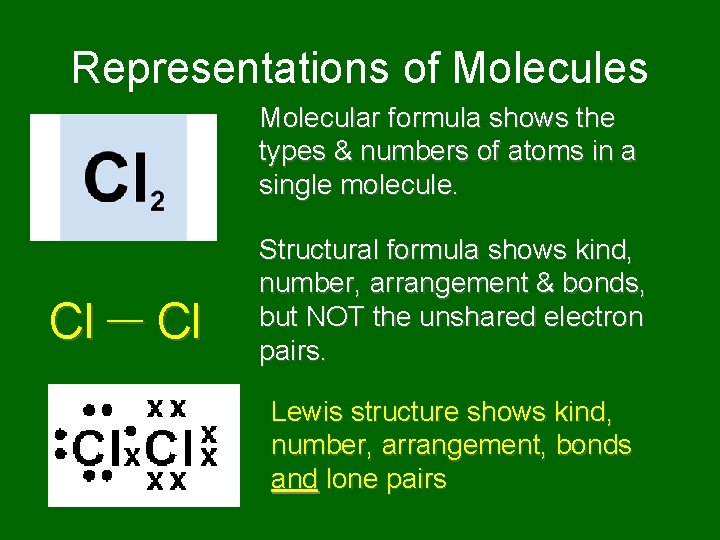
Representations of Molecules Molecular formula shows the types & numbers of atoms in a single molecule. Cl Structural formula shows kind, number, arrangement & bonds, but NOT the unshared electron pairs. Lewis structure shows kind, number, arrangement, bonds and lone pairs
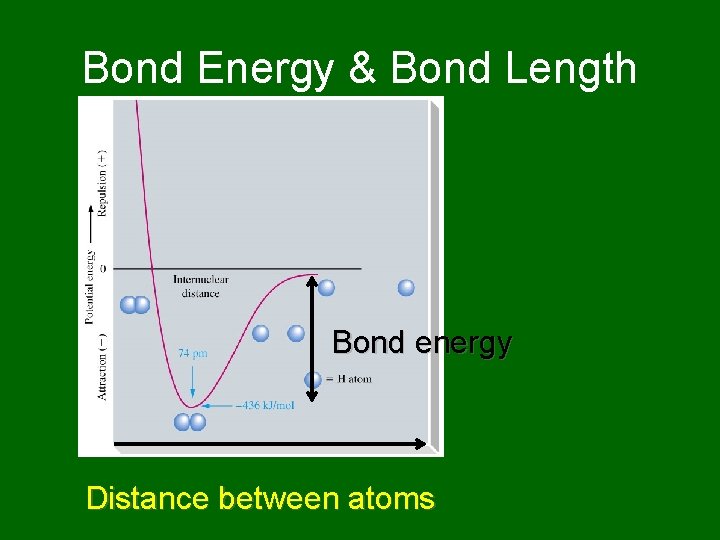
Bond Energy & Bond Length Bond energy Distance between atoms
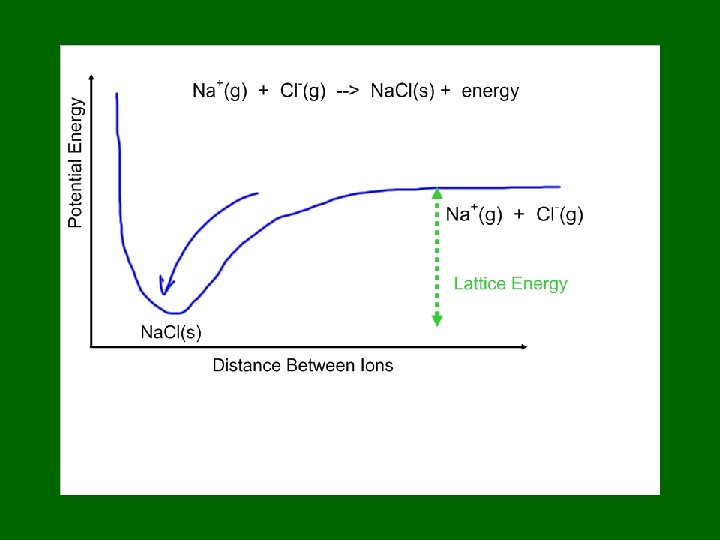

Atoms form bonds to … • Satisfy the octet rule. • But that simple statement doesn’t say much about HOW atoms share electrons. • Covalent Bonds result from the OVERLAP of two half-filled orbitals, one from each atom involved in the bond.
Recall orbitals

Geometry of orbitals • s orbitals are sherically symmetric, so no angular requirements. They can overlap any old way. • p orbitals have a definite geometry. They point along the x, y, & z axes, so you have to pay attention to the geometry.

Overlap of s orbitals • Even if you rotate the orbitals, they can still overlap.
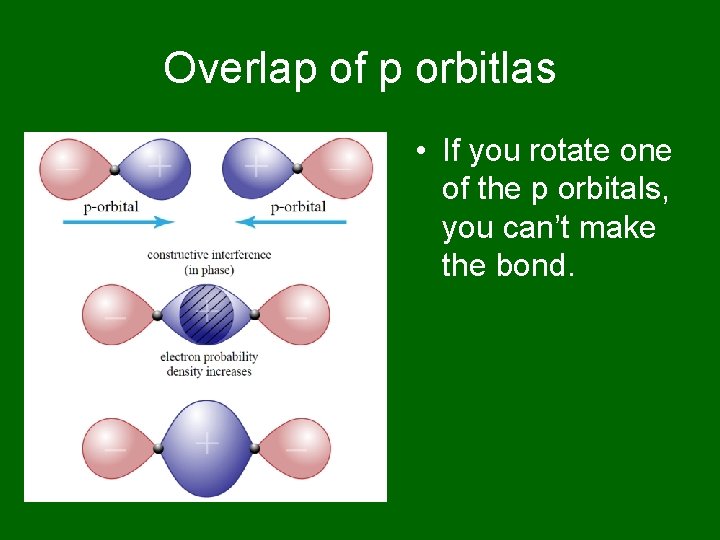
Overlap of p orbitlas • If you rotate one of the p orbitals, you can’t make the bond.
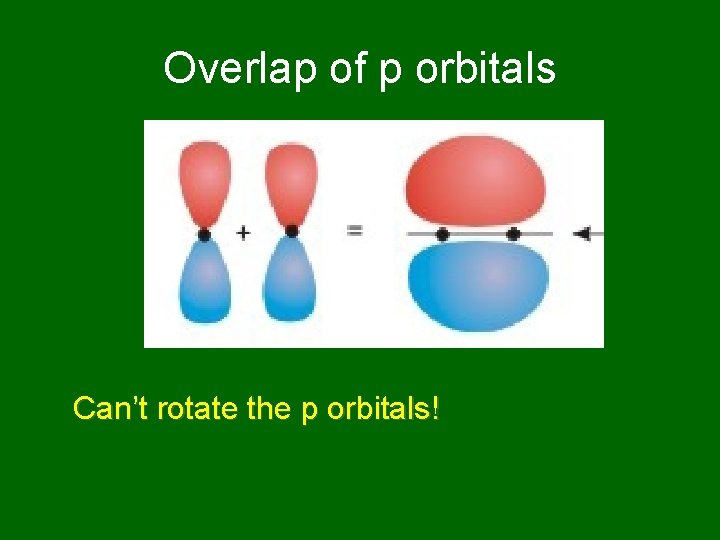
Overlap of p orbitals Can’t rotate the p orbitals!
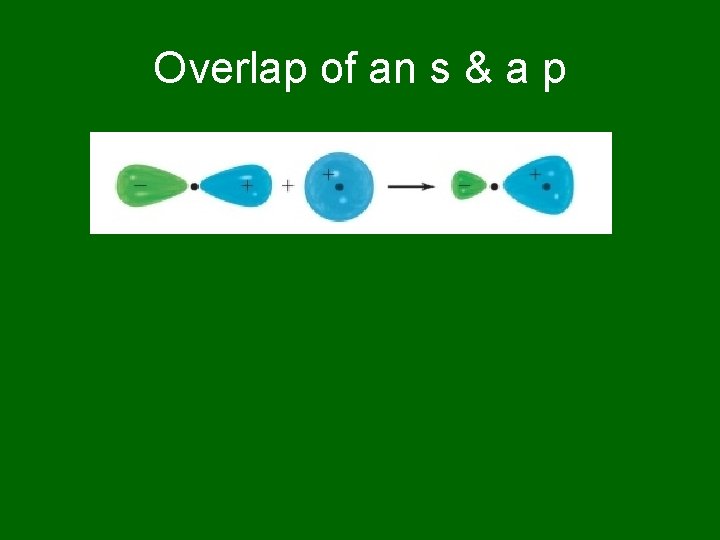
Overlap of an s & a p

Conclusion • Forming a bond by the overlap of half-filled orbitals means YOU MUST pay attention to the geometry of the orbitals. • Except for s orbitals, overlap requires specific orientations of orbitals. • More on this later ….
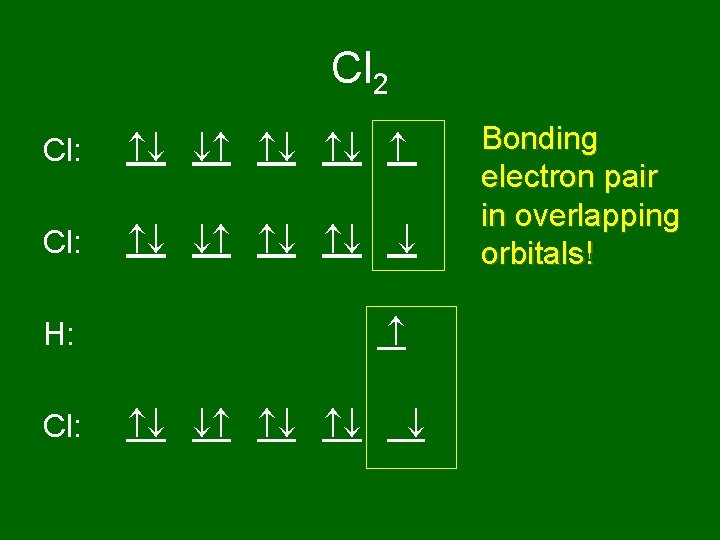
Cl 2 Cl: H: Cl: Bonding electron pair in overlapping orbitals!

Single vs. Multiple Bonds • Single bond: covalent bond in which one pair of electrons is shared between two atoms. • Double bond: covalent bond in which two pairs of electrons are shared between two atoms. • Triple bond: covalent bond in which three pairs of electrons are shared between two atoms.
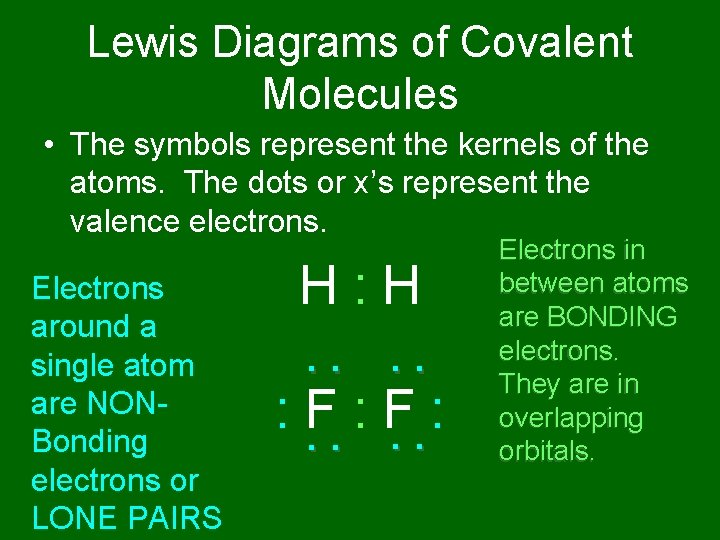
Lewis Diagrams of Covalent Molecules • The symbols represent the kernels of the atoms. The dots or x’s represent the valence electrons. : : H: H : : F: F: : Electrons around a single atom are NONBonding electrons or LONE PAIRS Electrons in between atoms are BONDING electrons. They are in overlapping orbitals.

Basic Rules for Lewis Diagrams 1. Count the valence electrons 2. Draw the skeleton, including all single bonds 3. Compare the # of electrons you need for each atom in the skeleton to have an octet (duet for H) and the # you have left to play with. 4. Distribute electrons / bonds as needed 5. Perform validity checks
- Slides: 35